Inclisiran Market Analysis in the USA: Patents, Exclusivity, and Market Entry Outlook
Overview
The current US market shows approval for Inclisiran sodium, representing a significant advancement in PCSK9 inhibitor therapy using RNA interference technology. As a novel cholesterol-lowering medication, Inclisiran has been approved for treatment of atherosclerosis, heterozygous familial hypercholesterolemia, hypercholesterolemia, and primary hyperlipidemia. The drug was developed by Alnylam Pharmaceuticals and is currently marketed by Novartis Pharmaceuticals Corp. Clinical trials have demonstrated impressive efficacy with approximately 50-55% reduction in LDL-C compared to placebo, positioning it as a potentially transformative therapy in cardiovascular disease management.
Detailed Description
Drug Information
Inclisiran sodium was developed by Alnylam Pharmaceuticals, Inc. and received approval in the United States.
| Approval Number | Approval Company | Approval Date | Dosage Form | Route | Specification | Indication | Approval Status |
|---|---|---|---|---|---|---|---|
| 214012 | Novartis Pharmaceuticals Corp. | 2021-12-22 | Solution | Subcutaneous | EQ 284MG BASE/1.5ML (EQ 189MG BASE/ML) | Atherosclerosis, Heterozygous familial hypercholesterolemia, Hypercholesterolemia, Primary Hyperlipidemia | Approved |
Special Review
| Organization | Indication | Special Review | Country | Approval Date |
|---|---|---|---|---|
| Novartis Pharmaceuticals Corp. | Homozygous familial hypercholesterolemia | Orphan Drug | United States | 2018-01-22 |
Registration Patent Barrier Analysis
The FDA Orange Book lists multiple patents for Inclisiran, with varying expiration dates and legal statuses:
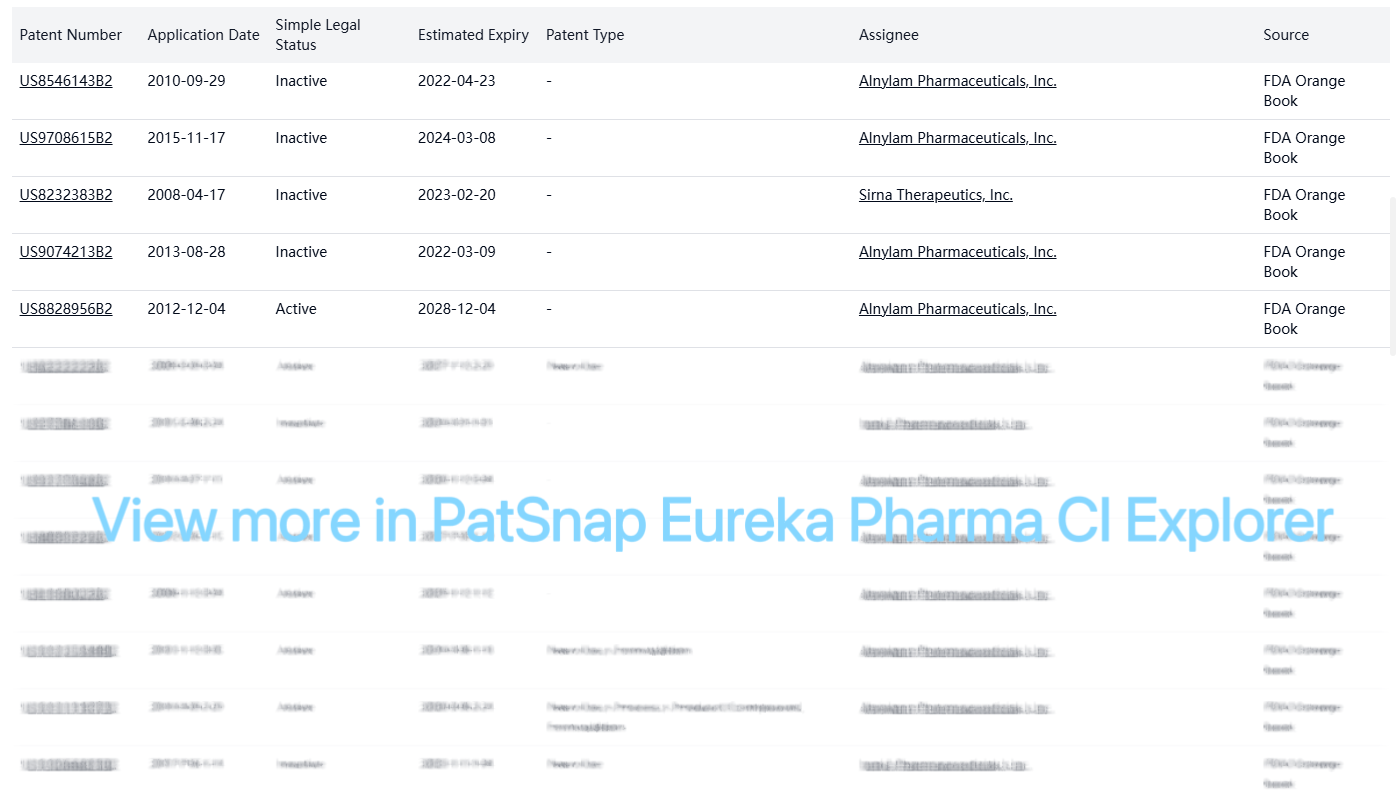
Other Patent Barrier Analysis
Original Company Patents
| Patent Number | Application Date | Simple Legal Status | Estimated Expiry | Patent Type | Assignee |
|---|---|---|---|---|---|
| US20130184324A1 | 2010-09-22 | Active | 2031-05-26 | Sequence | Alnylam Pharmaceuticals, Inc. |
| US20190292595A1 | 2019-02-06 | Inactive | - | Others | Alnylam Pharmaceuticals, Inc. |
| WO2009134487A2 | 2009-01-30 | PCT designated stage expired | - | New Use, Formulation | Alnylam Pharmaceuticals, Inc. |
| CN104651408A | 2010-06-15 | Inactive | - | Formulation | Alnylam Pharmaceuticals, Inc. |
Non-Original Company Patents
| Patent Number | Application Date | Simple Legal Status | Estimated Expiry | Patent Type | Assignee |
|---|---|---|---|---|---|
| WO2023170657A1 | 2023-03-10 | PCT designated stage expired | - | Process | Novartis AG |
| WO2024134505A1 | 2023-12-19 | PCT designated stage expired | - | Sequence, New Use, Process, Formulation, Others | Novartis AG |
| EP1407044B1 | 2001-11-29 | Inactive | 2021-11-29 | - | Max Planck Gesellschaft zur Förderung der Wissenschaften eV, The European Molecular Biology Laboratory |
| WO2018075658A1 | 2017-10-18 | PCT designated stage expired | - | New Use | The Medicines Co. |
Clinical Results
Phase 3 Clinical Trials (Study 1):
A multicenter, double‐blind, randomized, placebo‐controlled study enrolled 1,617 adults with clinical atherosclerotic cardiovascular disease (ASCVD) or at increased risk for ASCVD who were on maximally tolerated statin therapy. The trial assessed the mean percent change in LDL‑C levels from baseline to 18 months. In patients receiving inclisiran sodium, a significant reduction (~55%) in LDL‑C was observed compared to placebo (~3%).
Phase 3 Clinical Trials (Study 2 – ORION‑11):
This trial also adopted a multicenter, double‐blind, randomized, placebo‐controlled design. It enrolled 1,617 patients who required additional LDL‑C lowering alongside statin therapy. Patients received subcutaneous injections of 284 mg inclisiran on days 1, 90, 270, and 450, and the primary efficacy endpoint was the percent change in LDL‑C from baseline to day 510. The study demonstrated a mean percentage reduction in LDL‑C of approximately 50% in the inclisiran group compared to placebo.
Pharmacokinetic (PK) Studies:
Single‐dose and multiple‐dose studies showed that following a subcutaneous dose of 284 mg, peak plasma concentrations were reached around 4 hours post-dose, with a mean C_max of about 509 ng/mL. The drug exhibited linear and dose-proportional kinetics over a range of 25–800 mg, with an apparent volume of distribution of approximately 500 liters and a terminal half-life of about 9 hours, with no accumulation upon multiple dosing.
Population Pharmacodynamic Analysis:
Studies involving 4,328 patients evaluated variability in drug exposure and LDL‑C reductions across different demographic and clinical subgroups. Although higher plasma exposures were noted in patients with renal and hepatic impairments, the LDL‑C lowering effects remained similar.
Immunogenicity Assessment:
Evaluations in approximately 1,830 patients from placebo-controlled trials showed only a small percentage of patients developing binding anti‑inclisiran antibodies, which did not have a clinically significant impact on the drug's pharmacodynamics, safety, or efficacy over an 18‑month period.
Nonclinical Toxicology Studies:
Long-term carcinogenicity studies in rats and mice showed no evidence of carcinogenicity even at the highest doses tested. Genotoxicity tests confirmed that inclisiran was neither mutagenic nor clastogenic.
Fertility and Development Studies:
Studies in rats and rabbits demonstrated no evidence of embryo‑fetal toxicity or teratogenicity, and pre/postnatal development studies confirmed the absence of adverse effects on maternal performance, survival, growth, and reproductive performance.
Infringement Cases
Based on available information, there are no documented patent infringement incidents involving Inclisiran sodium, nor are there details related to litigation such as timeline, locations, parties involved, related products, or specific relevant patents.
Policy and Regulatory Risk Warning
After comprehensive research, it appears that Inclisiran may benefit from data exclusivity protection in the US as a new chemical entity (NCE), which typically provides 5 years of exclusivity from the approval date (December 22, 2021). This would extend protection until approximately December 2026, independent of patent protection. Additionally, as Inclisiran received orphan drug designation for homozygous familial hypercholesterolemia in January 2018, it may qualify for 7 years of market exclusivity for that specific indication.
Market Entry Assessment & Recommendations
Based on the patent landscape and regulatory status of Inclisiran in the USA:
Patent Protection Analysis: Inclisiran is protected by multiple patents with varying expiration dates, with the longest-lasting patent (US10851377B2) extending to 2036. Several key patents remain active, creating a significant barrier to generic entry.
Regulatory Exclusivity: As a new molecular entity, Inclisiran likely benefits from data exclusivity protection until approximately December 2026, and orphan drug exclusivity for homozygous familial hypercholesterolemia until approximately 2025.
Market Strategy for Innovator:
- Focus on building real-world evidence to support long-term cardiovascular outcomes beyond LDL-C reduction
- Develop patient support programs to improve adherence, leveraging the advantage of infrequent dosing (twice yearly after initial doses)
- Explore label expansion to additional indications and patient populations
- Consider combination therapies with complementary mechanisms of action
Generic Entry Considerations:
- Patent challenges would be complex due to the multiple layers of protection
- The earliest potential generic entry window appears to be after 2028 based on active patents
- The complex nature of the product (siRNA) may present manufacturing challenges for generic companies
- Consider biosimilar-like development approach given the complexity of the molecule
Market Access Recommendations:
- Develop value-based contracting models with payers based on LDL-C reduction outcomes
- Generate pharmacoeconomic data comparing twice-yearly Inclisiran administration to monthly PCSK9 antibody injections
- Target high-risk patients with demonstrated statin intolerance or inadequate response to maximize reimbursement potential
- Consider innovative pricing strategies to compete with other PCSK9 inhibitors while highlighting the convenience advantage
The robust patent portfolio, regulatory exclusivity periods, and technical complexity of manufacturing suggest that Inclisiran will maintain market exclusivity in the US for at least the next 5-7 years, with some protection extending potentially to 2036.
For more scientific and detailed information of Inclisiran , try PatSnap Eureka Pharma CI Explorer.