Request Demo
Last update 08 May 2025
CALB2
Last update 08 May 2025
Basic Info
Synonyms 29 kDa calbindin, CAB29, CAL2 + [4] |
Introduction Calretinin is a calcium-binding protein which is abundant in auditory neurons. |
Related
100 Clinical Results associated with CALB2
Login to view more data
100 Translational Medicine associated with CALB2
Login to view more data
0 Patents (Medical) associated with CALB2
Login to view more data
4,030
Literatures (Medical) associated with CALB201 Jun 2025·Molecular and Cellular Neuroscience
Olfactory bulb interneurons – The developmental timeline and targeting defined by embryonic neurogenesis
Article
Author: Martin-Lopez, Eduardo ; Greer, Charles A ; Spence, Natalie J ; Brennan, Bowen ; Lefèvre, Marion ; Lange, Nathaniel W ; Han, Kimberly
01 May 2025·Pain
Spinal lumbar Urocortin 3-expressing neurons are associated with both scratching and Compound 48/80-induced sensations
Article
Author: Ceder, Mikaela M. ; Lagerström, Malin C. ; Weman, Hannah M. ; Öhman-Mägi, Caroline ; Ahemaiti, Aikeremu ; Bengtsson, Erica ; Magnusson, Kajsa A. ; Henriksson, Katharina ; Franck, Marina C. M. ; Koning, Harmen K.
01 Apr 2025·Thoracic Cancer
Cytoplasmic HuR Expression Enhances Chemoresistance in Pleural Mesothelioma Through Increased Expression of CALB2 , Promotion of the E2F Pathway, and Suppression of the p53 Pathway
Article
Author: Okubo, Kenichi ; Taniguchi, Yusuke ; Sugita, Keisuke ; Kurata, Morito ; Ishibashi, Hironori ; Kirimura, Susumu ; Kinowaki, Yuko
Analysis
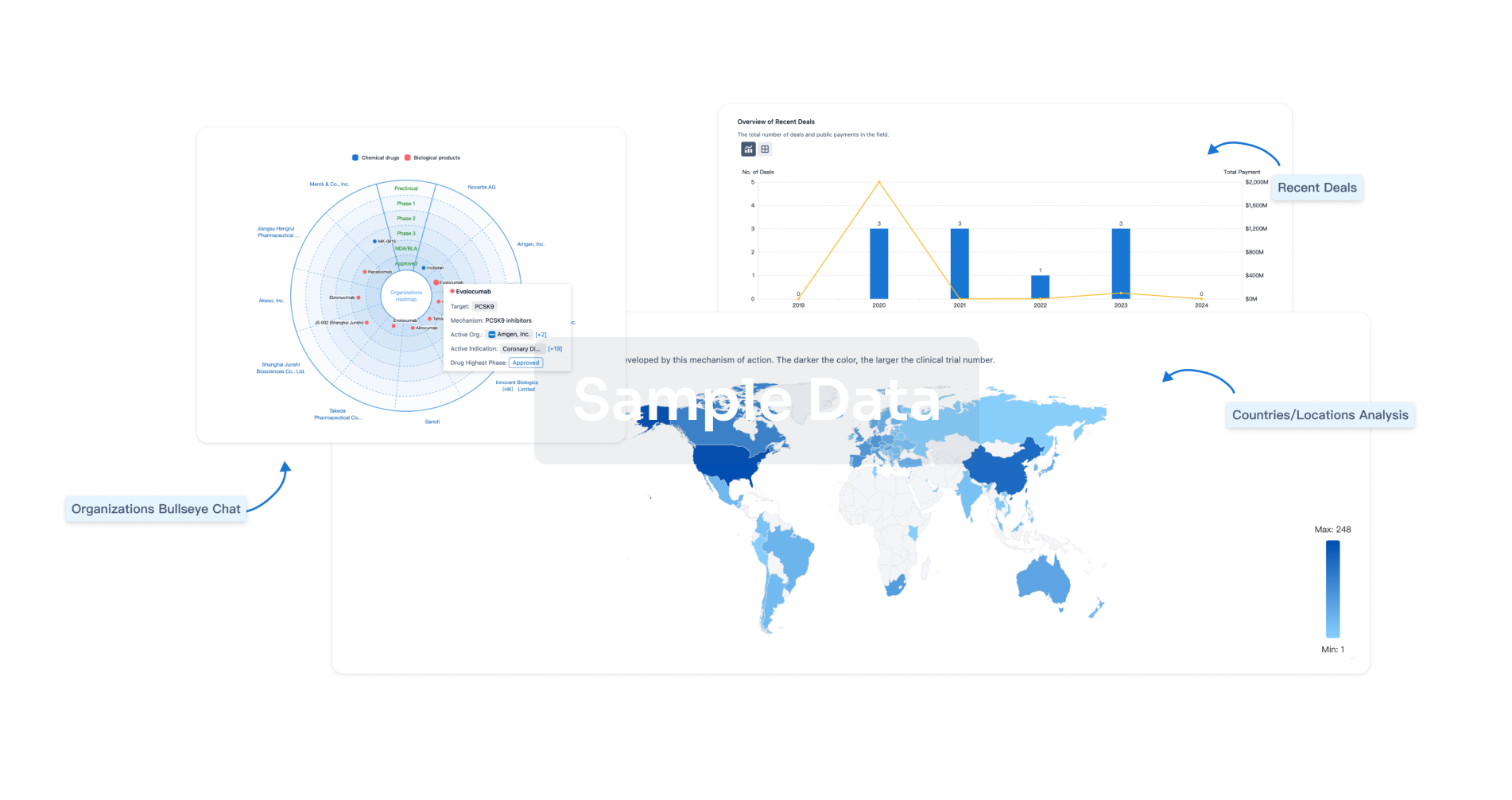
Perform a panoramic analysis of this field.
login
or
AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free