Ex vivo gene therapy offers a potential one-time, curative solution for a host of genetic diseases. Image: Getty Images/ Perception7
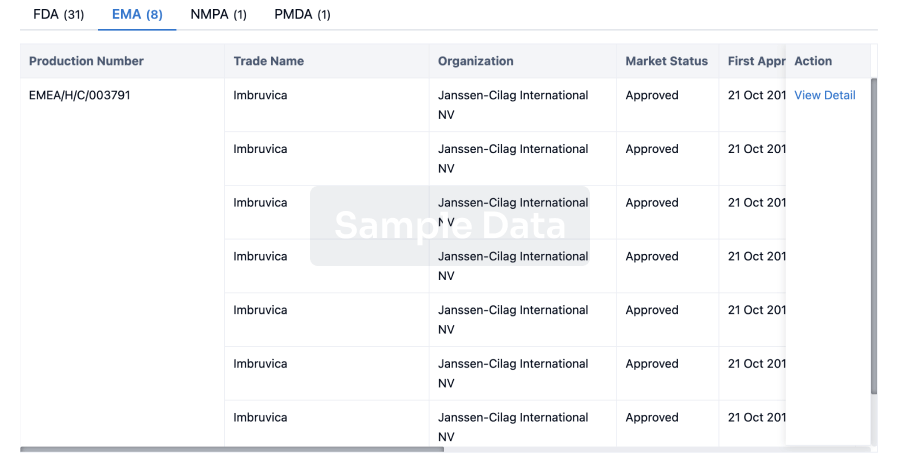
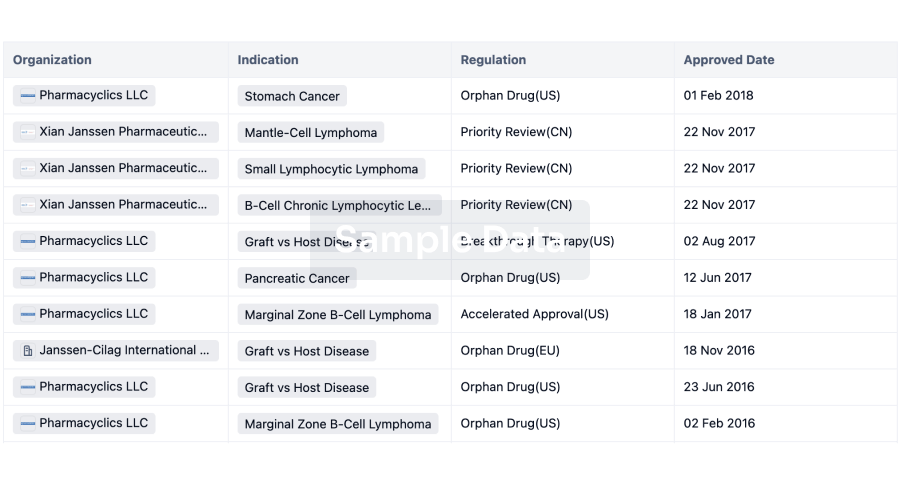
Bristol Myers Squibb
(BMS) has agreed to a research and licensing deal with Prime Medicine to develop ex vivo T cell therapies.
Under the terms of the deal announced yesterday (30 September), Massachusetts-based Prime Medicine will design gene editing reagents based on its proprietary PASSIGE technology with BMS taking these to develop ex vivo T cell therapies. Prime Medicine will receive a $55m upfront payment as well as $55m as an equity investment from BMS.
In addition, Prime could be eligible for over $3.5bn in milestone payments. These include up to $1.4bn from developmental milestones and over $2.1bn based on commercialisation. Prime will also be entitled to royalties on future sales.
The PASSIGE platform (Prime Assisted Site-Specific Integrase Gene Editing) combines prime editing with an integrase or other site-specific recombinase to stably insert and express genetic cargo in the genome of extracted cells. The technology does not utilise viral components or double-strand DNA breaks, intended to allow for precise genetic modification without off-target edits.
Prime’s lead candidate, PM-359, is currently in an open-label, single-arm Phase I/II clinical trial (NCT06559176) to treat chronic granulomatous disease (CGD). The cell therapy was granted orphan drug designation in January 2024 and an
Investigational New Drug
(IND) in April by the US Food and Drug Administration (FDA).
See Also:
mRNA Covid-19 vaccines may boost cancer immunotherapy effectiveness
J&J clinches EC approval for first single-pill PAH combo
In May 2024, Prime released
preclinical data
demonstrating that PM-359 precisely corrected CGD-causative mutations in more than 75% of patients’ CD34-positive cells. Furthermore, no off-target edits were detected in these cells post-engraftment.
Prime Medicine
launched
in 2021 with $315m in Series A and B financing. Since the company’s initial public offering in October 2022, its stock has steadily declined. Following the announcement of this deal, Prime’s share price opened at $4.23 on 30 September, 22% higher than at market close the previous Friday (27 September). The company’s market cap currently stands at $464.5m.
Also on 30 September, Prime unveiled a
strategically focussed pipeline
. This involved prioritising its candidate for Wilson’s disease, with an IND and potentially a clinical trial application (CTA) expected in 1H 2026. Additionally, the company said that data from the Phase I/II PM-359 trial could be released in 2025.
Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva
.
Editorial content is independently produced and follows the
highest standards
of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.
Free Whitepaper
Optimise your cell therapy process: a guide to cell thawing
Typically carried out at the point of care, errors in cell therapy thawing could compromise treatment efficacy, leading to significant patient impact as well as high costs and a compromised reputation for the product’s developer.
This guide addresses how cell thawing has historically developed into the new techniques used today, along with the physical and biological implications of key metrics and components such as warming rate and ice structure. Also included are reviews of key studies from scientific literature and a consideration of the interactions between cooling and warming rates, as applicable to cell and gene therapies.
Thank you.
You will receive an email shortly. Please check your inbox to download the Whitepaper.
By Cytiva Thematic
By downloading this Whitepaper, you acknowledge that GlobalData may share your information with
Cytiva Thematic
and that your personal data will be used as described in their
Privacy Policy