CN201 is a T cell engager bispecific antibody in development for the treatment of B cell non-Hodgkin lymphoma (B-NHL) and B cell acute lymphoblastic leukemia (B-ALL). Credits: Atmosphere1/Shutterstock.com
In a bid to further diversify its pipeline,
Merck
& Co (MSD) has announced that it is set to acquire B cell-targeting therapy, CN201, from Curon Biopharmaceutical.
As per the 9 August press release, MSD will purchase the clinical stage candidate through a subsidiary with an upfront cash payment of $700m. Under the terms of the agreement, Curon is eligible to receive up to $600m for developmental and regulatory milestones. MSD expects to complete the deal in Q3 2024.
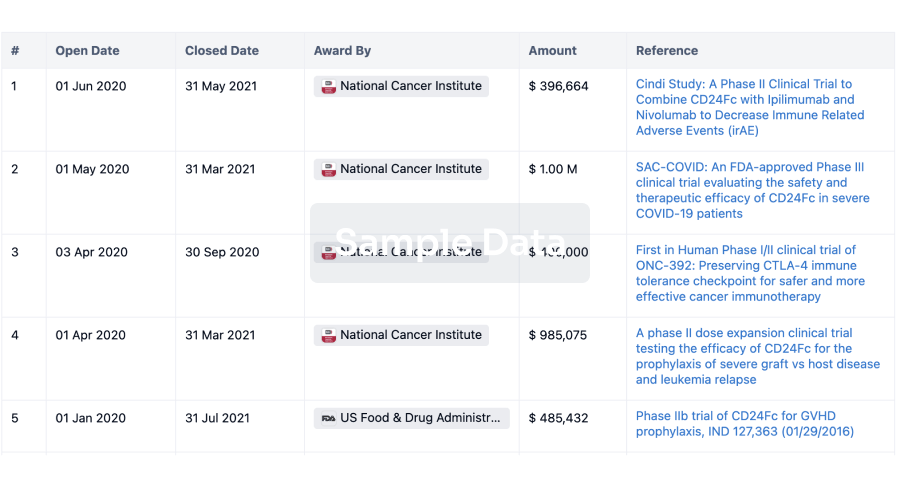
Intravenously administered CN201 is a CD3 and CD19-targeting T cell engager bispecific antibody designed to target B cells for elimination. The novel candidate is currently being evaluated in a Phase I study (NCT06189391) for the treatment of relapsed or refractory B cell non-Hodgkin lymphoma (B-NHL) and a Phase Ib/II study (NCT05579132) for the treatment of precursor B cell acute lymphoblastic leukemia (B-ALL).
MSD plans to assess this candidate’s potential in the treatment of B cell malignancies while also wading into the deeper waters with plans for evaluation in autoimmune diseases. MSD Research Laboratories president, Dr. Dean Li emphasised that early clinical data is “robust evidence” of CN201’s potential in treating a spectrum of malignant and autoimmune diseases.
Last month, the global pharma successfully
completed its $1.3bn acquisition
of EyeBio, a deal which gave MSD access to Restoret (EYE-103), a trispecific antibody designed to target the Wingless-related integration site (Wnt) signaling pathway. MSD plans to initiate a Phase IIb/III study of Restoret for the treatment of diabetic macular oedema (DME) in H2 2024.
See Also:
Eli Lilly boosts sales forecast by $3bn as diabetes and weight loss drugs propel revenue
Recursion and Exscientia merge to form drug discovery company
With MSD’s latest acquisition, the pharma giant is slowly but surely entering the bispecific antibody landscape, which used to be dominated by
Amgen
.
According to GlobalData healthcare analyst, Katarina Zahedi, GlobalData’s Pharma Intelligence Centre lists 1,147 ongoing and planned bispecific antibody clinical trials for oncology as of August 2024. Of these studies, Akeso Therapeutics leads with 52 trials with Amgen at the 8
th
place with 24 trials. Amgen previously
held the top spot in 2020
, running 19 of the 263 listed trials either ongoing or planned with a bispecific antibody.
However, Amgen continues to be a force for the development of bispecific T-cell engagers (BiTE), coming in behind Roche at 2
nd
place for the number of BiTE clinical studies.
Amgen’s trailblazing BiTE therapy, Blincyto (blinatumomab), is also designed to act as CD3xCD19-targeting T-cell engager. The therapy was first awarded US Food and Drug Administration (FDA) approval in 2014 for the treatment of ALL.
According to GlobalData’s consensus forecasts, Blincyto is expected to generate total sales of $2bn in 2030. GlobalData is the parent company of
Pharmaceutical Technology
.
GlobalData healthcare analysts expect that the bispecific antibody market for the treatment of cancer will reach $6bn globally by 2025.