Request Demo
Last update 06 Jul 2025

Variagenics, Inc.
Private Company | Subsidiary Company|1992|Massachusetts, United States|
100-250
| NASDAQ: VGNX| Private Company | Subsidiary Company|1992|Massachusetts, United States|
100-250
| NASDAQ: VGNX| Last update 06 Jul 2025
Overview
Related
100 Clinical Results associated with Variagenics, Inc.
Login to view more data
0 Patents (Medical) associated with Variagenics, Inc.
Login to view more data
13
Literatures (Medical) associated with Variagenics, Inc.22 Jan 2003·Bioinformatics (Oxford, England)Q3 · BIOLOGY
Simulating haplotype blocks in the human genome
Q3 · BIOLOGY
Article
Author: Wiuf, Carsten ; Posada, David
Abstract:
Summary: A bioinformatic tool was written to simulate haplotypes and SNPs under a modified coalescent with recombination. The most important feature of this program is that it allows for the specification of non-homogeneous recombination rates, which results in the formation of the so-called ‘haplotype blocks’ of the human genome. The program also implements different mutation models and flexible demographic histories. The samples generated can be very useful to better understand the architecture of the human genome and to investigate its impact in association studies searching for disease genes.Availability: The SNPsim package is available at http://www.evolgenics.com/softwareContact: dposada@evolgenics.com* To whom correspondence should be addressed.
01 Jul 2002·Pharmacogenomics
From the Lab to the Clinic: Integration of Pharmacogenics Into Clinical Development
Author: Hoban, Carolyn J
In the field of clinical genomics, pharmacogenic markers are being evaluated in order to discriminate between potential responders and nonresponders. In strategic alignment with the introduction of molecularly-designed drug candidates, the use of companion molecular diagnostic tests will help targeted drugs to achieve a greater therapeutic index. Realization of these potential advantages is evident from the increasing number of investigational new drug (IND) and new drug applications (NDA) as well as clinical study protocols that include tissue banking and pharmacogenic studies as integral component. An appraisal of critical issues and necessary steps to facilitate full integration of pharmacogenic tools in clinical development programs and medical care is presented. Expectation, promise, innovation, optimism are words which describe the enthusiasm for introducing pharmacogenic studies in the drug development process. The clinical utility of pharmacogenics to predict progression of disease or response to a drug therapy is a far reaching tool that will be added to treatment guidelines as part of the decision tree for evidence-based medicines. Veterans of the war on cancer are now eager to decipher the mysteries held within the human genome whose workings are subverted during the multiple steps that lead to oncogenesis. It is estimated that pharmaceutical companies will have 10,650 drugs in the pipeline by 2003 [101]. To evaluate the drugs in development in 2002, > 2.3 million people in the US participated in clinical trials [1]. The pharmaceutical industry drives the clinical development of drugs from Phase I through to Phase IV. However, both government agencies and pharmaceutical companies commission Phase I through to Phase III trials. In 2001, there were 750,000 trials funded by the government, with industry funding 850,000 trials in the US alone. In postmarket/Phase IV studies, industry dominated by funding 700,000 trials in the US. Oncology and AIDS/HIV drug therapy are the two areas where pharmacogenic markers have been successfully adopted as Food & Drug Administration (FDA)-approved molecular diagnostic tests. The worldwide cancer drug market, valued at US$30 billion, has doubled since 1998 and is rapidly expanding, with almost 400 cancerrelated drugs under development [101]. In oncology, there is an imperative to develop markers that apprise the physician and patient on whether to withhold therapies that might have serious adverse effects or treat with a specific drug that has the greatest possible benefit for the patient. Pharmacogenic studies are being integrated into every phase of drug development to select compounds with an assurance of safety. As a tool to eliminate failure early in development, pharmacogenic studies are valuable components of the drug development programme. Almost 80% of drugs fail due to safety concerns or poor efficacy. A review of pharmaceutical companies (1964–1985) reported that [2]:
01 Aug 1999·Molecular pharmacologyQ3 · MEDICINE
Selective Killing of Cancer Cells Based on Loss of Heterozygosity and Normal Variation in the Human Genome: A New Paradigm for Anticancer Drug Therapy
Q3 · MEDICINE
Article
Author: Rioux, Patrice ; Olson, Jeffrey C. ; Schievella, Andrea R. ; Stanton, Vincent P. Jr. ; Housman, David E. ; Monia, Brett P. ; Burns, Erica ; Basilion, James P. ; Lemonidis, Kristina M.
Most drugs for cancer therapy are targeted to relative differences in the biological characteristics of cancer cells and normal cells. The therapeutic index of such drugs is theoretically limited by the magnitude of such differences, and most anticancer drugs have considerable toxicity to normal cells. Here we describe a new approach for developing anticancer drugs. This approach, termed variagenic targeting, exploits the absolute difference in the genotype of normal cells and cancer cells arising from normal gene sequence variation in essential genes and loss of heterozygosity (LOH) occurring during oncogenesis. The technology involves identifying genes that are: 1) essential for cell survival; 2) are expressed as multiple alleles in the normal population because of the presence of one or more nucleotide polymorphisms; and 3) are frequently subject to LOH in several common cancers. An allele-specific drug inhibiting the essential gene remaining in cancer cells would be lethal to the malignant cell and would have minimal toxicity to the normal heterozygous cell that retains the drug-insensitive allele. With antisense oligonucleotides designed to target two alternative alleles of replication protein A, 70-kDa subunit (RPA70) we demonstrate in vitro selective killing of cancer cells that contain only the sensitive allele of the target gene without killing cells expressing the alternative RPA70 allele. Additionally, we identify several other candidate genes for variagenic targeting. This technology represents a new approach for the discovery of agents with high therapeutics indices for treating cancer and other proliferative disorders.
4
News (Medical) associated with Variagenics, Inc.03 Jan 2024
SOUTH SAN FRANCISCO, Calif.--(
BUSINESS WIRE
)--Switch Therapeutics, a biotechnology company pioneering a new way to use RNA science to treat diseases utilizing its proprietary CASi (Conditionally Activated siRNA) platform, today announced the appointment of Charles Allerson, Ph.D., as Chief Technology Officer (CTO). He succeeds Si-ping Han, Ph.D., co-founder of Switch Therapeutics, who will transition to an advisory role and serve on Switch’s Scientific Advisory Board.
“
Charles joins our leadership team with extensive expertise in oligonucleotide chemistry and RNA-based therapeutics, and we are delighted to welcome him to Switch. He will play a crucial role as we continue driving our novel CASi platform forward for the treatment of central nervous system (CNS) diseases,” said Dee Datta, Ph.D., co-founder and CEO of Switch Therapeutics. “
We also want to thank Dr. Han for his significant impact at Switch by leading the invention of the CASi platform, where efficient self-delivery, uptake and potency have been demonstrated
in vivo
. We look forward to his continued contributions as he joins our Scientific Advisory Board – a team of esteemed leaders in drug discovery, RNA science and CNS research – and are excited for what’s ahead in 2024.”
Dr. Allerson joins Switch Therapeutics after dedicating more than 20 years of his career building nucleic acid chemical tools and applying them to the development of RNA-based therapeutics. Most recently, he served as Senior Vice President of Chemistry at DTx Pharma from 2019 until its acquisition by Novartis in 2023. Dr. Allerson led efforts to develop the FALCON platform for the extrahepatic delivery of RNA therapeutics and to translate this platform into a portfolio of preclinical assets. Dr. Allerson began his career as a chemist at Variagenics, and subsequently joined the Medicinal Chemistry team at Isis (now Ionis) Pharmaceuticals. Following his role at Ionis, he led early-stage and development stage chemistry at Regulus Therapeutics, and subsequently joined Stoke Therapeutics as Vice President of Chemistry where he helped prepare the company’s TANGO platform for the clinic. Dr. Allerson earned his B.S. in Chemistry from Lafayette College, his Ph.D. in Chemistry from Harvard University, and spent several years as a postdoctoral fellow at the National Institutes of Health prior to his move into industry.
“
I am thrilled to join Switch at this pivotal time and look forward to partnering with my new colleagues to advance CASi into the clinic and beyond,” said Dr. Allerson, Ph.D. “
The three-stranded structures of CASi provide an opportunity to deploy novel chemistry and design strategies that will create unique medicines and open new therapeutic target spaces. I am eager to continue the innovation of the CASi platform, enabling Switch to translate the power of RNA science into a new generation of RNA medicines.”
About Switch Therapeutics
Switch Therapeutics is an emerging biotechnology company pioneering a new type of medicine that integrates nucleic acid nanotechnology and RNA interference (RNAi) science, with the goal of treating a range of diseases – affecting the central nervous system, peripheral nervous system and systemic indications – with significant unmet need. The company’s proprietary, three stranded RNA platform named CASi (Conditionally Activated siRNA), is based on technology developed by renowned researchers in the field of RNA and drug development from Caltech, Harvard Medical School and City of Hope. CASi combines advantageous properties of both single and double-stranded RNAs in a single molecule, allowing for improved biodistribution, functionality, efficacy and cell selective RNAi activity. Switch is based in South San Francisco, California. For more information, connect with Switch on
LinkedIn
or visit
www.switchthera.com
.
Executive ChangeAcquisitionOligonucleotide
17 Jul 2023
Pictured: Illustration of scientists working on biomedical research/iStock, Olga Kurbatova
There is a stark divide in public depictions of pharmaceutical innovation. The biopharma industry proudly advertises its accomplishments in launching new drugs for cancer, neurodegenerative disease, diabetes, weight loss and more without acknowledging the billions of dollars of government funding for basic and applied biomedical science that enabled these advances. Industry critics, meanwhile, argue that this government funding is responsible for pharmaceutical innovation, going as far as to propose that patients “pay twice” for medicines, once in the form of taxes that underwrite government research and again in purchasing these products.
Two recent papers published by my colleagues and me in the Journal of the American Medical Association’s Health Forum suggest that neither view is accurate. Our research illustrates the essential complementarity of government and industry spending on new medicines.
True Partners in Drug Development
There is no disagreement on the process leading to new drugs. Contemporary pharmaceutical innovation is based on a deep foundation of basic research that describes the biology of both health and disease as well as mechanisms and targets for medicines that may preserve or restore health, or at least reduce the morbidity and mortality associated with disease.
This leads to applied research to identify candidate drug molecules and understand their activity in laboratory and animal models. Promising compounds then enter clinical development, which explicitly focuses on the human trials necessary to establish the efficacy and safety of these products, establish manufacturing capacity and satisfy the requirements for marketing approval by the FDA or other regulatory agencies.
My colleagues and I at the Center for Integration of Science and Industry at Bentley University have undertaken a series of studies, supported by the Institute for New Economic Thinking, National Biomedical Research Foundation and National Pharmaceutical Council, to characterize the U.S. government’s investment in this process through the National Institutes of Health (NIH).
In our first two studies, published in 2018 and 2020, we identified $187 billion in NIH funding for research related to the 356 drugs approved between 2010 and 2019. Of this amount, ~83% represents basic research related to the drug target and ~17% applied research related to the drugs themselves. This April, we published a third study demonstrating that the value of the NIH investment in new drug approvals was comparable to reported levels of investment by industry.
A subsequent study, published July 14, focused explicitly on NIH funding for clinical development. Here we demonstrated that only 3.3% of all NIH funding related to new drug approvals pertains to clinical trials. That means that when it comes to this phase of drug development, the NIH invests only ~10% of the spending from the biopharma industry. We further showed that >90% of the NIH funding contributing to clinical development was in the form of grants supporting clinical consortia or centers, or translational science methods, capacity or training, rather than the clinical trials themselves.
© Nicole Bean
Taken together, these studies demonstrate the essential complementary nature of government and industry investments in pharmaceutical innovation. Industry has limited incentive to invest in basic science, which typically produces results that may not be patentable, are widely disseminated in scientific journals and provide uncertain returns to the company making the investment. By funding this research, the government reduces the industry’s total investment cost in new drug approvals by approximately half and creates economic efficiencies through spillover effects in which multiple companies are able to use the same scientific knowledge to develop different products.
In contrast, industry has multiple incentives to focus on clinical development, which not only allows commercialization of new products but also generates patents, know-how and trade secrets that contribute to market exclusivity and industry’s ability to set prices and maximize profits.
Ensuring Equitable Returns for All Parties
It is increasingly recognized that government plays an essential role as an early investor in technological innovation, making investments in research that is too uncertain or products that are too distant for industry to justify to their shareholders.
These investments reduce the risk of subsequent industry investments necessary to develop and commercialize practical applications of this research. As such, economists argue that taxpayers could expect returns on their investments in this process commensurate with those of pharmaceutical companies or their shareholders and the relative risks of these contributions. These may include both economic returns to public-sector organizations and social returns in the form of improved health or wellness, lower healthcare costs, new scientific insights or jobs.
Any attempt to deliver such public returns is undermined, however, by the disparate descriptions of the investments leading to new drugs promoted by the pharmaceutical industry and its critics—descriptions that belie the essential interdependence of public and private sector investments in this arena. Pharmaceutical innovation is not advanced by rhetoric that dismisses the contributions of either sector nor by partisan pressures that advocate for one sector at the expense of the other. Rather, effective innovation may be advanced through greater transparency concerning the investment and activities necessary for bringing drugs to market.
Fred D. Ledley/Bentley University
Only by acknowledging the contributions of both sectors will society be able to address unmet medical needs, identify potential economic and operational synergies that could make innovation more efficient and assure equitable returns on both public and private investments in pharmaceutical innovation.
Fred Ledley, M.D., is the director of the Center for Integration of Science and Industry at Bentley University in Massachusetts. He has served previously on the faculties of the Howard Hughes Medical Institute and Baylor College of Medicine, as a Founder and VP of R&D at GeneMedicine Inc. and as president and CEO of Variagenics Inc.
Executive Change
14 Jul 2023
Pictured: Illustration of scientists working on bi
Bringing new drugs to the market costs billions of dollars. It could not be done without investments by both the NIH and biopharma companies.
Pictured: Illustration of scientists working on biomedical research/iStock,
Olga Kurbatova
There is a stark divide in public depictions of pharmaceutical innovation. The biopharma industry proudly advertises its accomplishments in launching new drugs for cancer, neurodegenerative disease, diabetes, weight loss and more without acknowledging the billions of dollars of government funding for basic and applied biomedical science that enabled these advances. Industry critics, meanwhile, argue that this government funding is responsible for pharmaceutical innovation, going as far as to propose that patients “pay twice” for medicines, once in the form of taxes that underwrite government research and again in purchasing these products.
Two recent papers published by my colleagues and me in the
Journal of the American Medical Association
’s Health Forum suggest that neither view is accurate. Our research illustrates the essential complementarity of government and industry spending on new medicines.
True Partners in Drug Development
There is no disagreement on the process leading to new drugs. Contemporary pharmaceutical innovation is based on a deep foundation of basic research that describes the biology of both health and disease as well as mechanisms and targets for medicines that may preserve or restore health, or at least reduce the morbidity and mortality associated with disease.
This leads to applied research to identify candidate drug molecules and understand their activity in laboratory and animal models. Promising compounds then enter clinical development, which explicitly focuses on the human trials necessary to establish the efficacy and safety of these products, establish manufacturing capacity and satisfy the requirements for marketing approval by the FDA or other regulatory agencies.
My colleagues and I at the Center for Integration of Science and Industry at Bentley University have undertaken a series of studies, supported by the Institute for New Economic Thinking, National Biomedical Research Foundation and National Pharmaceutical Council, to characterize the U.S. government’s investment in this process through the National Institutes of Health (NIH).
In our first two studies, published in
2018
and
2020
, we identified $187 billion in NIH funding for research related to the 356 drugs approved between 2010 and 2019. Of this amount, ~83% represents basic research related to the drug target and ~17% applied research related to the drugs themselves. This April, we published a third
study
demonstrating that the value of the NIH investment in new drug approvals was comparable to reported levels of investment by industry.
A subsequent
study
, published July 14, focused explicitly on NIH funding for clinical development. Here we demonstrated that only 3.3% of all NIH funding related to new drug approvals pertains to clinical trials. That means that when it comes to this phase of drug development, the NIH invests only ~10% of the spending from the biopharma industry. We further showed that >90% of the NIH funding contributing to clinical development was in the form of grants supporting clinical consortia or centers, or translational science methods, capacity or training, rather than the clinical trials themselves.
© Nicole Bean
Taken together, these studies demonstrate the essential complementary nature of government and industry investments in pharmaceutical innovation. Industry has limited incentive to invest in basic science, which typically produces results that may not be patentable, are widely disseminated in scientific journals and provide uncertain returns to the company making the investment. By funding this research, the government reduces the industry’s total investment cost in new drug approvals by approximately half and creates economic efficiencies through spillover effects in which multiple companies are able to use the same scientific knowledge to develop different products.
In contrast, industry has multiple incentives to focus on clinical development, which not only allows commercialization of new products but also generates patents, know-how and trade secrets that contribute to market exclusivity and industry’s ability to set prices and maximize profits.
Ensuring Equitable Returns for All Parties
It is increasingly recognized that government plays an essential role as an early investor in technological innovation, making investments in research that is too uncertain or products that are too distant for industry to justify to their shareholders.
These investments reduce the risk of subsequent industry investments necessary to develop and commercialize practical applications of this research. As such, economists argue that taxpayers could expect returns on their investments in this process commensurate with those of pharmaceutical companies or their shareholders and the relative risks of these contributions. These may include both economic returns to public-sector organizations and social returns in the form of improved health or wellness, lower healthcare costs, new scientific insights or jobs.
Any attempt to deliver such public returns is undermined, however, by the disparate descriptions of the investments leading to new drugs promoted by the pharmaceutical industry and its critics—descriptions that belie the essential interdependence of public and private sector investments in this arena. Pharmaceutical innovation is not advanced by rhetoric that dismisses the contributions of either sector nor by partisan pressures that advocate for one sector at the expense of the other. Rather, effective innovation may be advanced through greater transparency concerning the investment and activities necessary for bringing drugs to market.
Fred D. Ledley/Bentley University
Jamison Wexler
Only by acknowledging the contributions of both sectors will society be able to address unmet medical needs, identify potential economic and operational synergies that could make innovation more efficient and assure equitable returns on both public and private investments in pharmaceutical innovation.
Fred Ledley, M.D., is the director of the Center for Integration of Science and Industry at Bentley University in Massachusetts. He has served previously on the faculties of the Howard Hughes Medical Institute and Baylor College of Medicine, as a Founder and VP of R&D at GeneMedicine Inc. and as president and CEO of Variagenics Inc.
Executive Change
100 Deals associated with Variagenics, Inc.
Login to view more data
100 Translational Medicine associated with Variagenics, Inc.
Login to view more data
Corporation Tree
Boost your research with our corporation tree data.
login
or
Pipeline
Pipeline Snapshot as of 29 Aug 2025
No data posted
Login to keep update
Deal
Boost your decision using our deal data.
login
or
Translational Medicine
Boost your research with our translational medicine data.
login
or
Profit
Explore the financial positions of over 360K organizations with Synapse.
login
or
Grant & Funding(NIH)
Access more than 2 million grant and funding information to elevate your research journey.
login
or
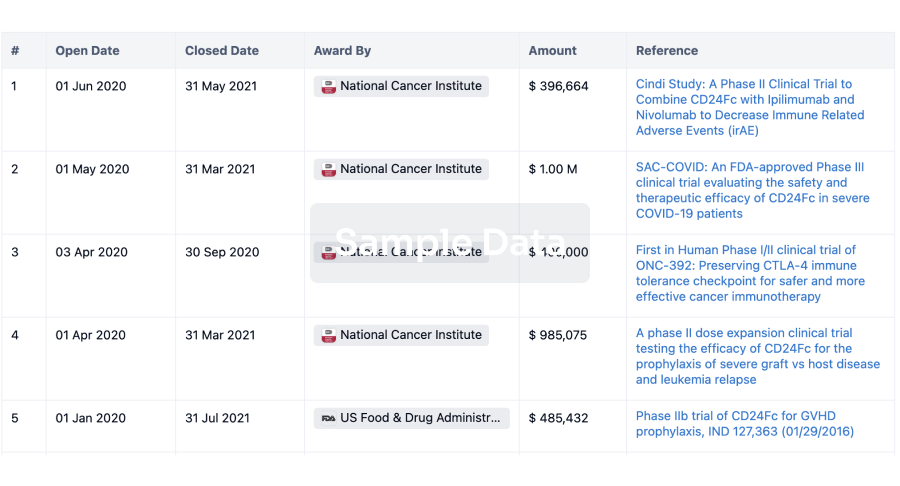
Investment
Gain insights on the latest company investments from start-ups to established corporations.
login
or
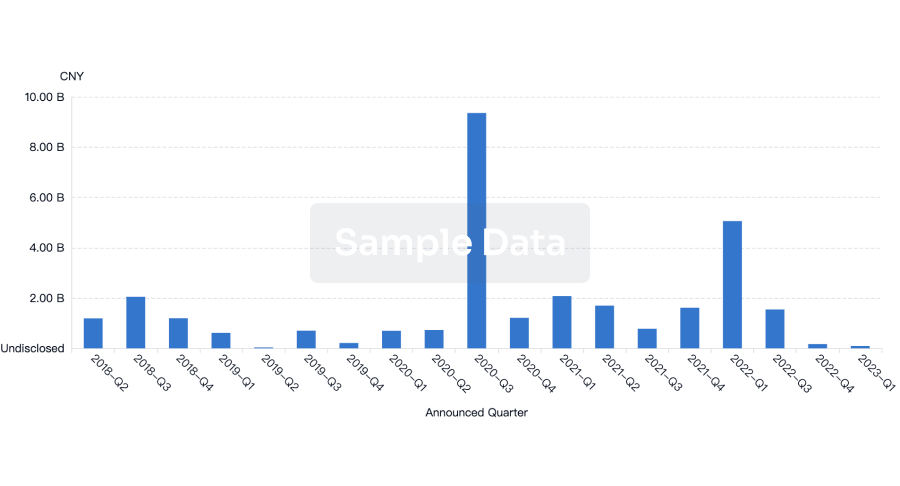
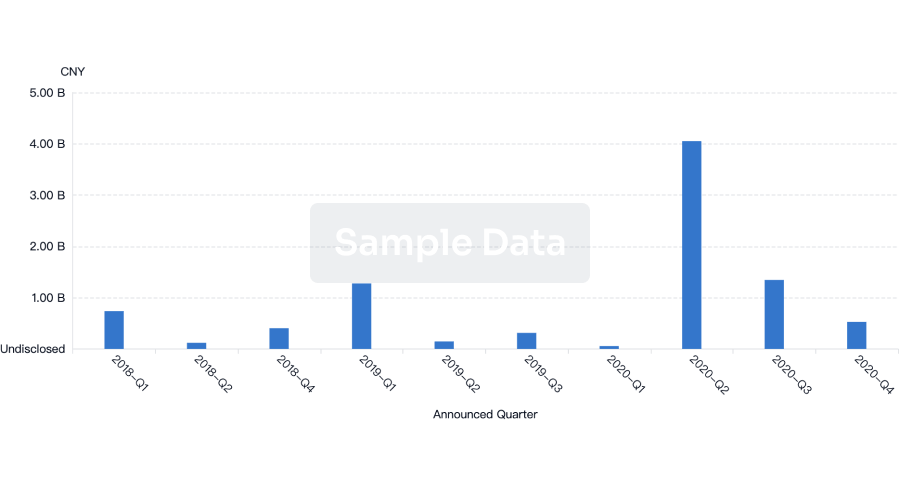
Financing
Unearth financing trends to validate and advance investment opportunities.
login
or
AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free