Market Analysis of Aceclofenac in the USA: Opportunities and Barriers for Market Entry
Overview
Based on our comprehensive analysis, aceclofenac is not currently approved in the USA market. However, it is a globally established non-steroidal anti-inflammatory drug (NSAID) with approvals in China and the European Union. The drug is primarily indicated for rheumatoid arthritis, ankylosing spondylitis, osteoarthritis, and other rheumatic diseases. It functions as a COX inhibitor, targeting the COXs pathway. While there is significant market presence in other countries with multiple manufacturers and formulations, particularly in China, the US market remains unexplored for this product.
Detailed Description
Drug Information
Aceclofenac was developed by Prodes SA and is approved in multiple countries. The drug is not currently approved in the USA.
China Approvals:
| Approval Number | Approval Company | Approval Date | Dosage Form | Specification | Administration Route | Indication | Approval Status |
|---|---|---|---|---|---|---|---|
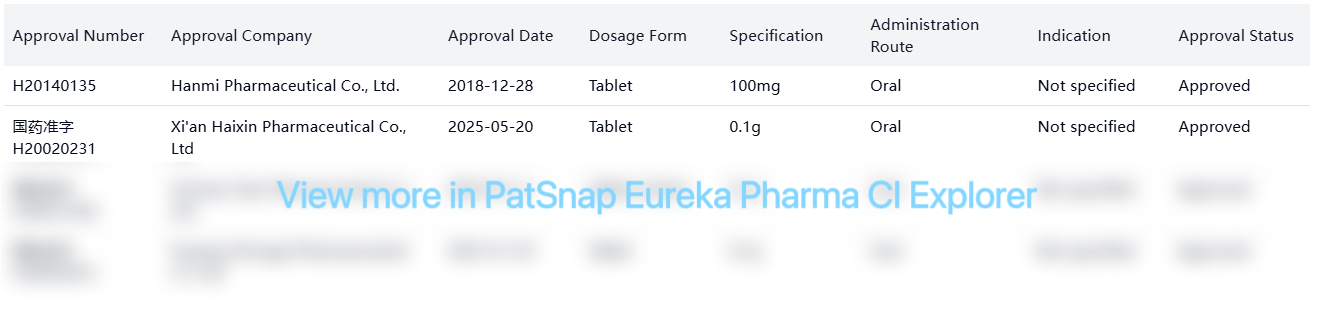
| H20140135 | Hanmi Pharmaceutical Co., Ltd. | 2018-12-28 | Tablet | 100mg | Oral | Not specified | Approved |
| 国药准字H20020231 | Xi'an Haixin Pharmaceutical Co., Ltd | 2025-05-20 | Tablet | 0.1g | Oral | Not specified | Approved |
| 国药准字H20031249 | Sichuan Viao Pharmaceutical Co., Ltd. | 2024-05-11 | Tablet, Enteric | 0.1g | Oral | Not specified | Approved |
| 国药准字H20050253 | Puyang Zhongyi Pharmaceutical Co. Ltd. | 2025-01-20 | Tablet | 0.1g | Oral | Not specified | Approved |
EU Approvals:
Structure:
Patent Barrier Analysis
Registration Patent Analysis
The core patents for aceclofenac have expired, indicating no primary patent barriers for generic entry:
| Patent Number | Simple Legal Status | Application Date | Estimated Expiry | Patent Type | Applicant | Source |
|---|---|---|---|---|---|---|
| EP0119932A1 | Inactive | 1984-03-19 | 2004-03-19 | Not specified | Prodes SA | SPC, PED |
| EP0119932B1 | Inactive | 1984-03-19 | 2004-03-19 | Not specified | Prodes SA | SPC, PED |
Other Patent Barrier Analysis
There are several secondary patents related to aceclofenac that could potentially impact market entry, particularly in formulation and process areas. Notable active patents in the US include:
| Patent Number | Simple Legal Status | Application Date | Estimated Expiry | Patent Type | Applicant |
|---|---|---|---|---|---|
| US20210228598A1 | Active | 2019-08-13 | 2040-07-16 | Formulation | AMEZCUA AMEZCUA FEDERICO |
There are also active patents in other countries that could be relevant if considering global markets:
| Patent Number | Simple Legal Status | Application Date | Estimated Expiry | Patent Type | Applicant |
|---|---|---|---|---|---|
| EP3028706B1 | Active | 2014-07-24 | 2034-07-24 | Formulation | LAB RAAM DE SAHUAYO S A DE |
| EP2583674B1 | Active | 2011-06-01 | 2031-06-01 | Formulation | KOREA UNITED PHARM, Inc. |
| CN103086907A | Active | 2013-02-06 | 2033-02-06 | Process | Henan Dongtai Pharmaceutical Co Ltd. |
Clinical Results
Based on clinical trial analysis, aceclofenac has shown positive results in treating inflammatory pain:
- Study: EUROPEAN OBSERVATIONAL COHORT STUDY, EULAR2002
- Phase: Not Applicable
- Population: Inflammatory pain
- Results: Acute back pain = 50% (positive evaluation)
- Published Time: 2002-06-12
Infringement Cases
Based on the provided references, there is no information regarding any patent infringement incidents involving the drug aceclofenac.
Policy and Regulatory Risk Warning
After a comprehensive search, there are no specific market exclusivity or data protection periods identified for aceclofenac in the USA, as the drug is not currently approved in this market. However, any company seeking to enter the US market would need to submit a full New Drug Application (NDA) with comprehensive clinical data, as there is no existing approval to reference.
Market Entry Assessment & Recommendations
US Market Entry Strategy:
- Consider filing an NDA with the FDA, leveraging existing clinical data from international markets.
- Focus on differentiation from existing NSAIDs in the US market by highlighting aceclofenac's safety profile and efficacy data from global markets.
- Partner with an established US pharmaceutical company to navigate the regulatory landscape and leverage existing distribution networks.
Patent Strategy:
- The original compound patents have expired, removing the primary barrier to market entry.
- Conduct a detailed analysis of existing US formulation patents (such as US20210228598A1) to design around or consider licensing options.
- Develop novel formulations or delivery methods that could qualify for new patent protection in the US market.
Clinical Development:
- Design US-specific clinical trials focusing on indications with unmet needs or advantages over existing NSAIDs.
- Consider comparative effectiveness studies against commonly prescribed NSAIDs in the US market.
- Explore specialized indications or patient populations where aceclofenac might offer advantages over current therapies.
Market Positioning:
- Position as an alternative to traditional NSAIDs with potentially improved gastrointestinal safety profile based on international data.
- Target specific therapeutic areas where current NSAIDs show limitations in efficacy or tolerability.
- Consider specialized formulations (extended-release, topical, etc.) to differentiate from generic NSAIDs.
Regulatory Pathway:
- Engage with FDA through pre-IND meetings to determine the most efficient regulatory pathway.
- Explore the 505(b)(2) pathway, which might allow partial reliance on existing data while requiring less extensive clinical trials than a traditional NDA.
- Monitor ongoing regulatory reforms that might create accelerated pathways for drugs with established safety profiles in other regulated markets.
The US market represents a significant opportunity for aceclofenac given the large NSAID market and the expired status of core patents. However, entry requires careful navigation of existing secondary patents and substantial investment in clinical development to meet FDA requirements.
For more scientific and detailed information of aceclofenac , try PatSnap Eureka Pharma CI Explorer.