Last update 19 Sep 2024
Janice M. Brown
Private Company|
North Carolina, United States
Private Company|
North Carolina, United States
Last update 19 Sep 2024
Overview
Related
100 Clinical Results associated with Janice M. Brown
Login to view more data
0 Patents (Medical) associated with Janice M. Brown
Login to view more data
1
News (Medical) associated with Janice M. Brown05 Nov 2013
510(K) SUMMARIES OR 510(K) STATEMENTS FOR FINAL
DECISIONS RENDERED DURING THE PERIOD October 2013
TOTAL 510(k)s THIS PERIOD 257 TOTAL WITH SUMMARIES 240
TOTAL WITH STATEMENTS 17
DEVICE: ACCESS TOTAL BHCG (5TH IS)
BECKMAN COULTER, INC. 510(k) NO: K130020(TRADITIONAL)
ATTN: GERALDINE L BAGLIEN PHONE NO : 952 368 7645
1000 LAKE HAZELTINE DR. SE DECISION MADE: 01-OCT-13
CHASKA MN 55318 1084 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ACE DIRECT TOTAL IRON-BINDING CAPACITY (TIBC) REAGENT, ACE TOTAL IRON REAGENT, ACE LDH-L REAGENT ALFA WASSERMANN DIAGNOSTIC TECHNOL510(k) NO: K131975(TRADITIONAL) ATTN: HYMAN KATZ PHONE NO : 973 852 0158 4 HENDERSON DRIVE SE DECISION MADE: 02-OCT-13 WEST CALDWELL NJ 07006 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INSTANT-VIEW MULTI-DRUG OF ABUSE URINE TEST (CUP, PANEL CASSETTE) ALFA SCIENTIFIC DESIGNS, INC. 510(k) NO: K131645(ABBREVIATED) ATTN: NAISHU WANG PHONE NO : 858 513 3888 13200 GREGG ST. SE DECISION MADE: 07-OCT-13 POWAY CA 92064 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VIDAS C DIFFICILE GDH BIOMERIEUX SA 510(k) NO: K132010(TRADITIONAL) ATTN: CAROLINE KOCH-MATHIAN PHONE NO : 33 478877 032 5 RUE DES AQUEDUCS SE DECISION MADE: 09-OCT-13 CRAPONNE FR 69290 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VITEK 2 AST-ST TIGECYCLINE BIOMERIEUX, INC. 510(k) NO: K131779(TRADITIONAL) ATTN: NATHAN HARDESTY PHONE NO : 314 731 8666 595 ANGLUM RD. SE DECISION MADE: 10-OCT-13 HAZELWOOD MO 63042 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ACCU-CHEK AVIVA EXPERT BLOOD GLUCOSE MONITORING SYSTEM ROCHE DIAGNOSTICS OPERATIONS INC 510(k) NO: K131366(TRADITIONAL) ATTN: NATHAN CARRINGTON PHONE NO : 317 521 4793 9115 Hague Rd SE DECISION MADE: 11-OCT-13 Indianapolis IN 46250 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMDX C.DIFFICILE FOR ABBOTT M2000 INTELLIGENT MEDICAL DEVICES, INC. 510(k) NO: K132235(TRADITIONAL) ATTN: FRAN WHITE PHONE NO : 978 927 3808 180 CABOT ST SE DECISION MADE: 11-OCT-13 BEVERLY MA 01915 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ADVIA CHEMISTRY ALBUMIN BCP REAGENT (ALBP), ADVIA CHEMISTRY ALBUMIN BCP CALIBRATOR SIEMENS HEALTHCARE DIAGNOSTICS, IN510(k) NO: K132664(TRADITIONAL) ATTN: KIRA GORDON PHONE NO : 914 524 2996 511 BENEDICT AVE SE DECISION MADE: 16-OCT-13 TARRYTOWN NY 10591 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FOCUS BLOOD GLUCOSE MONITORING SYSTEM, FOCUS PRO BLOOD GLUCOSE MONITORING SYSTEM NOVA BIOMEDICAL CORPORATION 510(k) NO: K131013(TRADITIONAL) ATTN: PAUL W MACDONALD, PHD PHONE NO : 781 894 0800 200 Prospect St SE DECISION MADE: 17-OCT-13 WALTHAM MA 02454 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MRSA/SA ELITE MGB, ELITE MGB SOFTWARE ELITECHGROUP EPOCH BIOSCIENCES 510(k) NO: K132468(TRADITIONAL) ATTN: DEBRA K HUTSON PHONE NO : 425 482 5555 21720 23rd Dr SE, Suite 150 SE DECISION MADE: 17-OCT-13 Bothell WA 98021 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: APTIMA COMBO 2 ASSAY (PANTHER SYSTEM) HOLOGIC / GEN-PROBE INCORPORATED 510(k) NO: K132251(TRADITIONAL) ATTN: PHONE NO : 10210 GENETIC CENTER DRIVE SE DECISION MADE: 17-OCT-13 SAN DIEGO CA 92121 4376 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: QUICKPROFILE SINGLE DRUG OF ABUSE DEVICE AND QUICK PROFILE MULTI-DRUGS OF ABUSE SCREEN DEVICE LUMIQUICK DIAGNOSTICS, INC. 510(k) NO: K130213(TRADITIONAL) ATTN: FENG-YU LEE PHONE NO : 949 951 3046 27001 LA PAZ RD, STE 266B SE DECISION MADE: 18-OCT-13 MISSION VIEJO CA 92691 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RAPIDFRET ORAL FLUID ASSAY FOR THC, THC CALIBRATOR SET, THC CONTROL SET, COLLECTOR BIOPHOR DIAGNOSTICS, INC. 510(k) NO: K132096(TRADITIONAL) ATTN: NATHANIEL G BUTLIN PHONE NO : 650 367 4954 1201 DOUGLAS AVE SE DECISION MADE: 18-OCT-13 REDWOOD CITY CA 94063 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOPLEX 2200 APLS IGM BIO-RAD LABORATORIES 510(k) NO: K130528(TRADITIONAL) ATTN: JUANG WANG PHONE NO : 510 741 4609 5500 E. 2ND STREET SE DECISION MADE: 21-OCT-13 BENICIA CA 94510 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMMULISA ENHANCED CELIAC FUSION (TTG/DGP) IGA/IGG ANTIBODY ELISA IMMCO DIAGNOSTICS, INC. 510(k) NO: K123713(TRADITIONAL) ATTN: KEVIN J LAWSON PHONE NO : 716 691 0091 60 PINEVIEW DR. SE DECISION MADE: 25-OCT-13 BUFFALO NY 14228 2120 510(k) STATEMENT
DEVICE: S TEST REAGENT CARTRIDGE BUN AND S TEST REAGENT CARTRIDGE CRE
HITACHI CHEMICAL DIAGNOSTICS, INC.510(k) NO: K132462(TRADITIONAL)
ATTN: ERIKA AMMIRATI PHONE NO : 650 949 2768
575 SHIRLYNN COURT SE DECISION MADE: 28-OCT-13
LOS ALTOS CA 94022 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PATIENT MONITOR SHENZHEN CREATIVE INDUSTRY CO., LT510(k) NO: K123711(TRADITIONAL) ATTN: CHARLIE MACK PHONE NO : 931 625 4938 12226 WASHINGTON LANE SE DECISION MADE: 01-OCT-13 PARKER AZ 85344 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MPACT DM LINER CONVERTER MEDACTA INTERNATIONAL 510(k) NO: K131458(TRADITIONAL) ATTN: ADAM GROSS PHONE NO : 805 322 3289 4725 CALLE QUETZAL, UNIT B SE DECISION MADE: 01-OCT-13 CAMARILLO CA 93012 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRINITY MICROLIFT FACIAL TONING DEVICE CAROL COLE COMPANY 510(k) NO: K131251(TRADITIONAL) ATTN: RAND DAOUD PHONE NO : 760 734 4545 1325 Sycamore Ave Ste A SE DECISION MADE: 01-OCT-13 Vista CA 92081 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FACIAL PREBENT PLATING WITH VIRTUAL SURGICAL PLANNING BIOMET MICROFIXATION 510(k) NO: K132600(SPECIAL) ATTN: ELIZABETH WHEELER PHONE NO : 904 741 4400 1520 TRADEPORT DR. SE DECISION MADE: 02-OCT-13 JACKSONVILLE FL 32218 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BD VERITOR SYSTEM FOR THE RAPID DETECTION OF FLU A+B BECTON, DICKINSON AND COMPANY 510(k) NO: K132693(SPECIAL) ATTN: GREGORY P PAYNE, RAC PHONE NO : 858 795 7890 10865 ROAD TO THE CURE SUITE 200 SE DECISION MADE: 02-OCT-13 SAN DIEGO CA 92121 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FINGERTIP PULSE OXIMETER BEIJING CHOICE ELECTRONIC TECHNOLO510(k) NO: K130947(TRADITIONAL) ATTN: LEI CHEN PHONE NO : 86 10 88798300 No.9 Shuangyuan Road, Badachu Hi-tSE DECISION MADE: 03-OCT-13 BEIJING CN 100041 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DISPOSABLE SILICONE FOLEY CATHETER GUANGDONG BAIHE MEDICAL TECHNOLOGY510(k) NO: K130908(TRADITIONAL) ATTN: DIANA HONG PHONE NO : 86 21 22815850 PO BOX 120-119 SE DECISION MADE: 03-OCT-13 SHANGHAI CH 200120 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CLEANSER ROOT CANAL CERKAMED WOJCIECH PAWLOWSKI 510(k) NO: K130180(TRADITIONAL) ATTN: KINGA WIERUCKA PHONE NO : 481 584235 85 KWIATKOWSKIEGO 1 STREET SE DECISION MADE: 03-OCT-13 STALOWA WOLA, PODKARPACIE PL 37510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LEDEX DENTMATE TECHNOLOGY CO., LTD. 510(k) NO: K131042(TRADITIONAL) ATTN: CHENG-FENG CHOU PHONE NO : 866 289769 226 8F, No.8-11, Sec.1, Zhongxing RoadSE DECISION MADE: 03-OCT-13 New Taipei City TW 24872 510(k) STATEMENT
DEVICE: EVEREST SPINAL SYSTEM K2M, INC. 510(k) NO: K132361(TRADITIONAL) ATTN: NANCY GIEZEN PHONE NO : 703 777 3155 751 MILLER DR.,SE SE DECISION MADE: 03-OCT-13 LEESBURG VA 20175 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MOTEX POWDER-FREE NITRILE SURGICAL GLOVES SHANGHAI MOTEX HEALTHCARE CO., LTD510(k) NO: K122557(TRADITIONAL) ATTN: JEN KE-MIN PHONE NO : 862 159 799888 NO. 369, JIASONG ZHONG ROAD HUAXINSE DECISION MADE: 03-OCT-13 SHANGHAI CH 201708 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OCCLUSION PERFUSION CATHETER ADVANCED CATHETER THERAPIES, INC. 510(k) NO: K130525(TRADITIONAL) ATTN: CHRIS HENZA PHONE NO : 262 842 1257 7401 104TH AVE SE DECISION MADE: 03-OCT-13 KENOSHA WI 53142 510(k) STATEMENT
DEVICE: NEO KNEE SYSTEM PIPELINE ORTHOPEDICS 510(k) NO: K131368(TRADITIONAL) ATTN: TERRY POWELL PHONE NO : 703 562 9800 901 KING STREET, SUITE 200 SE DECISION MADE: 03-OCT-13 ALEXANDRIA VA 22314 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AMCAD-UT DETECTION MODEL 2.0 LIN & ASSOCIATES, LLC. 510(k) NO: K122536(TRADITIONAL) ATTN: CHIU S LIN PHONE NO : 301 591 3895 9223 CAMBRIDGE MANOR COURT SE DECISION MADE: 03-OCT-13 POTOMAC MD 20854 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DICOM VIDEO BAR CODE COMPUTER LTD 510(k) NO: K132183(TRADITIONAL) ATTN: DAN LAOR PHONE NO : 972 4 8246632 6 SIRENI SE DECISION MADE: 03-OCT-13 HAIFA IS 32972 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ENSEAL G2 TISSUE SEALER, STRAIGHT JAW, ENSEAL G2 TISSUE SEALER, CURVED JAW, ENSEAL TRIO TISSUE SEALER, 3 MM CURVED JAW, ETHICON ENDO-SURGERY, LLC 510(k) NO: K131435(TRADITIONAL) ATTN: MARICA ARENTZ PHONE NO : 513 337 1066 4545 CREEK ROAD SE DECISION MADE: 03-OCT-13 CINCINNATI OH 45242 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ANYSCAN-S SPECT IMAGING SYSTEM ABSOLUTE IMAGING SOLUTIONS 510(k) NO: K131625(TRADITIONAL) ATTN: MARK SHINA PHONE NO : 336 643 2000 8205 B&G COURT SE DECISION MADE: 03-OCT-13 STOKESDALE NC 27357 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NATUS WARMETTE BLANKET AND DUAL CABINETS NATUS NICOLET IRELAND LTD 510(k) NO: K132110(TRADITIONAL) ATTN: MICHAEL GALVIN PHONE NO : 353 091 647451 IDA BUSINESS PARK SE DECISION MADE: 03-OCT-13 GORT, COUNTY GALWAY EI IE 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AVAFLEX VERTEBRAL BALLOON SYSTEM CAREFUSION 510(k) NO: K131824(TRADITIONAL) ATTN: JOY GREIDANUS PHONE NO : 847 473 7404 75 NORTH FAIRWAY DRIVE SE DECISION MADE: 03-OCT-13 VERNON HILLS IL 60061 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PICCOLO COMPOSITE NAILING SYSTEM CARBOFIX ORTHOPEDICS LTD. 510(k) NO: K132774(SPECIAL) ATTN: YAEL RUBIN PHONE NO : 972 9 9511511 11 HA’HOSHLIM STREET SE DECISION MADE: 03-OCT-13 HERZELIYA IS 46724 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BEAUTIFIL BULK FLOWABLE SHOFU DENTAL CORPORATION 510(k) NO: K132141(TRADITIONAL) ATTN: DAVID P MORAIS PHONE NO : 760 736 3277 1225 STONE DRIVE SE DECISION MADE: 03-OCT-13 SAN MARCOS CA 92078 510(k) STATEMENT
DEVICE: DEROYAL ELECTROSURGICAL PENCIL, ROCKER STYLE WITHOUT HOLSTER, DEROYAL ELECTROSURGICAL PENCIL, BUTTON STYLE WITHOUT HOLST DEROYAL INDUSTRIES, INC. 510(k) NO: K132199(TRADITIONAL) ATTN: COURTNEY RINEHART PHONE NO : 865 362 2122 200 DEBUSK LN. SE DECISION MADE: 03-OCT-13 POWELL TN 37849 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AAP LOQTEQ PROXIMAL MEDIAL TIBIA PLATE 3.5 AAP IMPLANTATE AG 510(k) NO: K132554(TRADITIONAL) ATTN: CHRISTIAN ZIETSCH PHONE NO : 004 930 75019193 LORENZWEG 5 SE DECISION MADE: 03-OCT-13 BERLIN GM 12099 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MODIFIED FLOWGATE BALLOON GUIDE CATHETER CONCENTRIC MEDICAL, INC. 510(k) NO: K131492(TRADITIONAL) ATTN: RHODA SANTOS PHONE NO : 510 413 2269 301 E Evelyn Ave SE DECISION MADE: 03-OCT-13 MOUNTAIN VIEW CA 94041 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PASS LP SPINAL SYSTEM MEDICREA INTERNATIONAL 510(k) NO: K132575(SPECIAL) ATTN: AUDREY VION PHONE NO : 33 47 2018787 14 Porte du Grand Lyon SE DECISION MADE: 03-OCT-13 NEYRON FR 01700 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: EMBOZENE MICROSPHERES CELONOVA BIOSCIENCES, INC. 510(k) NO: K132675(SPECIAL) ATTN: NICOLE BARBER PHONE NO : 210 497 6682 18615 Tuscany Stone Ste 100 SE DECISION MADE: 03-OCT-13 San Antonio TX 78258 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NAUTILUS SPINAL SYSTEM LIFE SPINE, INC 510(k) NO: K132760(SPECIAL) ATTN: RANDY LEWIS PHONE NO : 847 884 6117 2401 W. HASSELL RD SUITE 1535 SE DECISION MADE: 03-OCT-13 HOFFMAN ESTATES IL 60169 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MAGNIFUSE BONE GRAFT, MAGIFUSE II BONE GRAFT MEDTRONIC SOFAMOR DANEK USA, INC. 510(k) NO: K131673(TRADITIONAL) ATTN: KELLY ANGLIN PHONE NO : 901 396 3133 1800 PYRAMID PLACE SE DECISION MADE: 03-OCT-13 Memphis TN 38132 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: THERMOCHEM HT-2000 THERMASOLUTIONS, INC. 510(k) NO: K131583(TRADITIONAL) ATTN: M.W. ANDY ANDERSON PH.D. RAPHONE NO : 952 746 8079 5353 WAYZATA BOULEVARD SUITE 505 SE DECISION MADE: 03-OCT-13 MINNEAPOLIS MN 55416 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: COLONGLIDE(R) LUBRICANT PEDIATRIC PHARMACEUTICALS, INC. 510(k) NO: K131617(TRADITIONAL) ATTN: ROBERT M HARRIS PHONE NO : 732 739 2018 2137 ROUTE 35 SE DECISION MADE: 03-OCT-13 HOLMDEL NJ 07733 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LOCKING PLATE SYSTEM BEIJING LIBEIER BIO-ENGINEERING IN510(k) NO: K132642(TRADITIONAL) ATTN: DIANA HONG PHONE NO : 86 21 22815850 P.O. BOX 120-119 SE DECISION MADE: 03-OCT-13 SHANGHAI CH 200120 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AVAMAX VERTEBRAL BALLOON SYSTEM, 8G, AVAMAX VERTEBRAL BALLOON SYSTEM, 10G, AVAMAX VERTEBRAL BALLOON SYSTEM, 11G CAREFUSION 510(k) NO: K131820(TRADITIONAL) ATTN: JOY GREIDANUS PHONE NO : 847 473 7404 75 NORTH FAIRWAY DRIVE SE DECISION MADE: 04-OCT-13 VERNON HILLS IL 60061 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ISCHEMA VIEW RAPID ISCHEMA VIEW, INC. 510(k) NO: K121447(TRADITIONAL) ATTN: MARC GOODMAN PHONE NO : 949 548 3980 5405 ALTON PARKWAY 5A, #530 SE DECISION MADE: 04-OCT-13 IRVINE CA 92604 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INNER PEACE KAMABRY, INC., DBA INNER PEACE FOR510(k) NO: K122800(TRADITIONAL) ATTN: YOLANDA SMITH PHONE NO : 888 729 9674 1468 HARWELL HAVE SE DECISION MADE: 04-OCT-13 CROFTON MD 21114 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PATHFAST CK-MB-II CALIBRATORS MITSUBISHI CHEMICAL MEDIENCE CORPO510(k) NO: K130628(SPECIAL) ATTN: JUDI SMITH PHONE NO : 240 316 3377 2 BETHESDA METRO CENTER SUITE 850 SE DECISION MADE: 04-OCT-13 BETHESDA MD 20814 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: UGEO H60 DIAGNOSTIC ULTRASOUND SYSTEM SAMSUNG MEDISON CO., LTD 510(k) NO: K132861(TRADITIONAL) ATTN: KYEONG-MI PARK PHONE NO : 82 2 21941373 42, TEHERAN-RO 108-GIL GANGNAM-GU SE DECISION MADE: 04-OCT-13 SEOUL KS 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: THORATRAK MICS RETRACTOR SYSTEM, RACK, BLADES MEDTRONIC, INC. 510(k) NO: K132645(TRADITIONAL) ATTN: CHELSEA PIOSKE PHONE NO : 763 514 9838 7611 NORTHLAND DRIVE SE DECISION MADE: 04-OCT-13 MINNEAPOLIS MN 55428 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: XPRESS MULTI-SINUS DILATION TOOL ENTELLUS MEDICAL, INC. 510(k) NO: K132440(SPECIAL) ATTN: KAREN E PETERSON PHONE NO : 763 463 7066 3600 HOLLY LANE NORTH SUITE 40 SE DECISION MADE: 04-OCT-13 PLYMOUTH MN 55447 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: STAXX IBL SYSTEM SPINE WAVE, INC. 510(k) NO: K132719(SPECIAL) ATTN: ROAIDA RIZKALLAH PHONE NO : 203 944 9494 3 ENTERPRISE DR. SUITE 210 SE DECISION MADE: 04-OCT-13 SHELTON CT 06484 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: S8 EXP PORTABLE DIGITAL COLOR DOPPLER ULTRASOUND SYSTEM SONOSCAPE COMPANY LIMITED 510(k) NO: K132768(SPECIAL) ATTN: TOKI WU PHONE NO : 86 755 26722890 YIZHE BLDG, YUQUAN ROAD SE DECISION MADE: 04-OCT-13 SHENZHEN, GUANGDONG CH 518051 P510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: 200 MICRON BLOOD COMPONENT FILTER AND SYRINGE ADAPTER ARTERIOCYTE MEDICAL SYSTEMS, INC. 510(k) NO: K132905(TRADITIONAL) ATTN: ANN CHAREST PHONE NO : 508 497 8964 45 SOUTH STREET SE DECISION MADE: 04-OCT-13 HOPKINTON MA 01748 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: MICROPLEX COIL SYSTEM (MCS) MICROVENTION, INC. 510(k) NO: K131948(SPECIAL) ATTN: LARAINE PANGELINA PHONE NO : 714 247 8150 1311 VALENCIA AVE SE DECISION MADE: 04-OCT-13 TUSTIN CA 92780 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SINUS DILATION SYSTEM WITH CANNULATED INSTRUMENT ARTHROCARE CORP. 510(k) NO: K131709(SPECIAL) ATTN: ASHLEY DAWSON PHONE NO : 512 358 5773 7000 W.WILLIAM CANNON DRIVE BUILDISE DECISION MADE: 04-OCT-13 AUSTIN TX 78735 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VITAHEAT PATIENT WARMING SYSTEM VITAHEAT MEDICAL, LLC 510(k) NO: K132454(TRADITIONAL) ATTN: JACK SLOVICK PHONE NO : 763 639 0238 1585 BEVERLY DR. #121 SE DECISION MADE: 04-OCT-13 AURORA IL 60502 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: SMARTSITE VIALSHIELD YUKON MEDICAL, LLC 510(k) NO: K132863(TRADITIONAL) ATTN: PHONE NO : 2200 Gateway Centre Blvd Ste 208 SE DECISION MADE: 04-OCT-13 Morrisville NC 27560 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: KYMCO EQ20C SCOOTER KWANG YANG MOTOR CO., LTD. 510(k) NO: K130940(TRADITIONAL) ATTN: JUNNATA CHANG PHONE NO : 886 4 22465396 16F-2 (16A), NO. 462, SEC. 2, CHONSE DECISION MADE: 07-OCT-13 BEITUN DIST., TAICHUNG TW 406 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ENTUIT SECURE GASTROINTESTIONAL SUTURE ANCHOR SET, ENTUIT SECURE ADJUSTABLE GASTROINTESTINAL SUTURE ANCHOR SET COOK, INC. 510(k) NO: K131201(TRADITIONAL) ATTN: ERUM B NASIR PHONE NO : 812 339 2235 750 DANIELS WAY SE DECISION MADE: 07-OCT-13 Blmgtn IN 47404 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VISI MOBILE MONITORING SYSTEM SOTERA WIRELESS, INC 510(k) NO: K130709(TRADITIONAL) ATTN: EBEN GORDON PHONE NO : 858 373 4841 9444 WAPLES STREET SE DECISION MADE: 07-OCT-13 SAN DIEGO CA 92121 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRELLIS COLLAGEN RIBBON WRIGHT MEDICAL TECHNOLOGY, INC. 510(k) NO: K131143(TRADITIONAL) ATTN: SARAH HOLTGREWE PHONE NO : 901 867 4476 5677 AIRLINE RD. SE DECISION MADE: 07-OCT-13 ARLINGTON TN 38002 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CINCHLOCK KNOTLESS SUTURE ANCHOR, NANOTACK SUTURE ANCHOR 1.4MM PIVOT MEDICAL INC. 510(k) NO: K131769(TRADITIONAL) ATTN: JON COOK PHONE NO : 408 774 1452 247 HUMBOLDT CT SE DECISION MADE: 07-OCT-13 SUNNYVALE CA 94089 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CAREGIVER PROFESSIONAL CLINICAL THERMOMETER, PRO-TF SERIES THERMOMEDICS INC. 510(k) NO: K131771(TRADITIONAL) ATTN: PAUL LIU PHONE NO : 886 26625 8188X6134 6F, NO. 127, WUGONG 2ND RD., WUGU SE DECISION MADE: 07-OCT-13 NEW TAIPEI CITY TW 24888 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ESOFLIP BALLOON DILATION CATHETER CROSPON, LTD. 510(k) NO: K132337(TRADITIONAL) ATTN: PAUL DRYDEN PHONE NO : 239 498 2155 24301 WOODSAGE DRIVE SE DECISION MADE: 07-OCT-13 BONITA SPRINGS FL 34134 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: HEM-6221 OMRON HEALTHCARE, INC. 510(k) NO: K131749(TRADITIONAL) ATTN: PAUL DRYDEN PHONE NO : 239 498 2155 24301 WOODSAGE DRIVE SE DECISION MADE: 07-OCT-13 BONITA SPRINGS FL 34134 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: V8 SP NORMAL CONTROL, ABNORMAL CONTROL HELENA LABORATORIES 510(k) NO: K131479(TRADITIONAL) ATTN: JUSTIN PADIA PHONE NO : 409 842 3714 1530 LINDBERGH DR. SE DECISION MADE: 07-OCT-13 BEAUMONT TX 77704 510(k) STATEMENT
DEVICE: ESTECH COBRA(R) GENESIS BIPOLAR CLAMP ESTECH, INC. 510(k) NO: K133017(SPECIAL) ATTN: TAMER IBRAHIM PHONE NO : 925 543 2110 2603 CAMINO RAMON, STE 100 SE DECISION MADE: 07-OCT-13 SAN RAMON CA 94583 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PHILIPS PIXIUM 4343RC PHILIPS MEDICAL SYSTEMS DMC GMBH 510(k) NO: K131483(TRADITIONAL) ATTN: JENNIFER CARTLEDGE PHONE NO : 864 500 0097 409 WOODRIDGE DR. SE DECISION MADE: 07-OCT-13 SENECA SC 29672 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: UNIMED SURGICAL COATED ELECTROSURGICAL ELECTRODES UNIMED SURGICAL PRODUCTS, INC. 510(k) NO: K132224(TRADITIONAL) ATTN: LEE ALEXANDER PHONE NO : 727 546 1900 10401 BELCHER RD. SE DECISION MADE: 08-OCT-13 LARGO FL 33777 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: COMPREHENSIVE REVERSE SHOULDER – TITANIUM GLENOSPHERE BIOMET MANUFACTURING CORP. 510(k) NO: K131353(TRADITIONAL) ATTN: PATRICIA S BERES PHONE NO : 574 267 6639 56 EAST BELL DRIVE SE DECISION MADE: 08-OCT-13 WARSAW IN 46582 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: APEX KNEE MODULAR TIBIA SYSTEM OMNI LIFE SCIENCE, INC. 510(k) NO: K131472(TRADITIONAL) ATTN: BRANDON MOLINA PHONE NO : 774 226 1815 50 O’CONNELL WAY SE DECISION MADE: 08-OCT-13 EAST TAUNTON MA 02718 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: R7 CEFLA S.C. 510(k) NO: K130834(TRADITIONAL) ATTN: CLAUDE BERTHOIN PHONE NO : 386 672 2630 110 E. GRANADA BLVD. SUITE 207 SE DECISION MADE: 08-OCT-13 ORMOND BEACH FL 32176 510(k) STATEMENT
DEVICE: WECK EFX ENDO FASCIAL CLOSURE SYSTEM TELEFLEX MEDICAL, INC. 510(k) NO: K132362(TRADITIONAL) ATTN: NATALIE HICHAK PHONE NO : 919 361 4071 2917 WECK DR. SE DECISION MADE: 08-OCT-13 RESEARCH TRIANGLE PARK NC 27709 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SYNTHES 2.7MM AND 3.5MM VARIABLE ANGLE LCP MIDFOOT/HINDFOOT SYSTEM SYNTHES (USA) PRODUCTS LLC 510(k) NO: K131186(TRADITIONAL) ATTN: REBECCA G REITER PHONE NO : 610 719 1268 1301 GOSHEN PKWY. SE DECISION MADE: 08-OCT-13 WEST CHESTER PA 19380 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIODESIGN CRANIAL BASE DURAL GRAFT COOK BIOTECH, INC. 510(k) NO: K131015(TRADITIONAL) ATTN: MARY A FADERAN PHONE NO : 765 497 3355 1425 INNOVATION PLACE SE DECISION MADE: 08-OCT-13 West Lafayette IN 47906 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MEDDUSA BIPOLAR SYSTEM ELLIQUENCE LLC 510(k) NO: K130110(TRADITIONAL) ATTN: OSCAR R PAREDES PHONE NO : 516 277 9024 2455 Grand Avenue SE DECISION MADE: 08-OCT-13 Baldwin NY 11510 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INMODE WMBODY DEVICE INMODE MD LTD. 510(k) NO: K131362(TRADITIONAL) ATTN: AHAVA STEIN PHONE NO : 972 976700 02 20 HATA’AS STR., SUITE 102 SE DECISION MADE: 08-OCT-13 KFAR SABA IS 44425 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PRONTOSAN WOUND GEL X B. BRAUN MEDICAL, INC. 510(k) NO: K130857(TRADITIONAL) ATTN: NANCY SKOCYPEC PHONE NO : 610 266 0500 901 MARCON BLVD. SE DECISION MADE: 08-OCT-13 ALLENTOWN PA 18109 9341 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: QUADROX-I, QUADROX-ID, QUADROX-IR OXYGENATORS MAQUET CARDIOPULMONARY AG 510(k) NO: K132829(SPECIAL) ATTN: WHITNEY TORNING PHONE NO : 973 709 7994 45 BARBOUR POND DRIVE SE DECISION MADE: 08-OCT-13 WAYNE NJ 07470 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INTEL-GE CARE INNOVATIONS QUIETCARE INTEL-GE CARE INNOVATIONS LLC 510(k) NO: K131585(TRADITIONAL) ATTN: MAUREEN GLYNN PHONE NO : 916 847 7794 1721 DOUGLAS BLVD SUITE 100 SE DECISION MADE: 08-OCT-13 ROSEVILLE CA 95661 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NEUTRAL LUER ACTIVATED DEVICE (ONE-LINK NEEDLE-FREE IV CONNECTOR) AND EXTENSION SETS BAXTER HEALTHCARE CORP. 510(k) NO: K132734(SPECIAL) ATTN: GARY CHUMBIMUNE PHONE NO : 224 270 3312 32650 N WILSON RD. SE DECISION MADE: 08-OCT-13 ROUND LAKE IL 60073 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CD HORIZON SPINAL SYSTEM MEDTRONIC SOFAMOR DANEK USA, INC. 510(k) NO: K132471(SPECIAL) ATTN: SOWMYA SHETTY PHONE NO : 901 396 3133 1800 PYRAMID PLACE SE DECISION MADE: 08-OCT-13 MEMPHIS TN 38132 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ANTERIOR LUMBAR PLATE SYSTEM (PRESIDIO) LIFE SPINE, INC 510(k) NO: K132589(SPECIAL) ATTN: RANDY LEWIS PHONE NO : 847 884 6117 2401 W. HASSELL RD SUITE 1535 SE DECISION MADE: 08-OCT-13 HOFFMAN ESTATES IL 60169 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NERIA SOFT 90 INFUSION SET UNOMEDICAL A/S 510(k) NO: K132142(TRADITIONAL) ATTN: DEIRDRE BARROW PHONE NO : 440 170952 5256 UNIT B9, TAYLORS COURT, PARKGATE SE DECISION MADE: 08-OCT-13 ROTHERHAM, SOUTH YORKSHIRE UK S510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ESOPHAGEAL TTS STENT TAEWOONG MEDICAL CO., LTD 510(k) NO: K123205(TRADITIONAL) ATTN: CALLEY HERZOG PHONE NO : 720 883 3633 13417 QUIVAS ST SE DECISION MADE: 09-OCT-13 WESTMINSTER CO 80234 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: COMPREHENSIVE CONVERTIBLE GLENOID BIOMET MANUFACTURING, INC. 510(k) NO: K130390(TRADITIONAL) ATTN: PATRICIA SANDBORN BERES PHONE NO : 574 267 6639 56 EAST BELL DR. SE DECISION MADE: 09-OCT-13 WARSAW IN 46580 0587 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ROLLINS 7 OXYGEN MASK ROLLINS MEDICAL SOLUTIONS, INC. 510(k) NO: K130379(TRADITIONAL) ATTN: E. J SMITH PHONE NO : 888 729 9674 1468 HARWELL AVE. SE DECISION MADE: 09-OCT-13 CROFTON MD 21114 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FEMTO LDV(TM) Z-GENERATION FEMTOSECOND SURGICAL LASER SURGICAL INSTRUMENT ENGINEERING AG510(k) NO: K131207(TRADITIONAL) ATTN: KEVIN WALLS PHONE NO : 720 962 5412 33 GOLDEN EAGLE LANE SE DECISION MADE: 09-OCT-13 LITTLETON CO 80127 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DISTAL RADIUS VOLAR RIM PLATE STANDARD, DISTAL RADIUS VOLAR RIM PLATE NARROW BIOMET 510(k) NO: K132704(TRADITIONAL) ATTN: SUZANA OTANO PHONE NO : 305 269 6386 56 EAST BELL DR. P.O BOX 587 SE DECISION MADE: 09-OCT-13 WARSAW IN 46581 0587 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SYNERGY ODM TOPCON MEDICAL SYSTEMS, INC. 510(k) NO: K132667(TRADITIONAL) ATTN: MAUREEN O’CONNELL PHONE NO : 978 207 1245 5 TIMBER LANE SE DECISION MADE: 09-OCT-13 NORTH READING MA 01864 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PLAYTEX (SCENTED AND UNSCENTED) SPORT FRESH BALANCE TAMPONS PLAYTEX MANUFACTURING INC. 510(k) NO: K132819(SPECIAL) ATTN: PUSHPA RAO PHONE NO : 201 785 8070 75 COMMERCE DR. SE DECISION MADE: 09-OCT-13 ALLENDALE NJ 07401 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CEZANNE II INTERBODY FUSION SYSTEM ACCEL SPINE 510(k) NO: K131981(TRADITIONAL) ATTN: LISA PETERSON PHONE NO : 512 507 0746 14901 QUORUM DR. STE. 125 SE DECISION MADE: 09-OCT-13 DALLAS TX 75254 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AU BICARBONATE REAGENT BECKMAN COULTER IRELAND INC 510(k) NO: K131546(TRADITIONAL) ATTN: DAVID G DAVIS PHONE NO : 469 236 8180 250 S. KRAEMER BLVD. E1.SE.01 SE DECISION MADE: 09-OCT-13 BREA CA 92821 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: HAMILTON-G5 HAMILTON MEDICAL AG 510(k) NO: K131774(TRADITIONAL) ATTN: RALPH AGUILA PHONE NO : 41 91 6606845 VIA CRUSCH 8 SE DECISION MADE: 09-OCT-13 BONADUZ, GRISONS SZ 7402 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: POLARUS CONNECT ACUMED LLC 510(k) NO: K131636(TRADITIONAL) ATTN: BRITTANY CUNNINGHAM PHONE NO : 503 207 1467 5885 NW CORNELIUS PASS RD. SE DECISION MADE: 10-OCT-13 HILLSBORO OR 97124 9432 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMMULITE 2000 ACTH CALIBRATION VERIFICATION MATERIAL SIEMENS HEALTHCARE DIAGNOSTICS INC510(k) NO: K131662(ABBREVIATED) ATTN: GARO MIMARYAN PHONE NO : 914 524 3270 511 BENEDICT AVE. SE DECISION MADE: 10-OCT-13 TARRYTOWN NY 10591 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PEERSCOPE SYSTEM ENDOCHOICE, INC. 510(k) NO: K131422(TRADITIONAL) ATTN: TAMAR FUERST PHONE NO : 972 4 6327731 2 HATOCHEN ST., BUSINESS AND INDUSSE DECISION MADE: 10-OCT-13 CAESAREA IS 38900 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: T2 TIBIAL NAILING SYSTEM STRYKER TRAUMA AG 510(k) NO: K131365(TRADITIONAL) ATTN: ESTELA CELI PHONE NO : 201 831 6461 325 CORPORATE DRIVE SE DECISION MADE: 10-OCT-13 MAHWAH NJ 07430 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: POLARSTEM STANDARD AND LATERAL FEMORAL STEMS WITH TI/HA SMITH & NEPHEW, INC. 510(k) NO: K130728(TRADITIONAL) ATTN: SAMEER MANSOUR PHONE NO : 901 399 5579 7135 GOODLETT FARMS PARKWAY SE DECISION MADE: 10-OCT-13 CORDOVA TN 38016 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INERTIA MIS PEDICLE SCREW SYSTEM NEXXT SPINE LLC 510(k) NO: K132412(SPECIAL) ATTN: JUSTIN EGGLETON PHONE NO : 202 552 5800 1331 H STREET NW 12TH FLOOR SE DECISION MADE: 10-OCT-13 WASHINGTON DC 20005 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IDYS LIF CAGES CLARIANCE 510(k) NO: K131178(TRADITIONAL) ATTN: JANICE M HOGAN, ESQ. PHONE NO : 267 675 4611 1835 MARKET STREET 29TH FLOOR SE DECISION MADE: 10-OCT-13 PHILADELPHIA PA 19103 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMPLANET SPINE SYSTEM IMPLANET S.A. 510(k) NO: K132303(TRADITIONAL) ATTN: JANICE M HOGAN PHONE NO : 267 675 4611 1835 MARKET ST. 29TH FLOOR SE DECISION MADE: 10-OCT-13 PHILADELPHIA PA 19103 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NXSTAGE CONNECTED HEALTH SYSTEM NXSTAGE MEDICAL, INC. 510(k) NO: K131739(TRADITIONAL) ATTN: MARY LOU STROUMBOS PHONE NO : 978 687 4872 350 Merrimack St SE DECISION MADE: 10-OCT-13 LAWRENCE MA 01843 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOLOX(R) DELTA CERAMIC HEADS BIOMET, INC. 510(k) NO: K131684(TRADITIONAL) ATTN: BECKY EARL PHONE NO : 574 372 1518 56 EAST BELL DR. BOX 587 SE DECISION MADE: 10-OCT-13 WARSAW IN 46581 0587 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DELTEX MEDICAL CARDIOQ-EDM+ DELTEX MEDICAL LIMITED 510(k) NO: K132139(TRADITIONAL) ATTN: RONALD S WARREN PHONE NO : 858 753 1961 11440 W. BERNARDO CT. SUITE 300 SE DECISION MADE: 10-OCT-13 SAN DIEGO CA 92127 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BARRICADE EMBOLIZATION COIL SYSTEM BLOCKADE MEDICAL 510(k) NO: K132482(SPECIAL) ATTN: REBECCA K PINE PHONE NO : 760 809 5178 18 Technology Dr Ste 169 SE DECISION MADE: 10-OCT-13 Irvine CA 92618 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: 3B LASER NEEDLE LIVETEC GMBH 510(k) NO: K131797(TRADITIONAL) ATTN: KEVIN WALLS PHONE NO : 720 962 5412 33 GOLDEN EAGLE LANE SE DECISION MADE: 10-OCT-13 LITTLETON CO 80127 510(k) STATEMENT
DEVICE: THERAFLOW SINGLE LUMEN AND THERAFLOW DUAL LUMEN MEDICAL PRODUCTS, LLC 510(k) NO: K131023(TRADITIONAL) ATTN: DAVID MCGILL PHONE NO : 317 432 5114 7437 OAKLANDON RD. SE DECISION MADE: 11-OCT-13 INDIANAPOLIS IN 46236 510(k) STATEMENT
DEVICE: BASIC AND DOMINANT FLEX SUCTION PUMPS MEDELA AG 510(k) NO: K130123(TRADITIONAL) ATTN: ADRIENNE LENZ PHONE NO : 262 290 0023 W324S3649 COUNTY ROAD E SE DECISION MADE: 11-OCT-13 DOUSMAN WI 53118 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ACUSON S2000 AND S3000 SIEMENS MEDICAL SOLUTIONS USA, INC510(k) NO: K130881(TRADITIONAL) ATTN: SHELLY PEARCE PHONE NO : 650 694 5988 1230 SHOREBIRD WAY SE DECISION MADE: 11-OCT-13 MOUNTAIN VIEW CA 94043 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PPA/BT-RB STETHOSCOPE RNK PRODUCTS, INC. 510(k) NO: K132405(TRADITIONAL) ATTN: CHARLES R ABBRUSCATO PHONE NO : 321 610 3980 8247 Devereux Dr Ste 101 SE DECISION MADE: 11-OCT-13 Melbourne FL 32940 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AUDICOR CPAM WITH SDB (SINGLE SENSOR) INOVISE MEDICAL, INC. 510(k) NO: K130660(TRADITIONAL) ATTN: EARL ANDERSON PHONE NO : 503 431 3800 8770 SW NIMBUS AVE. SUITE D SE DECISION MADE: 11-OCT-13 Beaverton OR 97008 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: REVERSE MEDICAL MICROCATHETER REVERSE MEDICAL CORPORATION 510(k) NO: K130858(TRADITIONAL) ATTN: JEFFREY VALKO PHONE NO : 949 215 0660 13700 ALTON PARKWAY SUITE 167 SE DECISION MADE: 11-OCT-13 IRVINE CA 92618 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MODIFIED CONCENTRIC MICROCATHETER CONCENTRIC MEDICAL, INC. 510(k) NO: K131437(TRADITIONAL) ATTN: CHRISTINA ROWE PHONE NO : 510 413 2486 301 E. EVELYN AVE. SE DECISION MADE: 11-OCT-13 MOUNTAIN VIEW CA 94041 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CLASSIC INTERMITTENT CATHETER FLEXICARE MEDICAL LTD. 510(k) NO: K132619(TRADITIONAL) ATTN: CHRISTOPHER WATKINS PHONE NO : 44 1443 474560 CYNON VALLEY BUSINESS PARK MOUTAINSE DECISION MADE: 11-OCT-13 MID GLAMORGAN UK CF45 4ER 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: MTM(R) CLEAR ALIGNER DENTSPLY INTERNATIONAL, INC. 510(k) NO: K132145(TRADITIONAL) ATTN: HELEN LEWIS PHONE NO : 717 487 1332 SUSQUEHANNA COMMERCE CTR., 221 W. SE DECISION MADE: 11-OCT-13 YORK PA 17404 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PCP-USB STETHOSCOPE RNK PRODUCTS, INC. 510(k) NO: K132560(TRADITIONAL) ATTN: CHARLES R ABBRUSCATO PHONE NO : 621 610 3980 8247 Devereux Dr Ste 101 SE DECISION MADE: 11-OCT-13 Melbourne FL 32940 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: HIGH ENERGY LINEAR ACCELERATOR VARIAN MEDICAL SYSTEMS, INC. 510(k) NO: K131807(TRADITIONAL) ATTN: PETER J CORONADO PHONE NO : 650 424 6320 3100 HANSEN WAY SE DECISION MADE: 11-OCT-13 PALO ALTO CA 94304 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PRECICE INTRAMEDULLARY LIMB LENGTHENING SYSTEM ELLIPSE TECHNOLOGIES INC. 510(k) NO: K131677(TRADITIONAL) ATTN: JOHN MCINTYRE PHONE NO : 949 837 3600 13900 ALTON PKWY STE 123 SE DECISION MADE: 11-OCT-13 IRVINE CA 92618 0000 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TURBO-JECT PERIPHERALLY INSERTED CENTRAL VENOUS CATHETER (PICC) SET COOK INCORPORATED 510(k) NO: K132885(SPECIAL) ATTN: SEAN SPENCE PHONE NO : 812 335 3575 750 DANIELS WAY SE DECISION MADE: 11-OCT-13 BLOOMINGTON IN 47404 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: KATANA ZIRCONIA KURARAY NORITAKE DENTAL INC. 510(k) NO: K131534(TRADITIONAL) ATTN: IVAN WASSERMAN PHONE NO : 202 585 6529 700 12TH ST SW SUITE 100 SE DECISION MADE: 11-OCT-13 WASHINGTON DC 20005 510(k) STATEMENT
DEVICE: BALANCED KNEE SYSTEM HIGH FLEX VITAMIN E PS TIBIAL INSERT AND PATELLA ORTHO DEVELOPMENT CORP. 510(k) NO: K131337(TRADITIONAL) ATTN: TOM HAUETER PHONE NO : 801 553 9991 12187 SOUTH BUSINESS PARK DR. SE DECISION MADE: 11-OCT-13 DRAPER UT 84020 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: 2008T HEMODIALYSIS MACHINE WITH OPTIONAL CRIT-LINE MONITOR (CLIC) FRESENIUS MEDICAL CARE RENAL THERA510(k) NO: K131908(TRADITIONAL) ATTN: DENISE OPPERMANN PHONE NO : 781 699 4479 920 WINTER STREET SE DECISION MADE: 11-OCT-13 WALTHAM MA 02451 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SEATTLE – PAP SEATTLE CHILDRENS HOSPITAL 510(k) NO: K131502(TRADITIONAL) ATTN: PAUL DRYDEN PHONE NO : 239 498 2155 1900 NINTH AVENUE SE DECISION MADE: 11-OCT-13 SEATTLE WA 98101 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OPTETRAK LOGIC PS 17MM & 19MM THICK INSERTS, OPTETRAK LOGIC PSC 17MM & 19MM THICK INSERTS EXACTECH, INC. 510(k) NO: K132161(TRADITIONAL) ATTN: PATRICK HUGHES PHONE NO : 352 327 4762 2320 N.W. 66TH CT. SE DECISION MADE: 11-OCT-13 GAINESVILLE FL 32653 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NON-CONTACT INFRARED THERMOMETER JINXINBAO ELECTRONIC CO.,LTD 510(k) NO: K130231(ABBREVIATED) ATTN: HELEN NAN PHONE NO : 086 577886 06182 ROOM 404, BLDG 7, JINHUICHANG HOMESE DECISION MADE: 15-OCT-13 WENZHOU, ZHEJIANG CH 325000 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LUNGPOINT TOOLS (LUNGPOINT SHEATH, LUNGPOINT DILATION BALLOON) BRONCUS MEDICAL INC 510(k) NO: K131234(TRADITIONAL) ATTN: MAHTAB FATEMI PHONE NO : 650 428 1600 1400 N. Shoreline Blvd., Bldg A, SSE DECISION MADE: 15-OCT-13 Mountain View CA 94043 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: ENDOCHOICE HOT BIOPSY FORCEPS ENDOCHOICE, INC. 510(k) NO: K131991(TRADITIONAL) ATTN: DANIEL HOEFER PHONE NO : 678 708 4743 11810 WILLS ROAD SE DECISION MADE: 15-OCT-13 ALPHARETTA GA 30009 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MULTICHEM U TECHNO-PATH MANUFACTURING LTD. 510(k) NO: K131993(ABBREVIATED) ATTN: STEPHANIE G GARTH PHONE NO : 469 230 0959 325 BIG ELM ST. SE DECISION MADE: 15-OCT-13 HIGHLAND VILLAGE TX 75077 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NASAL COMFORT FREEDOM LAKEVIEW SLEEP CENTER 510(k) NO: K130686(TRADITIONAL) ATTN: ANDY BLACK PHONE NO : 847 620 7990 400 NORTH RAND RD SE DECISION MADE: 16-OCT-13 NORTH BARRINGTON IL 60010 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PROW FUSION NLT SPINE LTD 510(k) NO: K130254(TRADITIONAL) ATTN: JONATHAN KAHAN PHONE NO : 202 637 5794 555 THIRTEENTH STREET NW SE DECISION MADE: 16-OCT-13 WASHINGTON DC 20004 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OVATION 10/12 HIP STEM ORTHO DEVELOPMENT CORP. 510(k) NO: K131022(TRADITIONAL) ATTN: TOM HAUETER PHONE NO : 801 553 9991 12187 SOUTH BUSINESS PARK DR. SE DECISION MADE: 16-OCT-13 DRAPER UT 84020 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AEQUALIS SHOULDER FRACTURE SYSTEM, AEQUALIS REVERSED SHOULDER PROSTHESIS, AEQUALIS REVERSED FRACTURE SHOULDER PROTHESIS TORNIER SAS 510(k) NO: K131231(TRADITIONAL) ATTN: SEVERINE BONNETON PHONE NO : 003 347 6613500 161 RUE LAVOISIER SE DECISION MADE: 16-OCT-13 MONTBONNOT SAINT MARTIN FR 3833510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MEDLINE SUCTION COAGULATOR MEDLINE INDUSTRIES, INC. 510(k) NO: K122949(TRADITIONAL) ATTN: MATT CLAUSEN PHONE NO : 847 643 4785 ONE MEDLINE PLACE SE DECISION MADE: 16-OCT-13 MUNDELEIN IL 60060 510(k) STATEMENT
DEVICE: PEDIATRIC FOLDING WHEELCHAIR COLUMBIA MEDICAL MFG, LLC 510(k) NO: K130644(ABBREVIATED) ATTN: GARY WERSCHMIDT PHONE NO : 562 282 0244 11724 WILLAKE ST SE DECISION MADE: 16-OCT-13 SANTA FE SPRINGS CA 90670 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FINGERTIP PULSE OXIMETER BEIJING CHOICE ELECTRONIC TECHNOLO510(k) NO: K131047(TRADITIONAL) ATTN: LEI CHEN PHONE NO : 86 10 88794630 NO. 9 SHUANGYUAN ROAD, BADACHU HI-SE DECISION MADE: 16-OCT-13 SHIJINGSHAN DISTRICT, BEIJING C510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BRAEMAR CARDIOKEY HOLTER RECORDER BRAEMAR MANUFACTURING LLC 510(k) NO: K130294(SPECIAL) ATTN: KENT SAYLER PHONE NO : 619 243 7500 750 B. STREET, STUITE 1400 SE DECISION MADE: 16-OCT-13 SAN DIEGO CA 92101 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: A.I.M. SUTURE ANCHOR ANCHOR INNOVATION MEDICAL, INC. 510(k) NO: K132461(TRADITIONAL) ATTN: HOWARD L SCHRAYER PHONE NO : 5410 EDSON LANE SUITE 308 SE DECISION MADE: 16-OCT-13 ROCKVILLE MD 20852 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VANTAGE TITAN 3T TOSHIBA MEDICAL SYSTEMS CORPORATIO510(k) NO: K132160(TRADITIONAL) ATTN: PAUL BIGGINS PHONE NO : 714 730 5000 2441 MICHELLE DRIVE SE DECISION MADE: 16-OCT-13 TUSTIN CA 92780 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: .DECIMAL PROTON APERTURE .DECIMAL, INC. 510(k) NO: K132236(TRADITIONAL) ATTN: KIMBERLY RUPP PHONE NO : 407 330 3300 121 CENTRAL PARK PLACE SE DECISION MADE: 16-OCT-13 SANFORD FL 32771 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IFUSE IMPLANT SYSTEM SI-BONE, INC. 510(k) NO: K131405(TRADITIONAL) ATTN: CINDY DOMECUS PHONE NO : 650 343 4813 1171 BARROILHET DR SE DECISION MADE: 16-OCT-13 HILLSBOROUGH CA 94010 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SMOOTH OR THREADED METALLIC BONE FASTENER BIOMEDICAL ENT., INC. 510(k) NO: K131640(TRADITIONAL) ATTN: JOE SOWARD PHONE NO : 210 677 0354 14785 OMICRON DR., SUITE 205 SE DECISION MADE: 16-OCT-13 SAN ANTONIO TX 78245 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LEGACY MEDICAL ECO-CHOICE DISPOSABLE BLOOD PRESSURE CUFF LEGACY MEDICAL, INC. 510(k) NO: K130942(TRADITIONAL) ATTN: DEB ROBERTSON PHONE NO : 952 322 7240 9039 Horizon Dr SE DECISION MADE: 17-OCT-13 Shakopee MN 55379 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: UNIVATION
AESCULAP IMPLANT SYSTEMS, LLC 510(k) NO: K131167(TRADITIONAL)
ATTN: JULIE T WING PHONE NO : 610 984 9147
3773 CORPORATE PARKWAY SE DECISION MADE: 17-OCT-13
CENTER VALLEY PA 18034 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TVC IMAGING SYSTEM, TVC INSIGHT CATHETER INFRAREDX, INC. 510(k) NO: K130719(TRADITIONAL) ATTN: STEVE CHARTIER PHONE NO : 781 345 9593 34 THIRD AVE SE DECISION MADE: 17-OCT-13 BURLINGTON MA 01803 510(k) STATEMENT
DEVICE: INTELLIVUE PATIENT MONITOR MX500, INTELLIVUE PATIENT MONITOR MX550 PHILIPS MEDIZINSYSTEME BOEBLINGEN 510(k) NO: K131872(SPECIAL) ATTN: MARKUS STACHA PHONE NO : 49 7031 4632840 HEWLETT-PACKARD STR.2 SE DECISION MADE: 17-OCT-13 BOEBLINGEN GM D 71034 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRUE SPINAL FIXATION SYSTEM INNOVATIVE SURGICAL DESIGNS 510(k) NO: K130958(TRADITIONAL) ATTN: ED MORRIS PHONE NO : 812 774 9297 2660 E. 2ND STREET #10 SE DECISION MADE: 17-OCT-13 BLOOMINGTON IN 47401 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NBM-200MP ORSENSE LTD 510(k) NO: K124041(TRADITIONAL) ATTN: MARK A HELLER PHONE NO : 202 346 4107 901 NEW YORK AVE, N.W. SE DECISION MADE: 17-OCT-13 WASHINGTON DC 20001 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOROOT RCS NOVOCOL, INC. 510(k) NO: K130601(ABBREVIATED) ATTN: CORA BRACHO-TROCONIS PHONE NO : 303 665 7535 416 SOUTH TAYLOR AVE. SE DECISION MADE: 17-OCT-13 LOUISVILLE CO 80027 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RESPIGUIDE DELIVERY SYSTEM RESPICARDIA 510(k) NO: K130378(TRADITIONAL) ATTN: BONNIE LABOSKY PHONE NO : 952 540 4470 12400 WHITEWATER DRIVE SUITE 150 SE DECISION MADE: 17-OCT-13 MINNETONKA MN 55343 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AAP K-WIRE, STEINMANN PIN, CERCLAGE WIRE AAP IMPLANTATE AG 510(k) NO: K131459(TRADITIONAL) ATTN: CHRISTIAN ZIETSCH PHONE NO : 49 307501 9193 LORENZWEG 5 SE DECISION MADE: 17-OCT-13 BERLIN GM 12099 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: HARMONIC ACE+ SHEARS 23 CM LENGTH WITH ADVANCED HEMOSTASIS, HARMONIC ACE + LAPAROSCOPIC SHEARS 36 CM LENGTH WITH ADVANCE ETHICON ENDO-SURGERY, LLC 510(k) NO: K132612(TRADITIONAL) ATTN: BRIAN GODWIN PHONE NO : 513 337 3623 4545 CREEK ROAD SE DECISION MADE: 17-OCT-13 CINCINNATI OH 45242 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRANSCEND AUTO SOMNETICS INTERNATIONAL, INC. 510(k) NO: K132127(TRADITIONAL) ATTN: MELINDA SWANSON PHONE NO : 651 621 1800 33 5TH AVE NW, STE 500 SE DECISION MADE: 17-OCT-13 NEW BRIGHTON MN 55112 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SITE-RITE VISION II ULTRASOUND SYSTEM C.R. BARD, INC. 510(k) NO: K132942(SPECIAL) ATTN: KERRIE HAMBLIN PHONE NO : 801 522 4909 605 NORTH 5600 WEST SE DECISION MADE: 17-OCT-13 SALT LAKE CITY UT 84116 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DYONICS PLAN HIP IMPINGEMENT PLANNING SYSTEM SMITH & NEPHEW, INC. 510(k) NO: K132636(TRADITIONAL) ATTN: KATHLEEN SOLOMON PHONE NO : 978 749 1605 150 MINUTEMAN RD. SE DECISION MADE: 17-OCT-13 ANDOVER MA 01810 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: UNIVERSAL NEURO 3 SYSTEM STRYKER 510(k) NO: K131775(TRADITIONAL) ATTN: MANISH PATEL PHONE NO : 269 388 4271 750 TRADE CENTRE WAY STE 200 SE DECISION MADE: 17-OCT-13 PORTAGE MI 49002 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: STERLING OVER-THE-WIRE (OTW) PTA BALLOON DILATATION CATHETER BOSTON SCIENTIFIC CORP. 510(k) NO: K132430(TRADITIONAL) ATTN: CHRISTOPHER DACHEL PHONE NO : 763 494 2718 ONE SCIMED PLACE SE DECISION MADE: 17-OCT-13 MAPLE GROVE MN 55311 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SAPPHIRE INFUSION PUMP Q CORE MEDICAL LTD 510(k) NO: K123049(TRADITIONAL) ATTN: RHONA SHANKER PHONE NO : 301 251 9570 12154 DARNESTOWN ROAD #236 SE DECISION MADE: 17-OCT-13 GAITHERBURG MD 20878 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SOMNOLYZER 24X7 RESPIRONICS, INC. 510(k) NO: K131994(TRADITIONAL) ATTN: DANIELA AIZPITARTE PHONE NO : 724 387 7954 1740 GOLDEN MILE HIGHWAY SE DECISION MADE: 17-OCT-13 MONROEVILLE PA 15146 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMAGENET 5 PC SOFTWARE SYSTEM TOPCON MEDICAL SYSTEMS, INC. 510(k) NO: K132438(TRADITIONAL) ATTN: MAUREEN O’CONNELL PHONE NO : 978 207 1245 5 TIMBER LANE SE DECISION MADE: 17-OCT-13 NORTH READING MA 01864 510(k) STATEMENT
DEVICE: GLOOKO DEVICE SYSTEM FOR GLOOKO APPLICATION GLOOKO, INC. 510(k) NO: K132272(SPECIAL) ATTN: SHILPA MYDUR PHONE NO : 650 720 5310 170A UNIVERSITY AVENUE SE DECISION MADE: 17-OCT-13 PALO ALTO CA 94301 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PHILIPS HEARTSTART FR3 AUTOMATED EXTERNAL DEFIBRILLATOR PHILIPS MEDICAL SYSTEMS 510(k) NO: K130684(TRADITIONAL) ATTN: NANCY BURKE PHONE NO : 425 487 7000 22100 BOTHELL EVERETT HIGHWAY SE DECISION MADE: 18-OCT-13 BOTHELL WA 98041 3003 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ASA ORAL APPLIANCES AMERICAN SLEEP ASSOCIATION 510(k) NO: K130504(TRADITIONAL) ATTN: KAREN E WARDEN, PHD PHONE NO : 440 729 8457 PO BOX 566 SE DECISION MADE: 18-OCT-13 CHESTERLAND OH 44026 2141 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NO-PROFILE SCREWS NO-PROFILE WASHER NO-PROFILE SCREWS WASHERLOC WASHERS BIOMET SPORTS MEDICINE 510(k) NO: K122437(TRADITIONAL) ATTN: ELIZABETH WRAY PHONE NO : 574 371 1176 56 EAST BELL DRIVE P.O. BOX 587 SE DECISION MADE: 18-OCT-13 WARSAW IN 46581 0587 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CRANIAL MAP NEURO STRYKER CORPORATE 510(k) NO: K131214(TRADITIONAL) ATTN: LILIAN ECKERT PHONE NO : 49 761 4512117 BOETZINGER STR. 41 SE DECISION MADE: 18-OCT-13 FREIBURG, BADEN-WUERTTEMBERG GM510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ELECSYS CK-MB STAT IMMUNOASSAY, ELECSYS CK-MB IMMUNOASSAY ROCHE DIAGNOSTICS 510(k) NO: K132571(TRADITIONAL) ATTN: KELLI TURNER PHONE NO : 317 521 4515 9115 HAGUE ROAD SE DECISION MADE: 18-OCT-13 INDIANAPOLIS IN 46250 0416 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DIREXION AND DIREXION HI-FLO TORQUEABLE MICROCATHETERS BOSTON SCIENTIFIC CORP. 510(k) NO: K132947(SPECIAL) ATTN: MAUREEN SUNDEEN PHONE NO : 763 494 2903 ONE SCIMED PLACE SE DECISION MADE: 18-OCT-13 MAPLE GROVE MN 55311 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MULTIFUNCTIONAL SERIES BEIJING HONKON TECHNOLOGIES CO., L510(k) NO: K131859(TRADITIONAL) ATTN: DIANA HONG PHONE NO : 86 21 22815850 P.O. BOX 120-119 SE DECISION MADE: 18-OCT-13 SHANGHAI CH 200030 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ECHELON ENDOSCOPIC LINEAR CUTTER RELOAD, BLACK ETHICON ENDO-SURGERY, LLC 510(k) NO: K131663(TRADITIONAL) ATTN: CHRISTINA CANTER PHONE NO : 513 337 7321 4545 CREEK ROAD SE DECISION MADE: 18-OCT-13 CINCINNATI OH 45242 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: REMSTAR AUTO A-FLEX HT RESPIRONICS, INC. 510(k) NO: K131982(SPECIAL) ATTN: MICHELLE BRINKER PHONE NO : 724 387 4146 1740 GOLDEN MILE HIGHWAY SE DECISION MADE: 18-OCT-13 MONROEVILLE PA 15146 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LENSTEC LC INJECTION SYSTEM LENSTEC, INC. 510(k) NO: K122848(TRADITIONAL) ATTN: JIMMY CHACKO PHONE NO : 727 571 2272 1765 Commerce Ave N SE DECISION MADE: 18-OCT-13 Saint Petersburg FL 33716 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: PROVOX VEGA PUNCTURE SET ATOS MEDICAL AB 510(k) NO: K131947(TRADITIONAL) ATTN: FERENC DAHNER PHONE NO : 46 415198 00 KRAFTGATAN 8 SE DECISION MADE: 18-OCT-13 HORBY SW SE-24222 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: GYRUS ACMI URO-EZDILATE URETERAL BALLOON DILATION CATHETER OLYMPUS SURGICAL TECHNOLOGIES AMER510(k) NO: K132181(TRADITIONAL) ATTN: NEIL KELLY PHONE NO : 908 804 2600 136 TURNPIKE ROAD SE DECISION MADE: 21-OCT-13 SOUTHBOROUGH MA 01772 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NEO DENTA ZIRCONIA NEO GROUP LIMITED 510(k) NO: K130198(TRADITIONAL) ATTN: MIKE ADAMS PHONE NO : 852 235 69700 ROOM Q1D G/F, KAISER EST 3RD PHASESE DECISION MADE: 21-OCT-13 HUNG HOM, KOWLOON HK HK 852 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PURITAN AMIES MEDIUM COLLECTION AND TRANSPORT SYSTEM PURITAN MEDICAL PRODUCTS LLC 510(k) NO: K131630(TRADITIONAL) ATTN: MEHDI KARAMCHI PHONE NO : 207 876 3311 31 SCHOOL STREET P.O. BOX 149 SE DECISION MADE: 21-OCT-13 GUILFORD ME 04443 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SWIFT AIR RESMED LTD. 510(k) NO: K132013(TRADITIONAL) ATTN: JIM CASSI PHONE NO : 858 836 6081 9001 SPECTRUM CENTER BLVD. SE DECISION MADE: 21-OCT-13 SAN DIEGO CA 92123 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LESSER METATARSAL HEAD IMPLANT SYSTEM SOLANA SURGICAL, LLC 510(k) NO: K131187(TRADITIONAL) ATTN: JOE CLIFT PHONE NO : 901 818 1860 6363 POPLAR AVENUE SUITE 312 SE DECISION MADE: 22-OCT-13 MEMPHIS TN 38119 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: PERIFLUX SYSTEM 6000 PERIMED AB 510(k) NO: K131253(ABBREVIATED) ATTN: MARIA LILJEVRET PHONE NO : 46 8 58011990 DATAVAGEN 9 A SE DECISION MADE: 22-OCT-13 JARFALLA SE 17543 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RANFAC BONE MARROW ASPIRATION NEEDLE RANFAC CORP. 510(k) NO: K131157(TRADITIONAL) ATTN: CHRISTOPHER P WHELAN PHONE NO : 508 588 4400 30 Doherty Avenue, P.O. Box 635 AvSE DECISION MADE: 22-OCT-13 Avon MA 02322 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MEDCOMP VASCU-PICC WITH VALVE MEDCOMP (MEDICAL COMPONENTS) 510(k) NO: K130897(TRADITIONAL) ATTN: JESSICA LEO PHONE NO : 215 256 4201 1499 DELP DRIVE SE DECISION MADE: 22-OCT-13 HARLEYSVILLE PA 19438 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: 5F & 6F LAUNCHER GUIDE CATHETER, 5F SHERPA ACTIVE NX GUIDE CATHETER, 6F Z4 GUIDING CATHETER MEDTRONIC INC. 510(k) NO: K132673(SPECIAL) ATTN: HEATHER MOROSE PHONE NO : 978 739 3187 37A CHERRY HILL DRIVE SE DECISION MADE: 22-OCT-13 DANVERS MA 01923 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMMULITE 2000; HCG CALIBRATION VERIFICATION MATERIAL, INSULIN CALIBRATION VERIFICATION MATERIAL, PYRILINKS-D CALIBRATION SIEMENS HEALTHCARE DIAGNOSTICS INC510(k) NO: K133128(ABBREVIATED) ATTN: ASHA GARTLAND PHONE NO : 914 524 3257 511 BENEDICT AVE. SE DECISION MADE: 22-OCT-13 TARRYTOWN NY 10591 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TAUTONA INJECTOR DEVICE TAUTONA GROUP, LLC 510(k) NO: K131963(TRADITIONAL) ATTN: JOE RIMSA PHONE NO : 650 331 2450 4040 CAMPBELL AVE, STE 110 SE DECISION MADE: 22-OCT-13 MENLO PARK CA 94025 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OCCLUSION BALLOON CATHETER BOSTON SCIENTIFIC CORP. 510(k) NO: K132990(SPECIAL) ATTN: DIANE NELSON PHONE NO : 763 255 0813 ONE SCIMED PLACE SE DECISION MADE: 22-OCT-13 MAPLE GROVE MN 55311 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MULTICHEM S PLUS / S PLUS (ASSAYED) TECHNO-PATH MANUFACTURING LTD. 510(k) NO: K132751(ABBREVIATED) ATTN: STEPHANIE G GARTH PHONE NO : 469 230 0959 325 BIG ELM ST. SE DECISION MADE: 22-OCT-13 HIGHLAND VILLAGE TX 75077 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: GEM FLOW COUPLER DEVICE AND SYSTEM SYNOVIS LIFE TECHNOLOGIES, INC. 510(k) NO: K132727(SPECIAL) ATTN: TROY THOME PHONE NO : 651 796 7300 2575 UNIVERSITY AVE., STE.180 SE DECISION MADE: 22-OCT-13 ST. PAUL MN 55114 1024 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SMITH & NEPHEW GLOBAL FEMORAL HEAD TRIALS SMITH & NEPHEW, INC. 510(k) NO: K132435(TRADITIONAL) ATTN: MEGAN FESSENDEN PHONE NO : 901 399 1104 7135 GOODLETT FARMS PARKWAY SE DECISION MADE: 22-OCT-13 CORDOVA TN 38016 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VISCOELASTIC INJECTOR SIGHT SCIENCES, INC. 510(k) NO: K132494(TRADITIONAL) ATTN: JUDY F GORDON D.V.M. PHONE NO : 949 715 0609 733 BOLSANA DRIVE SE DECISION MADE: 22-OCT-13 LAGUNA BEACH CA 92651 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CALIX LUMBAR SPINAL IMPLANT SYSTEM X-SPINE SYSTEMS, INC 510(k) NO: K131350(TRADITIONAL) ATTN: DAVID KIRSCHMAN PHONE NO : 937 847 8400 452 Alexandersville Rd SE DECISION MADE: 23-OCT-13 Miamisburg OH 45342 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OPTUM TELEHEALTH APPLICATION OPTUMHEALTH CARE SOLUTIONS, INC. 510(k) NO: K130971(TRADITIONAL) ATTN: ROSHANA AHMED, MA, RAC PHONE NO : 905 690 5792 4 INNOVATION DR SE DECISION MADE: 23-OCT-13 DUNDAS, ON CA L9H 7P3 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RENASYS EZ MAX NEGATIVE PRESSURE WOUND THERAPY DEVICE SMITH & NEPHEW, INC. 510(k) NO: K132446(SPECIAL) ATTN: LAURA REYNOLDS PHONE NO : 727 399 7702 970 LAKE CARILLON DR. SUITE 110 SE DECISION MADE: 23-OCT-13 ST. PETERSBURG FL 33716 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: COLLAGEN WOUND DRESSING KERECIS LIMITED 510(k) NO: K132343(TRADITIONAL) ATTN: GUDMUNDUR F SIGURJONSSON PHONE NO : 354 562 2601 EYRARGATA 2 SE DECISION MADE: 23-OCT-13 ISAFIJORDUR IC 400 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: WMT IMPLANTABLE K-WIRE WRIGHT MEDICAL TECHNOLOGY, INC. 510(k) NO: K132895(TRADITIONAL) ATTN: LESLIE FITCH PHONE NO : 901 867 4409 5677 AIRLINE RD. SE DECISION MADE: 23-OCT-13 ARLINGTON TN 38002 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AUTOMATIC DIGITAL BLOOD PRESSURE MONITOR HONSUN (NANTONG) CO., LTD. 510(k) NO: K132627(SPECIAL) ATTN: SELINA QIAN PHONE NO : 86 0513 80580116 NO.8, Tongxing Road, Nantong EconoSE DECISION MADE: 23-OCT-13 NANTONG, JIANGSU CH 226009 510(k) STATEMENT
DEVICE: EVOLUTION MP ADAPTIVE PS TIBIAL INSERT WRIGHT MEDICAL TECHNOLOGY, INC. 510(k) NO: K131679(TRADITIONAL) ATTN: THERESA LEISTER PHONE NO : 901 290 5898 5677 AIRLINE RD. SE DECISION MADE: 23-OCT-13 ARLINGTON TN 38002 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INNERSENSE ESOPHAGEAL TEMPERATURE PROBE / FEEDING TUBE PHILIPS MEDICAL SYSTEMS 510(k) NO: K131590(TRADITIONAL) ATTN: CHRISTINE TREFETHEN PHONE NO : 978 659 3890 3000 Minuteman Rd SE DECISION MADE: 23-OCT-13 ANDOVER MA 01810 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DYNAMESH -CICAT FEG TEXTILTECHNIK FORSCHUNGS-UND E510(k) NO: K131530(TRADITIONAL) ATTN: NEIL R ARMSTRONG PHONE NO : 011 44 1763222505 HERLINGTON HOUSE, ORTON MALBORNE SE DECISION MADE: 23-OCT-13 PETERBOROUGH, CAMBS. UK PE2 5XS510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RANDOX CSF CONTROLS LEVELS 2 AND 3 RANDOX LABORATORIES, LTD. 510(k) NO: K132156(TRADITIONAL) ATTN: PAULINE ARMSTRONG PHONE NO : 028 944 22413 55 DIAMOND RD. SE DECISION MADE: 23-OCT-13 CRUMLIN, COUNTY ANTRIM UK BT29 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: VARIAX 2 SYSTEM STRYKER TRAUMA AG 510(k) NO: K132502(TRADITIONAL) ATTN: ELIJAH N WREH PHONE NO : 201 3 5691 325 CORPORATE DR SE DECISION MADE: 23-OCT-13 MAHWAH NJ 07430 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ENDO GIA RADIAL RELOAD WITH TRI-STAPLE TECHNOLOGY COVIDIEN LLC 510(k) NO: K132493(SPECIAL) ATTN: KATHERINE KASSIM PHONE NO : 203 492 5299 60 MIDDLETOWN AVENUE SE DECISION MADE: 23-OCT-13 NORTH HAVEN CT 06473 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LOTUS SPINAL ELEMENTS, INC. 510(k) NO: K131427(TRADITIONAL) ATTN: BENJAMIN A KIMBALL PHONE NO : 760 607 1816 3115 MELROSE DRIVE STE 200 SE DECISION MADE: 24-OCT-13 CARLSBAD CA 92010 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOPRO – INFINITY PLATE ANCHOR SYSTEM BIOPRO, INC. 510(k) NO: K132510(TRADITIONAL) ATTN: AL LIPPINCOTT PHONE NO : 952 492 5858 3150 E. 200TH ST SE DECISION MADE: 24-OCT-13 PRIOR LAKE MN 55372 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: CORE 3D ABUTMENT SYSTEM FOR DIGITAL PROSTHETIC SOLUTIONS CORE 3D PROTECH, S.L. 510(k) NO: K122295(ABBREVIATED) ATTN: ANNA CORTINA PHONE NO : 349 022828 48 POL. IND. SANTA ANNA APARTAT 20 SE DECISION MADE: 24-OCT-13 SANTPEDOR SP 08251 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MAK OSS FEMORAL KNEE COMPONENTS BIOMET ORTHOPEDICS CORP. 510(k) NO: K131393(TRADITIONAL) ATTN: GARY BAKER PHONE NO : 574 267 6639 56 EAST BELL DRIVE SE DECISION MADE: 24-OCT-13 WARSAW IN 46582 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ACUSON S1000 / S2000 / S3000 SIEMENS MEDICAL SOLUTIONS USA, INC510(k) NO: K132804(TRADITIONAL) ATTN: CHRISTINE DUNN PHONE NO : 685 EAST MIDDLEFIELD ROAD SE DECISION MADE: 24-OCT-13 MOUNTAIN VIEW CA 94043 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: OMBRA TABLE TOP COMPRESSOR TRUDELL MEDICAL INTL. 510(k) NO: K131881(TRADITIONAL) ATTN: DARRYL FISCHER PHONE NO : 519 455 7060 725 THIRD ST. SE DECISION MADE: 24-OCT-13 LONDON CA N5V 5G4 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ULTRACEM ULTRADENT PRODUCTS INC. / ORATECH 510(k) NO: K132255(TRADITIONAL) ATTN: DIANE E ROGER PHONE NO : 801 553 4491 10200 SOUTH 505 WEST SE DECISION MADE: 24-OCT-13 SOUTH JORDAN UT 84095 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DA01 BLOOD GLUCOSE MONITORING SYSTEM DELBIO INCORPORATION 510(k) NO: K132338(SPECIAL) ATTN: ERICA LI PHONE NO : 886 3359 6268 X8672 3F,6F, No.252, Shangying Road, GuiSE DECISION MADE: 24-OCT-13 Taoyuan County TW 33341 510(k) STATEMENT
DEVICE: SHEATHLESS EAUCATH CORONARY GUIDE CATHETER ASAHI INTECC CO., LTD. 510(k) NO: K132556(TRADITIONAL) ATTN: YOSHI TERAI PHONE NO : 949 756 8252 2500 RED HILL AVE, SUITE 210 SE DECISION MADE: 24-OCT-13 SANTA ANA CA 92705 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RIGHTEST CONTROL SOLUTION BIONIME CORPORATION 510(k) NO: K133003(TRADITIONAL) ATTN: JIGAR SHAH PHONE NO : 516 482 9001 55 NORTHERN BLVD. SUITE 200 SE DECISION MADE: 24-OCT-13 GREAT NECK NY 11021 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TROJAN JAGUAR MALE NATURAL RUBBER LATEX CONDOM WITH WARMING AND TINGLING LUBRICANT CHURCH & DWIGHT CO., INC. 510(k) NO: K131887(TRADITIONAL) ATTN: EMILY PEREZ PHONE NO : 609 806 1430 500 CHARLES EWING BLVD SE DECISION MADE: 24-OCT-13 EWING NJ 08628 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: (QUANTA SYSTEM) SURGICAL LASER FIBERS QUANTA SYSTEM, S.P.A. 510(k) NO: K131473(TRADITIONAL) ATTN: MAURIZIO BIANCHI PHONE NO : 39 0331 376797 VIA IV NOVEMBRE,116 SE DECISION MADE: 24-OCT-13 SOLBIATE OLONA IT 21058 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AIRVO 2 HUMIDIFIER, MYAIRVO 2 HUMIDIFIER FISHER & PAYKEL HEALTHCARE, LTD. 510(k) NO: K131895(TRADITIONAL) ATTN: BRETT WHISTON PHONE NO : 64 9 5740100 15 MAURICE PAYKEL PLACE SE DECISION MADE: 24-OCT-13 EAST TAMAKI NZ 2013 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRIA FAN SYSTEM TRIA BEAUTY, INC. 510(k) NO: K130459(TRADITIONAL) ATTN: JONATHAN KAHAN PHONE NO : 202 638 5794 555 13TH STREET, NW COLUMBIA SQUARSE DECISION MADE: 25-OCT-13 WASHINGTON DC 20004 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: LIQUICHEK IMMUNOASSAY PREMIUM CONTROL-TRILEVEL, LIQUICHEK IMMUNOASSAY PREMIUM CONTROL-LEVEL I, LIQUICHEK IMMUNOASSAY BIO-RAD LABORATORIES 510(k) NO: K132227(TRADITIONAL) ATTN: SUZANNE PARSONS PHONE NO : 949 598 1467 9500 JERONIMO RD. SE DECISION MADE: 25-OCT-13 IRVINE CA 92618 2017 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TRELLIS-8 PERIPHERAL INFUSION SYSTEM COVIDIEN 510(k) NO: K130904(TRADITIONAL) ATTN: JENNIFER SULLIVAN PHONE NO : 508 452 1659 15 HAMPSHIRE STREET SE DECISION MADE: 25-OCT-13 MANSFIELD MA 02048 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOLIGHT BIODYNAM BIOLIGHT AB 510(k) NO: K130866(SPECIAL) ATTN: PAR OLAUSSON LIDO PHONE NO : 46 0 86225270 WARFVINGES VAG 26 SE DECISION MADE: 25-OCT-13 STOCKHOLM SW SE 112 51 510(k) STATEMENT
DEVICE: COVAMESH BIOMUP S.A. 510(k) NO: K130428(TRADITIONAL) ATTN: VALERIE CENTIS PHONE NO : 33 04 86573610 8 ALLEE IRENE JOLIOT-CURIE SE DECISION MADE: 25-OCT-13 ST-PRIEST FR 69800 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OSSEOSPEED PROFILE EV DENTSPLY IMPLANTS MANUFACTURING GM510(k) NO: K130999(TRADITIONAL) ATTN: LINDA K SCHULZ PHONE NO : 858 792 1235 12264 EL CAMINO REAL, SUITE 400 SE DECISION MADE: 25-OCT-13 SAN DIEGO CA 92130 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: RAUMEDIC-PTO-MONITORING-SYSTEM RAUMEDIC AG 510(k) NO: K130529(TRADITIONAL) ATTN: REINER THIEM PHONE NO : 004 992523 592782 HERRMANN-STAUDINGER-STR. 2 SE DECISION MADE: 25-OCT-13 HELMBRECHTS GM 95233 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: MIRADRY SYSTEM MIRAMAR LABS, INC. 510(k) NO: K131162(TRADITIONAL) ATTN: KATHY O’SHAUGHNESSY PHONE NO : 408 940 8700 445 INDIO WAY SE DECISION MADE: 25-OCT-13 SUNNYVALE CA 94085 4203 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: IMMULITE 2000; TOTAL T3 CALIBRATION VERIFICATION MATERIAL, TOTAL T4 CALIBRATION VERIFICATION MATERIAL, TOTAL TBG CALIBRA SIEMENS HEALTHCARE DIAGNOSTICS INC510(k) NO: K133124(ABBREVIATED) ATTN: GARO MIMARYAN PHONE NO : 914 524 3270 511 BENEDICT AVE. SE DECISION MADE: 25-OCT-13 TARRYTOWN NY 10591 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MARAUDER CERVICAL-THORACIC SPINAL FIXATION SYSTEM INTELLIGENT IMPLANT SYSTEMS, LLC 510(k) NO: K132900(TRADITIONAL) ATTN: MICHAEL NUTT PHONE NO : 704 424 1009 3300 INTERNATIONAL AIRPORT DRIVE SSE DECISION MADE: 25-OCT-13 CHARLOTTE NC 28208 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MAMMOGRAPHY VIEWER SMV658 FUJIFILM MEDICAL SYSTEM U.S.A., IN510(k) NO: K132188(TRADITIONAL) ATTN: JYH-SHYAN LIN PHONE NO : 301 251 1092 419 WEST AVE. SE DECISION MADE: 25-OCT-13 STAMFORD CT 06902 6300 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MEDUSA VASCULAR PLUG ENDOSHAPE, INC. 510(k) NO: K123696(TRADITIONAL) ATTN: MICHAEL PARMENTER PHONE NO : 303 951 6898 2450 Central Ave Ste I SE DECISION MADE: 25-OCT-13 Boulder CO 80301 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ADVANCE 34LP LOW PROFILE PTA BALLOON DILATATION CATHETER COOK, INC. 510(k) NO: K132020(SPECIAL) ATTN: STEVEN LAWRIE PHONE NO : 812 335 3575 750 DANIELS WAY P.O. BOX 489 SE DECISION MADE: 25-OCT-13 BLOOMINGTON IN 47402 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INSTINCT ENDOSCOPIC HEMOCLIP WILSON-COOK MEDICAL INC. 510(k) NO: K132809(SPECIAL) ATTN: SCOTTIE FARIOLE PHONE NO : 336 744 0157 4900 Bethania Station Rd SE DECISION MADE: 25-OCT-13 Winston Salem NC 27105 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DIAGNOSTIC ULTRASOUND SYSTEM ADVANCED INSTRUMENTATIONS, INC. 510(k) NO: K132059(TRADITIONAL) ATTN: JORGE MILLAN PHONE NO : 305 925 1260 601 WEST 20TH STREET SE DECISION MADE: 25-OCT-13 HIALEAH FL 33010 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: TX WHEELCHAIR CHASSIS JCM SEATING SOLUTIONS LTD 510(k) NO: K131336(TRADITIONAL) ATTN: STUART R GOLDMAN PHONE NO : 512 327 9997 816 CONGRESS AVE. SUITE 1400 SE DECISION MADE: 28-OCT-13 AUSTIN TX 78701 510(k) SUMMARY AVAILABLE FROM FDA THIRD PARTY REVIEW
DEVICE: JIAJIAN POINTOSELECT DIGITAL WUXI JIANJIAN MEDICAL INSTRUMENT C510(k) NO: K123958(TRADITIONAL) ATTN: DORIS DONG PHONE NO : 86 21 31261348 RM 1706, NO. 128 SONGLE RD. SONGJISE DECISION MADE: 28-OCT-13 SHANGHAI CH 201600 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NAUTILUS SPINAL SYSTEM LIFE SPINE, INC 510(k) NO: K132866(TRADITIONAL) ATTN: RANDY LEWIS PHONE NO : 847 884 6117 2401 W. HASSELL RD SUITE 1535 SE DECISION MADE: 28-OCT-13 HOFFMAN ESTATES IL 60169 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INTEGRA JARIT HEPARIN CANNULAS, INJECTORS, AND NEEDLES INTEGRA LIFESCIENCES CORPORATION 510(k) NO: K131446(TRADITIONAL) ATTN: STEPHANIE N SHEESLEY PHONE NO : 717 840 3522 589 DAVIES DRIVE SE DECISION MADE: 28-OCT-13 YORK PA 17402 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AFFINITY FUSION CARDIOTOMY/VENOUS RESERVOIR WITH BALANCE BIOSURFACE MEDTRONIC, INC. 510(k) NO: K132972(SPECIAL) ATTN: JULIA A NELSON PHONE NO : 763 514 9844 7611 NORTHLAND DRIVE SE DECISION MADE: 28-OCT-13 MINNEAPOLIS MN 55428 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SPECTRUM SPINE FENESTRATED FACET SCREW SYSTEM SPECTRUM SPINE, LLC 510(k) NO: K132126(TRADITIONAL) ATTN: OTTIE PENDLETON PHONE NO : 404 372 0389 100 E. SOUTH MAIN ST. P.O. BOX 145SE DECISION MADE: 28-OCT-13 WAXHAW NC 28173 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AZUR PURE PERIPHERAL COIL SYSTEMS, PUSHABLE 35 MICRO VENTION, INC. 510(k) NO: K132083(TRADITIONAL) ATTN: LARAINE PANGELINA PHONE NO : 714 247 8000 1311 VALENCIA AVENUE SE DECISION MADE: 28-OCT-13 TUSTIN CA 92780 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: ITCLAMP 50 INNOVATIVE TRAUMA CARE INC 510(k) NO: K132651(TRADITIONAL) ATTN: RICHARD WAITE PHONE NO : 210 582 5850 3463 MAGIC DR. SUITE 120 SE DECISION MADE: 28-OCT-13 SAN ANTONIO TX 78229 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: MYPATCH DMS-SERVICE LLC 510(k) NO: K131190(TRADITIONAL) ATTN: LYNDA S COLE PHONE NO : 775 315 0660 2800 NEILSON WAY, STE. 1 SE DECISION MADE: 29-OCT-13 SANTA MONICA CA 90405 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: INSPIRATION VENTILATOR SYSTEM 5I/7I EVENT MEDICAL LTD 510(k) NO: K130178(TRADITIONAL) ATTN: RICK WATERS PHONE NO : 949 492 8312 971CALLE AMANECER SUITE 101 SE DECISION MADE: 29-OCT-13 SAN CLEMENTE CA 92673 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: WOUNDWASH SALINE SPRAY NEILMED PHARMACEUTICALS, INC. 510(k) NO: K123910(TRADITIONAL) ATTN: KETAN MEHTA PHONE NO : 707 525 3784 601 AVIATION BLVD. SE DECISION MADE: 29-OCT-13 SANTA ROSA CA 95403 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: 2.5MM INLINE FUSION PLATE BIOMET, INC. 510(k) NO: K131867(TRADITIONAL) ATTN: SUZANA OTANO PHONE NO : 305 269 6386 56 EAST BELL DR. BOX 587 SE DECISION MADE: 29-OCT-13 WARSAW IN 46581 0587 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: NETWORK EYE SPEARS, POINTS, DRAINS, WICKS AND SHIELDS NETWORK MEDICAL PRODUCTS, LTD. 510(k) NO: K130117(TRADITIONAL) ATTN: ALISON MARCH PHONE NO : 440 176560 955 Coronet House, Kearsley Road SE DECISION MADE: 29-OCT-13 Ripon UK HG4 2SG 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: BIOLIGN ACIF SYSTEMS, BIOLIGN STACC ACIF SYSTEM, BIOLIGN TLIF SYSTEM ELITE SURGICAL SUPPLIES (PTY) LTD.510(k) NO: K130274(TRADITIONAL) ATTN: JOHN O’TOOLE PHONE NO : 27 12 3860012 184 BESSEMER ROAD SE DECISION MADE: 29-OCT-13 PRETORIA WEST INDU., GAUTENG SF510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: REVERSE SHOULDER PROSTHESIS MONOBLOCK ENCORE MEDICAL, L.P. 510(k) NO: K130048(TRADITIONAL) ATTN: TEFFANY HUTTO PHONE NO : 512 834 6255 9800 METRIC BLVD. SE DECISION MADE: 29-OCT-13 AUSTIN TX 78758 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: GUIDOR BIORESORBABLE MATRIX BARRIER SUNSTAR AMERICAS, INC. 510(k) NO: K132325(ABBREVIATED) ATTN: DEBORAH L GRAYESKI PHONE NO : 202 550 7395 575 8TH AVE, STE 1212 SE DECISION MADE: 29-OCT-13 NEW YORK NY 10018 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: DIPLOMAT DIPLOMAT DENTAL S.R.O. 510(k) NO: K130534(TRADITIONAL) ATTN: STUART R GOLDMAN PHONE NO : 512 327 9997 816 CONGRESS AVENUE, SUITE 1400 SE DECISION MADE: 30-OCT-13 AUSTIN TX 78701 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SPIROTEL MIR MEDICAL INTL. RESEARCH SRL 510(k) NO: K130784(SPECIAL) ATTN: GERDA VAN HOUTS PHONE NO : 39 06 22754777 VIA DEL MAGGIOLINO 125 SE DECISION MADE: 30-OCT-13 ROMA IT 00155 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SUNTOUCH EYE SPEAR HUIZHOU FORYOU MEDICAL DEVICES CO,510(k) NO: K132796(TRADITIONAL) ATTN: DIANA HONG PHONE NO : 86 21 22815850 P.O. BOX 120-119 SE DECISION MADE: 30-OCT-13 SHANGHAI CH 200120 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: SMITH & NEPHEW ULTRATAPE SMITH & NEPHEW, INC. 510(k) NO: K132357(SPECIAL) ATTN: MELODY BI PHONE NO : 508 337 4035 150 MINUTEMAN RD. SE DECISION MADE: 30-OCT-13 ANDOVER MA 01810 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: AMICO CE SERIES SURGICAL LIGHTS, MIRA LED SERIES MINOR SURGICAL LIGHTS AJW TECHNOLOGY CONSULTANTS, INC. 510(k) NO: K131893(TRADITIONAL) ATTN: JANET DOUGLAS PHONE NO : 813 645 2855 445 APOLLO BEACH BLVD SE DECISION MADE: 30-OCT-13 APOLLO BEACH FL 33572 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: FLOWER BONE SCREW SET FLOWER ORTHOPEDICS CORPORATION 510(k) NO: K132248(TRADITIONAL) ATTN: JANICE M HOGAN PHONE NO : 267 675 4611 1835 MARKET ST, 29TH FLOOR SE DECISION MADE: 30-OCT-13 PHILADELPHIA PA 19103 510(k) SUMMARY AVAILABLE FROM FDA
DEVICE: OPHTHALMOSCOPES DIAGNOSTIC INSTRUMENT KITS MEDICAL DEVICES (PVT) LTD. 510(k) NO: K131719(TRADITIONAL) ATTN: TAUSEEF AJMAL PHONE NO : 009 252357 1214 WAZIRABAD RD. SE DECISION MADE: 30-OCT-13 UGOKI-SIALKOT PK PK 510(k) STATEMENT
DEVICE: MULTIVIEW HOLOGIC, INC. 510(k) NO: K132316(TRADITIONAL) ATTN: EILEEN M BOYLE PHONE NO : 781 999 7781 35 CROSBY DR. SE DECISION MADE: 30-OCT-13 BEDFORD MA 01730 510(k) STATEMENT
DEVICE: MESO BILAYER SURGICAL MESH KENSEY NASH CORPORATION DBA DSM BI510(k) NO: K132025(TRADITIONAL) ATTN: SUSAN PILEGGI PHONE NO : 484 713 2173 735 PENNSYLVANIA DRIVE SE DECISION MADE: 30-OCT-13 EXTON PA 19341 510(k) SUMMARY AVAILABLE FROM FDA
Antibody
100 Deals associated with Janice M. Brown
Login to view more data
100 Translational Medicine associated with Janice M. Brown
Login to view more data
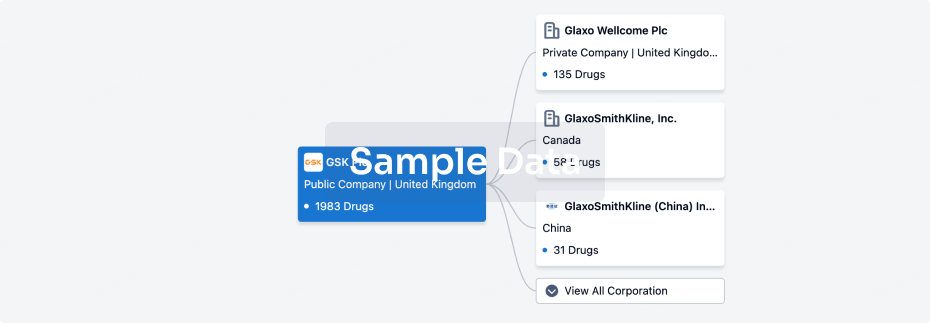
Corporation Tree
Boost your research with our corporation tree data.
login
or
Pipeline
Pipeline Snapshot as of 06 Oct 2024
No data posted
Login to keep update
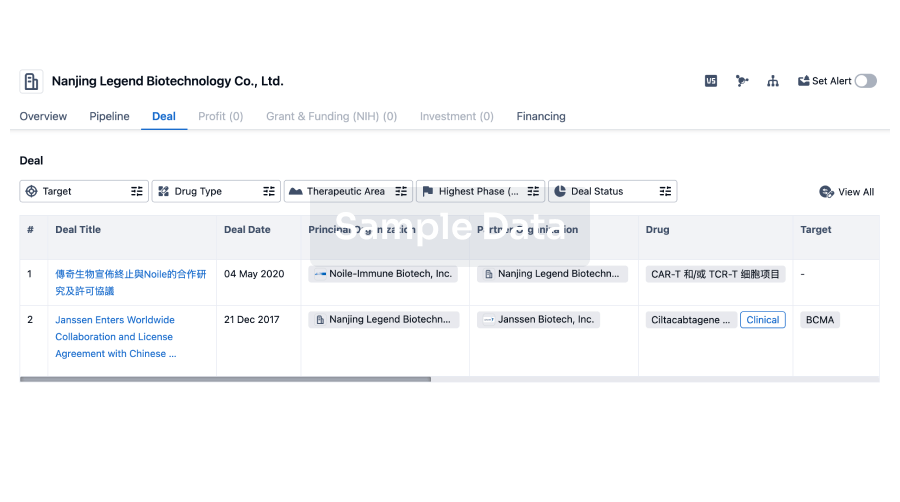
Deal
Boost your decision using our deal data.
login
or
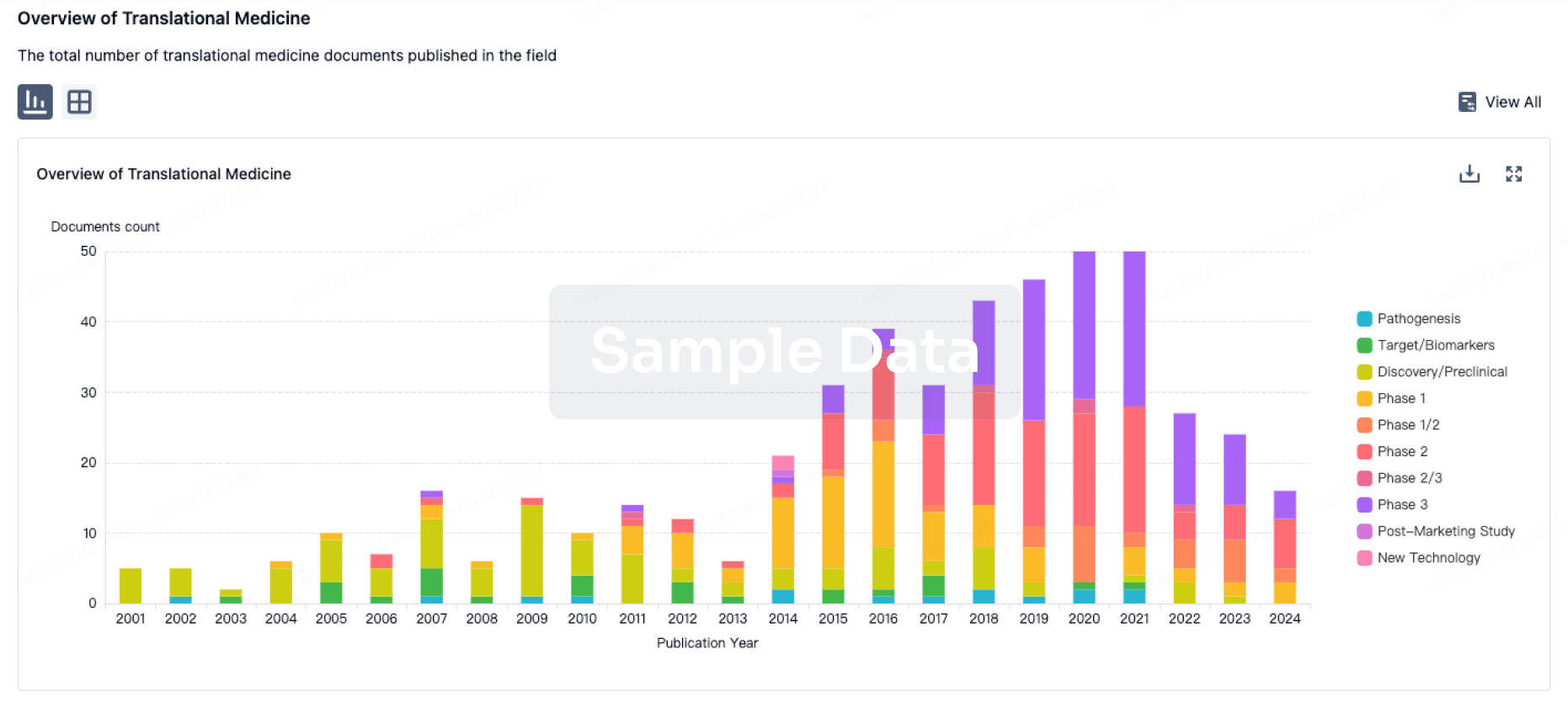
Translational Medicine
Boost your research with our translational medicine data.
login
or
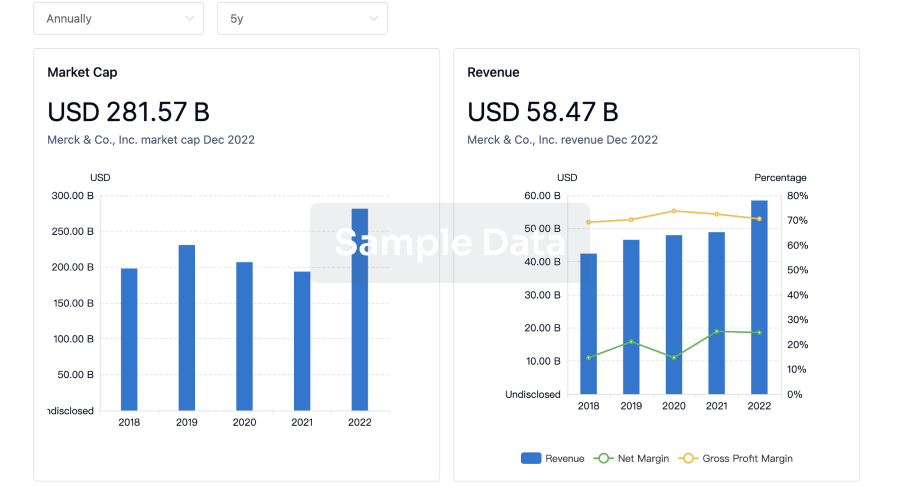
Profit
Explore the financial positions of over 360K organizations with Synapse.
login
or
Grant & Funding(NIH)
Access more than 2 million grant and funding information to elevate your research journey.
login
or
Investment
Gain insights on the latest company investments from start-ups to established corporations.
login
or
Financing
Unearth financing trends to validate and advance investment opportunities.
login
or
Chat with Hiro
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free