Vital@Work: a Personalized Reintegration Program for Employees With Stress-related Complaints
The objective of this study, Vital@Work reintegration program, is to support sick-listed workers with stress-related complaints in their return to work based on a personalized program. Therefore, the aim of this study is test whether participants supported by a tailored eHealth program, and if needed, additionally supported by a structured and stepwise Participatory Approach (PA) involving the sick-listed worker, their direct supervisor and a neutral party, show a faster and sustainable return to work as compared to participants in the control condition. This program is investigated in four different organizations, which differ in sector, size (small and large organizations), type of organization (private or public) and type of work.
Vital@Work: an Evidence-based Intervention Program for Prevention of Mental Health Complaints at Work, Tailored to Specific Organisational Needs and Contextual Factors
The objective of the current study, Vital@Work, is to keep employees in the Netherlands vital on the long term by preventing work-related mental health problems. Therefore, we test an evidence based Participatory Approach (PA) to compose a mental health program tailored to behavioral, organizational & contextual (BOC) determinants of individuals and populations at risk. The PA is investigated in four different organizations, which differ in sector, size (small and large organizations), type of organization (private or public) and type of work. The varied organizations, each characterized by unique BOC-determinants, provide an opportunity for researching tailored intervention activities that aim to effectively prevent work-related mental health problems. By identifying common factors across these diverse organizations, we aim to grasp patterns of mechanisms for interventions to mental health at work. By doing this, we come closer to opening black box of how and why interventions work and thus closer to tailoring interventions to needs of both individuals and organizations
Potassium Correction for Renin-angiotensin-aldosterone System Optimization in Chronic Kidney Disease
The goal of this placebo-controlled, double-blinded cross-over trial is to test whether patiromer, compared with placebo, better enables up-titration of RAAS-blocker treatment in patients with chronic kidney disease stage 3b/4.
The main questions it aims to answer are:
Does patiromer allow uptitration of irbesartan, resulting in a significant reduction in albuminuria and blood pressure?
Does patiromer allow uptitration of irbesartan, resulting in a significant reduction in blood pressure?
The trial contains the following interventions:
Participants will be switched from their ACEi/ARB to a standardised dose of irbesartan (150 mg/d).
During two 12-week study periods, participants will receive either patiromer 8.4 g/d or placebo. The order of study periods is randomized.
At the start of each study period irbesartan will be up-titrated to 300 mg/d.
After 1 and 6 weeks, at both periods, plasma potassium will be measured and the irbesartan dose will be reduced to 150 mg/d in case plasma potassium exceeds 5.0 mmol/L.
At 12 weeks from the start of the study period, the endpoints will be assessed.
Between the two study periods, there is a 6-week washout. Irbesartan dose during the wash-out period will be 150mg/d. After washout, participants will switch from the patiromer arm to the placebo arm or vice versa.
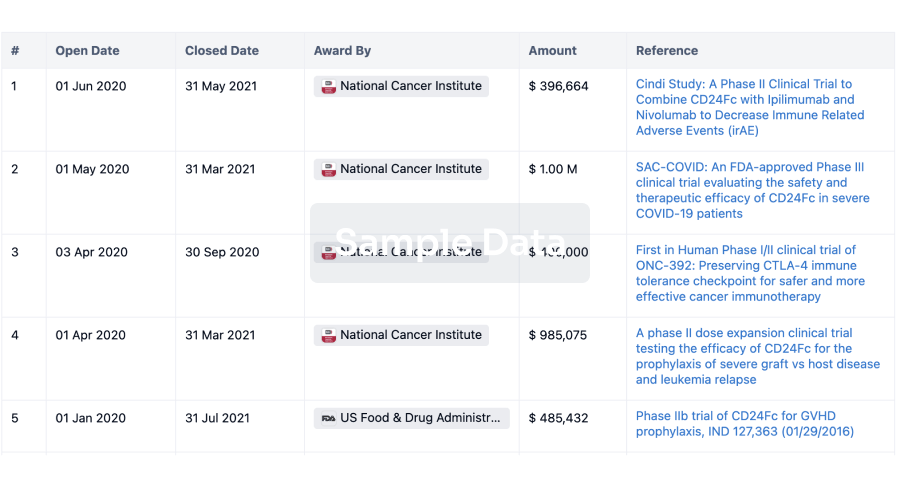
100 Clinical Results associated with Health-Holland
0 Patents (Medical) associated with Health-Holland
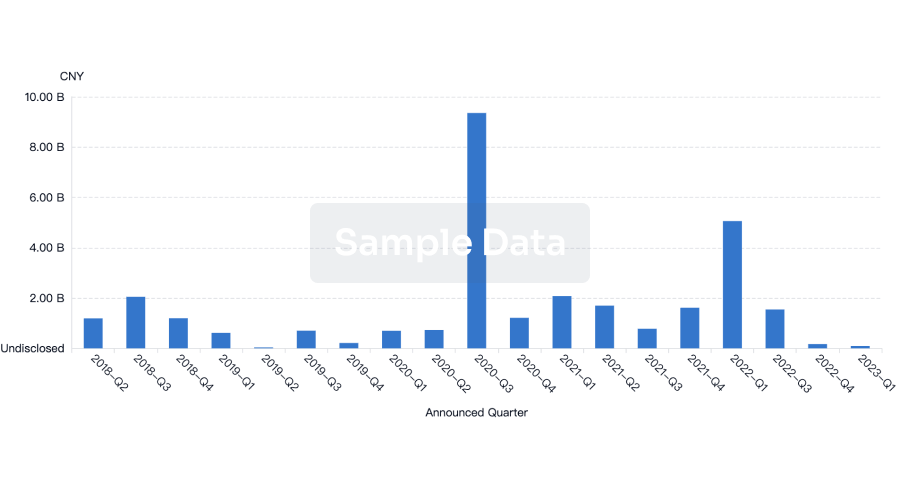
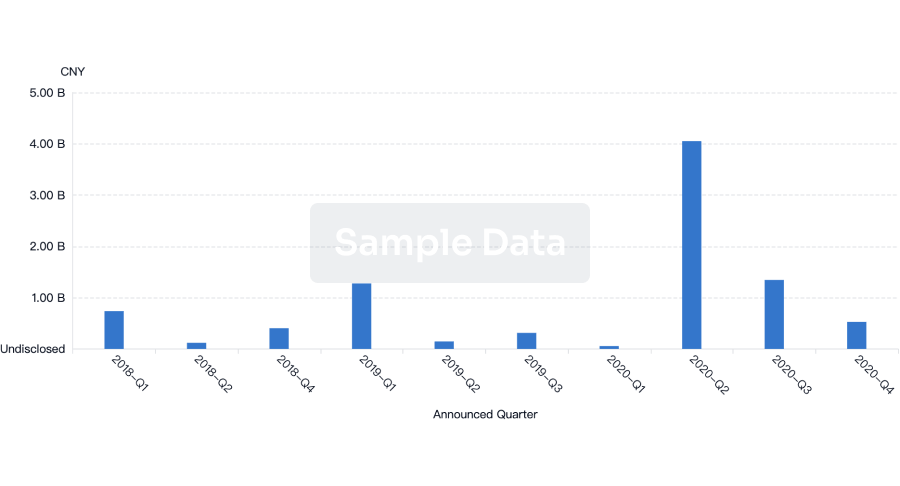
100 Deals associated with Health-Holland
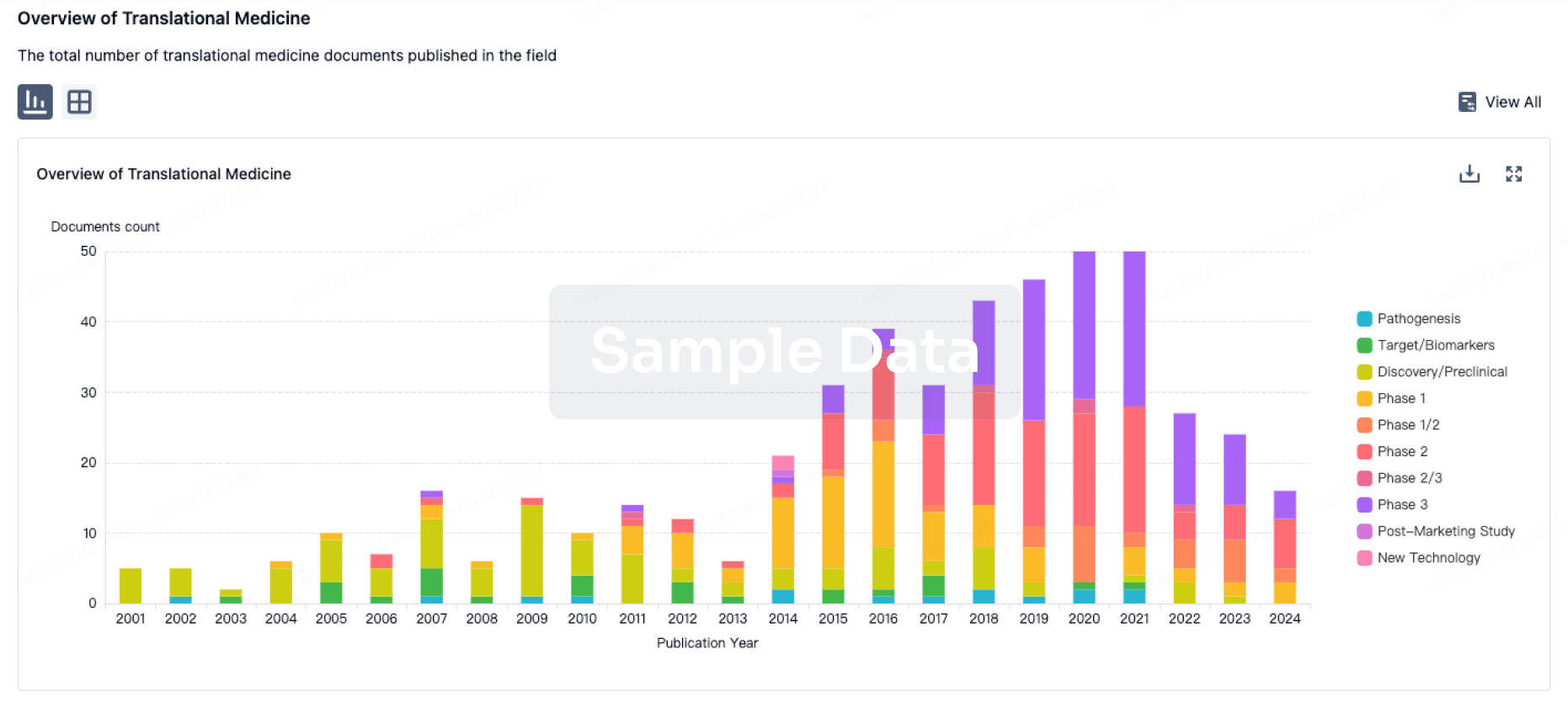
100 Translational Medicine associated with Health-Holland