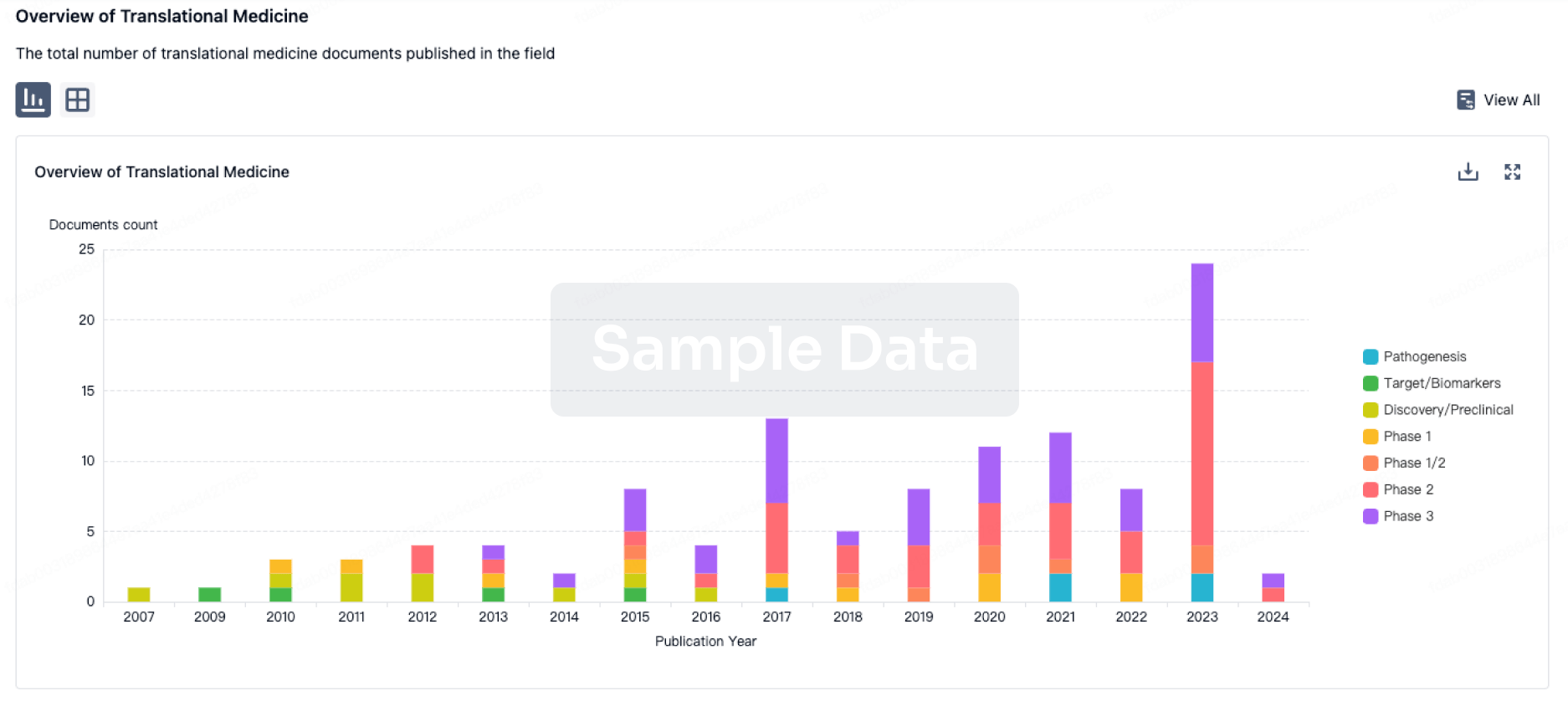
Recent publications by Chhabra et al.1 and Palchaudhuri et al.2 reported two novel approaches to safely opening marrow niches without using toxic alkylating agents or ionizing radiation (Figure 1).These studies in mouse models using clin. relevant reagents achieved high levels of donor cell chimerism with either congenic or allogeneic mismatched donor stem cells.Currently, the only clin. approach for creating marrow space for hematopoietic cell transplantation (HCT) that achieves significant donor chimerism requires an allogeneic setting and the use of moderate doses of alkylating agents or total body irradiation (TBI).This may be acceptable when treating patients with malignant diseases in which the ultimate goal is to ablate any residual tumor cells.However, for patients with nonmalignant disorders including hemoglobinopathies, primary immune deficiencies, inborn errors of metabolism, and marrow failure syndromes such as Fanconi anemia, any approach that avoids these toxic agents and their acute complications and long-term side effects would be a significant advance.Allogeneic HCT was initially used for a patient with severe combined immunodeficiency (SCID) and an HLA-matched sibling donor without any conditioning.Although chemoradiotherapy for most patients with typical SCID is not essential to achieve at least T-cell reconstitution,3,4 for all other diseases a conditioning or preparative regimen is required to prevent rejection of the donor cells and open marrow niches to allow engraftment of hematopoietic stem cells (HSCs).For patients with malignancies the goal is to achieve 100% donor chimerism.For many nonmalignant diseases such as the hemoglobinopathies, marrow failure syndromes, SCID, and some other primary immune deficiencies, only partial donor chimerism is necessary for disease correction.However, even in these cases, some reduced-intensity conditioning with lower doses of alkylating agents and/or TBI is required.There is a growing body of information regarding the marrow niche with respect to both the hematopoietic and the leukemic stem cell.5 Based on this information, agents have been evaluated that (i) directly attack the host stem cell, (ii) alter adhesion of the host stem cell in the niche, or (iii) enhance donor stem cell adhesion in the niche.The first exciting discovery, reported by Czechowicz et al.,6 was that the anti-murine c-Kit monoclonal antibody (ACK2) brought about significant engraftment of congenic donor cells in severely immunodeficient mice.Unfortunately, this approach was not effective in immune-competent mice, although synergy was observed between ACK2 and nonmyeloablative TBI.Cellular targeting of HSCs using sensitized donor T cells treated with psoralin and UVA light to prevent graft-vs.-host disease in murine SCID recipients of mismatched donor cells was also effective; multilineage engraftment was obtained, but graft-vs.-host disease could not be completely eliminated.7 Anti-CD45 monoclonal antibody administration along with immunosuppression was evaluated in one study of HCT for a variety of primary immunodeficiency diseases.8 Although sufficient engraftment was seen, it was difficult to determine the exact role of the antibody in determining outcome.Finally, a potentially attractive approach that has been evaluated in a dog model of autologous gene transfer is anti-CD45 conjugated to 211astatine, an α-emitter with a short path, high linear energy, and t1/2 of 7 h.9 Despite successful myelosuppression and minimal off-target effects, only minimal durable engraftment occurred, although the transduced cell dose was low.Among the initial approaches to targeting host HSC adhesion involved the use of AMD3100 (now known as plerixafor), a mol. that is an antagonist of the alpha-chemokine receptor CXCR4, which is important for HSC homing in the bone marrow and HSC quiescence.Unfortunately, though effective in opening niches to a limited degree in mouse models, it appears to be ineffective for HCT in SCID patients,10 and we have found it ineffective with or without ACK2 in a mouse model of Artemis-deficient SCID (unpublished results).Finally, two approaches to providing donor HSCs with an adhesion advantage over host HSCs have used either diprotin A, a CD26 inhibitor,11 or prostaglandin E2, a Wnt signaling agonist.Prostaglandin E2 has shown efficacy in murine and nonhuman primate models.12 This latter approach will be effective only in combination with other more direct measures as described above.In the first of the two studies, Chhabra et al.1 report on the synergy between ACK2 and CD47 antagonists in opening marrow niches.They found that the primary mechanism of ACK2-mediated HSC depletion is dependent on the Fc portion of the anti-c-Kit antibody and involves antibody-dependent cell-mediated cytotoxicity and phagocytosis (ADCC/ADCP).CD47 is a transmembrane protein expressed on many cells, including HSCs; it confers a "don't eat me" signal on the ADCC/ADCP process via its interaction with signal regulatory protein α (SIRPα) expressed on neutrophils and macrophages, representing one mechanism by which cancer cells are protected from phagocytosis.The investigators hypothesized that by blocking the "don't eat me" signal either with anti-mCD47 or engineered high-affinity fragments of human SIRPα, HSCs targeted by ACK2 would be even more susceptible to ADCC/ADCP.They found a greater than 10,000-fold reduction in the number of HSCs in treated wild-type mice.This was quite profound compared to ACK2 alone and involved not only HSCs but other hematopoietic progenitors; treated mice developed anemia and leukopenia.The effects were transient, with recovery of hematopoiesis by about three weeks.In an F1 → parent model, the investigators showed that ACK2 + CD4-SIRPα blockade achieved ∼60% multilineage engraftment compared with <5% with ACK2 alone.In a mismatched allogeneic transplant model, they found that by adding immunosuppression with anti-CD4 and anti-CD8, ACK2/CD47 blockade resulted in multilineage engraftment, although with only ∼15% donor chimerism.The main drawback to ACK2 alone has been that it was only effective in SCID mice and required the addition of low-dose TBI in T- and B-cell competent animals.7 The addition of a CD47 antagonist to anti-c-Kit has eliminated this requirement.A clin. trial of AMG191, an anti-human c-Kit monoclonal antibody, is planned in SCID patients undergoing allogeneic transplantation, and two anti-CD47 clin. trials are ongoing in patients with acute myeloid leukemia and solid tumors (ClinicalTrials.gov NCT02678338 and NCT02216409, resp.).Thus, the possibility of combining these two agents in a clin. trial of allogeneic transplantation in the future seems quite reasonable depending on the toxicity profiles that emerge from these trials.In mice, the principal toxicity was transient pancytopenia.However, c-Kit is expressed on other tissues, and treated mice turn gray and have reduced spermatogonia.One wonders whether other anti-c-Kit antibodies that bind to different epitopes of c-Kit might eliminate these off-target effects.The main concern regarding CD47 blockade is its effect on ADCC/ADCP and whether it will result in increased infections and/or whether it might be contraindicated in infected patients.In the second study, Palchaudhuri et al.2 used a different approach to opening marrow niches by combining an internalizing anti-CD45 monoclonal antibody with saporin (SAP), a catalytic N-glycosidase ribosome-inactivating protein that halts protein synthesis.They showed that this immunotoxin selectively depleted HSCs and hematopoietic progenitors.They found that mice pretreated with anti-CD45-SAP prior to transplant with 10 × 106 congenic whole bone marrow cells durably engrafted with 75-90% multilineage donor cells; 1 × 106 cells resulted in ∼20% chimerism.When 2,000 highly purified stem cells were used, ∼60% chimerism was achieved compared to virtually no chimerism when no pretreatment was given.They also compared the effects on bone marrow architecture and the thymus of nonmyeloablative doses of TBI vs. immunotoxin; TBI had a much greater adverse impact on the marrow and thymus.Furthermore, hematopoietic and T- and B-cell immune reconstitution appeared earlier in the immunotoxin-pretreated animals compared to those receiving TBI, and leukocyte, red blood cell, hematocrit, and Hb levels remained unaffected.Finally, using a knock-in mouse model of human sickle-cell disease the investigators showed that pretreatment with higher doses of anti-CD45-SAP followed by transplantation of 10 × 106 congenic wild-type bone marrow cells resulted in >90% durable chimerism with complete normalization of erythropoiesis.Although no significant off-target effects of this immunotoxin were apparent, clin. trials will be necessary to define potential toxicities.Allogeneic transplants were not evaluated in this study.The potential applications of either anti-c-Kit/CD47 blockade or anti-CD45-SAP, both of which target HSCs and appear to open marrow niches in mice quite effectively, are huge.Currently, autologous gene therapy for inborn errors of metabolism, hemoglobinopathies, and primary immune deficiencies all require varying degrees of conditioning with an alkylating agent (busulfan).For SCID it appears that low-dose exposure to busulfan is sufficient to achieve T- and B-cell reconstitution following autologous gene therapy.13,14 This may also be true for some other primary immunodeficiencies, but for the hemoglobinopathies and inborn errors of metabolism, ablative doses are necessary,15 thus eliminating at least one of the potential advantages of this approach unless adequate levels of chimerism can be achieved.It is also possible that combining these two approaches, which target the HSCs via different mechanisms, might achieve full donor chimerism.To eliminate the need for alkylating agents in the definitive treatment of these diseases would be a major breakthrough.For allogeneic transplantation, especially for nonmalignant disorders, eliminating alkylator therapy and/or TBI would significantly reduce major complications and side effects.Immunosuppression to prevent rejection would still be necessary, but with agents such as fludarabine replacing alkylators, acute and late effects would be minimized.Even for patients undergoing allogeneic transplantation for malignancies with these nonchemotherapeutic approaches, one could imagine being able to focus solely on rejection and more targeted anticancer therapy while minimizing the toxic effects of the standard conditioning regimens.Finally, for malignancies that express CD45 and/or anti-c-Kit, it might be possible that these novel approaches could replace alkylating chemotherapy and radiation altogether.