Request Demo
Last update 08 May 2025

Geneseeq Technology, Inc.
Last update 08 May 2025
Overview
Related
100 Clinical Results associated with Geneseeq Technology, Inc.
Login to view more data
0 Patents (Medical) associated with Geneseeq Technology, Inc.
Login to view more data
159
Literatures (Medical) associated with Geneseeq Technology, Inc.22 Mar 2024·Cancer Research
Abstract 7408: Robust profiling of cancer-related gene fusions: Analytical validation of PANCARNA for multiple cancer types
Author: Liu, Sisi ; Wu, Shuyu ; Liu, Rui ; Tang, Haimeng ; Shao, Yang ; Wu, Xue ; Bao, Hua
04 Apr 2023·Cancer Research
Abstract 940: Comprehensive characterization of MET exon 14 skipping mutations in non-small cell lung cancer
Author: Zhang, Yaru ; Wu, Xue ; Tang, Haimeng ; Shao, Yang ; Yin, Jiani C. ; Pang, Jiaohui ; Lin, Li
04 Apr 2023·Cancer Research
Abstract 939: Genomic characterization of PMBCL, cHL and DLBCL utilizing tissue and liquid biopsies
Author: Shao, Yang ; Yin, Jiani C. ; Shen, Lu ; Tang, Haimeng ; Zhu, Liuqing ; Wang, Xiaonan
13
News (Medical) associated with Geneseeq Technology, Inc.03 Jan 2024
TORONTO, Jan. 3, 2024 /PRNewswire/ - Geneseeq announced that its multi-cancer early detection solution, CanScan™, has been granted Breakthrough Device Designation by the US Food and Drug Administration (FDA).
CanScan™ utilizes low-depth whole-genome sequencing (WGS) on circulating cell-free DNA (cfDNA) from a single tube of peripheral blood, extracting genetic and fragmentomic features to detect early cancer signals with 99% specificity and predict the tissue of origin (TOO) of cancers to help guide next steps for cancer diagnosis. CanScan™ exhibits promising potential to address unmet medical needs in clinical diagnosis and treatment, particularly for individuals aged 50 and above with an average risk of cancer. The test outperforms current standard of care (SOC) screening methods in common cancer types, such as prostate, lung and liver cancers. It also detects cancer types currently without effective SOC screening methods, such as esophagus, endometrial, gastric, pancreatic cancers and lymphoma.
Built on Geneseeq's highly sensitive MERCURY™ multi-omics technology, the performance of CanScan™ has been validated in large-scale clinical study series, DECIPHER (Detecting Early Cancer by Inspecting ctDNA Features), in over thirteen cancer types. CanScan™ is currently under real-world evaluation in the Jinling Cohort (NCT06011694), a large-scale prospective multi-center trial. The ongoing recruitment of 15,000 individuals for the phase I trial within the Jinling Cohort is approaching completion.
"The Jinling Cohort aims to further validate the technical performance of CanScan™ in the targeted screening population," Dr.Xue Wu, Geneseeq Toronto's CEO said in a statement, noting that "we will release more results of the Jinling Cohort in 2024."
This FDA Breakthrough Device Designation follows the CanScan™ assay kit's CE approval in January 2023, marking another significant recognition from an internationally authoritative institution.
SOURCE Geneseeq Technology Inc.
Phase 1
13 Oct 2023
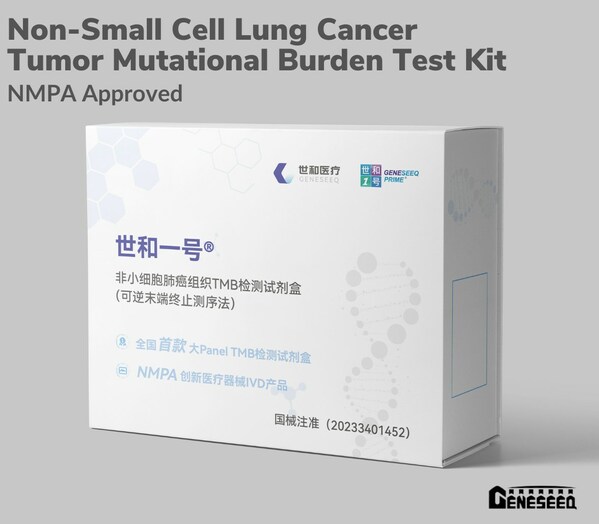
TORONTO, Oct. 13, 2023 /PRNewswire/ - Geneseeq Technology Inc. announced on Thursday that Geneseeq's Non-Small Cell Lung Cancer Tumor Mutational Burden Test Kit (Reversible terminator sequencing method) (NSCLC TMB Kit) has gained approval from the Chinese National Medical Products Administration (NMPA) on October 12, 2023 as a Breakthrough Medical Device.
Continue Reading
Non-small cell lung cancer tumor mutational burden test kit approved by NMPA (CNW Group/Geneseeq Technology Inc.)
This NSCLC TMB kit entered the NMPA special examination and approval procedures for Breakthrough Medical Devices in January 2020. With the NMPA approval, this kit is used for in vitro qualitative detection of TMB in FFPE tissue samples from patients with EGFR/ALK-negative non-squamous NSCLC. This is the first next-generation sequencing (NGS)-based TMB test kit approved in China with a large gene panel covering 425 cancer-associated genes (GENESEEQPRIME™). TMB is a predictive biomarker for the efficacy of immune checkpoint inhibitor therapy.
Prior to this, Geneseeq already had one NGS-based CDx kit for NSCLC,
ESSENCARE™ (EGFR/ALK/ROS1/BRAF/KRAS/HER2 mutation testing kit), approved by the NMPA through the Breakthrough Medical Devices pathway. GENESEEQPRIME™ was also CE-IVD marked by the European Medicines Agency in August 2023 for detecting single nucleotide variants, gene amplifications/deletions, translocations, TMB, and microsatellite instability in patients with solid tumors.
"This approval will significantly benefit the clinical implementation of immunotherapy in China with a standardized TMB assessment assay," Dr. Yang Shao, founder and CEO of Geneseeq Group, said in a statement.
SOURCE Geneseeq Technology Inc.
OligonucleotideClinical Study
23 Aug 2023
GeneseeqPrime and GeneseeqPrime HRD are indicated for solid tumour genomic profiling while Hemasalus DNA/Hemarna RNA is approved for haematological cancer genomic profiling
Geneseeq gets CE Mark for three cancer test kits. (Credit: National Cancer Institute on Unsplash)
Geneseeq Technology has secured the European Union’s CE Mark approval for GENESEEQ Homologous Recombination Deficiency Detection Kit (GeneseeqPrime HRD), GENESEEQPRIME NGS Tumor Gene Detection Kit (GeneseeqPrime), and GENESEEQ Blood Cancer Gene Detection Kit (Hemasalus DNA/Hemarna RNA).
The Canada-based precision oncology company said that all three tests are next-generation sequencing (NGS)-based cancer genetic testing kits.
GeneseeqPrime and GeneseeqPrime HRD are indicated for solid tumour genomic profiling while Hemasalus DNA/Hemarna RNA is approved for haematological cancer genomic profiling.
According to Geneseeq Technology, the GeneseeqPrime kit uses a set of 437 genes that are associated with solid tumours. The reportable range consists of tumour mutational burden (TMB), microsatellite instability (MSI), and DNA mismatch repair (MMR) genes.
Additionally, the range includes single nucleotide variants (SNVs), insertions and deletions (indels), copy number variants (CNVs), and gene translocations and large genomic rearrangements (LGR).
It provides information on key oncogenic genes and potential drug resistance pathways to guide therapeutic decisions, the precision oncology company said.
In addition to the data offered by GeneseeqPrime, the GeneseeqPrime HRD report integrates the homologous recombination deficiency (HRD) status to suggest PARP inhibitor therapies.
The third test kit which received the CE Mark, which is Hemasalus DNA/Hemarna RNA, examines genetic variants and translocations in 475 genes at the DNA level. This is in addition to detecting translocations in 232 genes at the RNA level.
The kit is said to simplify clinical diagnosis and therapy planning for people with haematological malignancies.
Furthermore, the Geneseeq cancer genetic testing kit features an assay kit for sequencing, an automated reporting system, and an end-to-end service that includes assay setup and customer assistance.
Geneseeq Canada CEO Xue Wu said: “The CE Mark validation for the GeneseeqPrime, GeneseeqPrime HRD, and Hemasalus DNA/Hemarna RNA kits underscores our dedication’s commitment to high quality testing standards on a global level.”
The Canadian company presently offers five CE-marked cancer genetic testing kits designed for different clinical scenarios.
Earlier this year, Geneseeq launched the CE-marked GENESEEQ multi-cancer minimal residual disease detection (Shielding ULTRA MRD) and GENESEEQ multi-cancer early detection (CanScan MCED) kits.
siRNAOligonucleotide
100 Deals associated with Geneseeq Technology, Inc.
Login to view more data
100 Translational Medicine associated with Geneseeq Technology, Inc.
Login to view more data
Corporation Tree
Boost your research with our corporation tree data.
login
or

Pipeline
Pipeline Snapshot as of 17 May 2026
No data posted
Login to keep update
Deal
Boost your decision using our deal data.
login
or

Translational Medicine
Boost your research with our translational medicine data.
login
or

Profit
Explore the financial positions of over 360K organizations with Synapse.
login
or

Grant & Funding(NIH)
Access more than 2 million grant and funding information to elevate your research journey.
login
or

Investment
Gain insights on the latest company investments from start-ups to established corporations.
login
or

Financing
Unearth financing trends to validate and advance investment opportunities.
login
or

AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free