On March 18th, Cell published online a research paper about a new mechanism involving the gut in regulating cholesterol metabolism. The researchers discovered a gut-derived hormone called Cholesin, which is triggered by cholesterol intake and produced within intestinal cells. Cholesin achieves inhibition of the PKA-ERK1/2 signaling pathway in the liver by binding to the GPR146 receptor there, thereby downregulating the cholesterol synthesis process regulated by SREBP2. This means that when the gut absorbs cholesterol, the Cholesin-GPR146 axis acts to suppress cholesterol production in the liver, helping to maintain stable blood cholesterol levels and combat hypercholesterolemia and atherosclerosis.
DOI: 10.1016/j.cell.2024.02.024To investigate the interaction between intestinal cholesterol absorption and hepatic cholesterol synthesis, the researchers designed experiments to induce mice to consume diets with varying cholesterol content (regular diet and high-cholesterol Western diet) and observed changes in cholesterol levels in the intestine and liver. Under a high-cholesterol diet, intestinal cholesterol content increased, accompanied by downregulated expression of the cholesterol biosynthesis marker HMGCR in the intestine, suggesting a negative feedback mechanism where absorbed cholesterol can influence its own synthesis.
Against this backdrop, the researchers identified a new gut-derived hormone induced by cholesterol – Cholesin. Cholesin is triggered by cholesterol absorption mediated by NPC1L1, with its gene encoded in humans as C7orf50 and in mice as 3110082I17Rik. After secretion by intestinal cells, Cholesin binds to a specific orphan GPCR member, GPR146, and suppresses the PKA-ERK1/2 signaling pathway, reducing the hepatic cholesterol synthesis activity regulated by SREBP2.
To further confirm the relationship between Cholesin and its receptor GPR146, researchers employed a variety of experimental approaches, including but not limited to binding assays on frozen tissue sections, genome-wide CRISPR-Cas9 screenings, and immunohistochemistry techniques. Ultimately, it was established that GPR146 is an effective receptor for Cholesin, and the interaction between Cholesin and GPR146 directly influences the regulation of cholesterol metabolism. Through CRISPR screenings, protein expression purification, and a range of experimental validations, Cholesin's mechanism of action was elucidated, and it was demonstrated that both the sole use of Cholesin and its combination with statin drugs significantly reduce total plasma cholesterol levels and alleviate atherosclerotic lesions. Moreover, Cholesin can also decrease body weight gain, lower triglyceride levels, and improve hepatic lipid accumulation and inflammation markers.
About AtherosclerosisAtherosclerosis is a chronic inflammatory disease, primarily characterized by the accumulation of substantial amounts of cholesterol and other lipid substances on the vessel walls, forming plaques. These plaques gradually enlarge and can lead to the narrowing of the arterial lumen, impeding blood flow. Additionally, these plaques may become unstable and rupture, causing thrombosis, which in turn can trigger serious events such as myocardial infarction or stroke.
Cholesterol is an essential lipid for the human body, involved in the construction of cell membranes, hormone synthesis, and various other physiological functions. However, when cholesterol levels in the blood are too high, especially when low-density lipoprotein cholesterol (LDL-C) concentrations are excessive, the risk for cardiovascular diseases increases, with the most notable being the onset and progression of atherosclerosis. Studies have shown that an excess of cholesterol, particularly oxidized LDL cholesterol, penetrating beneath endothelial cells, can trigger an inflammatory response through a complex series of biochemical processes. This stimulates smooth muscle cell migration and proliferation, causes macrophages to engulf lipids and become foam cells, forming what are called fatty streaks, which then evolve into mature atherosclerotic plaques.
In addition to the abnormal accumulation of lipids, the state of imbalance between immune responses and their clearance is also believed to be primarily shaped by the migration and homeostasis of leukocytes, which are regulated by chemokines and their receptors. In recent years, new pro-inflammatory and anti-inflammatory pathways that link lipid biology with inflammation have been discovered, and gene expression profiling studies have revealed various factors involved in human coronary artery disease.
Cardiovascular diseases are the leading cause of death globally, with an estimated 17.8 million people dying each year. Inflammation is considered to be one of the main driving forces in the development of atherosclerosis and is recognized as one of the major etiologies of cardiovascular disease.
Despite the increasing body of research evidence over the past three decades showing that immune components play an indispensable role in the formation and chronicity of atherosclerosis, through a series of immunotherapeutic targets, the benefits and challenges brought about by targeting inflammation and the immune system in cardiovascular diseases have been demonstrated.
DOI: 10.1038/s41569-021-00668-4DOI: 10.1038/nm.2538Using the diagram above as an example, chemokines and their receptors also play an important role in the pathogenesis of atherosclerosis. The process of leukocyte recruitment includes rolling, firm adhesion, lateral migration, and transendothelial migration, which are finely regulated by chemokines. On the one hand, soluble chemokines directly mediate the recruitment of leukocytes; on the other hand, chemokines immobilized by glycosaminoglycans (GAGs) on the surface of activated endothelial cells can trigger the docking of leukocytes through GPCRs and activate leukocyte integrins, thus achieving firm adhesion. The cell-specific and lesion stage-specific utilization of chemokines and their receptors underscores the robustness and complexity of the chemokine system, ensuring the formation of specific chemokine combinations at each stage of the lesion that attract specific subtypes of leukocytes. Synergistic effects among chemokines in the recruitment of leukocytes can lead to cooperative interactions, potentially mediating the process between leukocytes and atherosclerotic lesions.
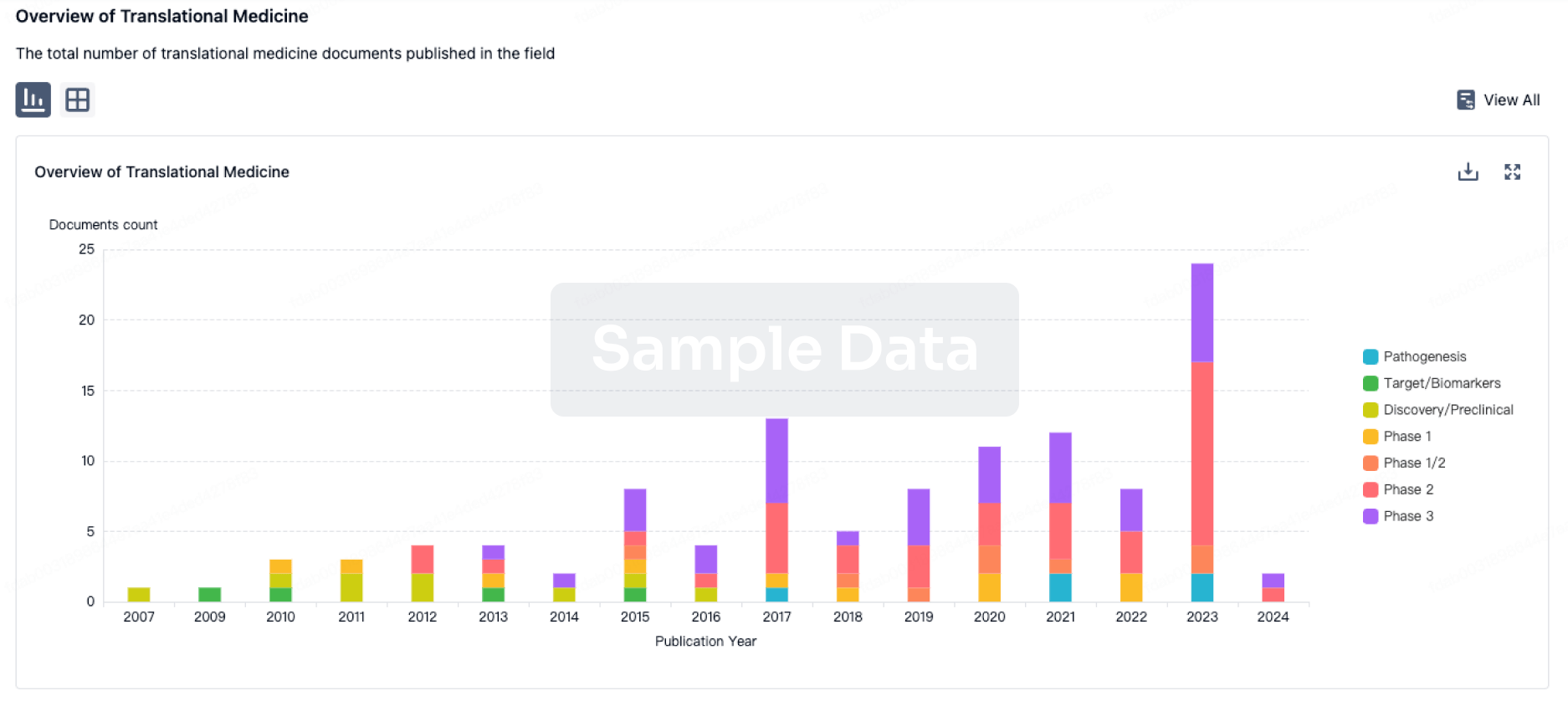
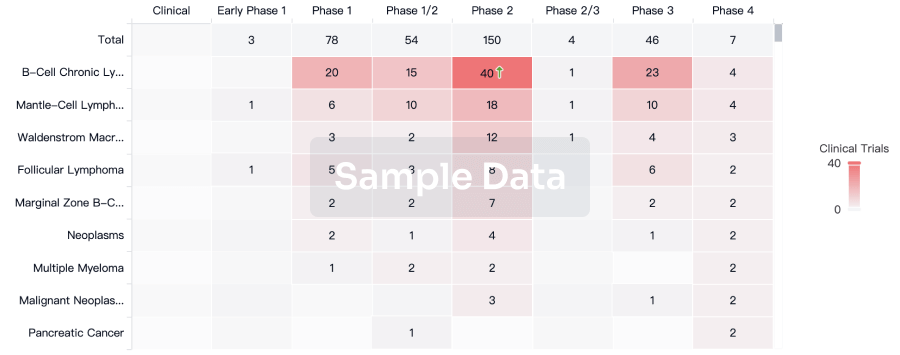
According to statistics from the Synapse database, as of now, there are more than 500 therapeutic drug pipelines globally, originating from over 400 institutions, encompassing over 200 targets, and involving 4095 clinical trials. The therapeutic drugs for this disease have a diverse range of Mechanisms of Action (MOA).
At present, the primary therapeutic drugs for the treatment of atherosclerosis include statins, which function primarily by inhibiting the key enzyme in cholesterol biosynthesis—hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase. Examples include Lovastatin (Merck, approved in 1987), Fluvastatin (Novartis, approved in 1993), Atorvastatin (Pfizer, approved in 1996), Rosuvastatin (AstraZeneca, approved in 2002).
P2Y12 receptor antagonists are another class of therapeutic drugs for atherosclerosis, acting by selectively blocking the function of the P2Y12 receptor to inhibit excessive activation and aggregation of platelets. This is crucial because platelets play a key role in the development of atherosclerosis and thrombosis, especially in the progression of cardiovascular diseases. Common drugs in this category include Clopidogrel (Sanofi, approved in 1997), Ticagrelor (AstraZeneca, approved in 2010), Cangrelor (CHIESI, approved in 2015).
Additionally, in recent years, PCSK9 has emerged as a therapeutic candidate for the treatment of hypercholesterolemia and atherosclerosis-related diseases. The primary function of PCSK9 is to regulate the number of low-density lipoprotein cholesterol (LDL-C) receptors on the membrane of liver cells. When PCSK9 binds to LDL receptors, it leads to their degradation within lysosomes, thereby reducing the uptake and clearance of circulating LDL-C by the liver, resulting in elevated levels of LDL-C in the blood. Therapeutic drugs related to this mechanism include Evolocumab (Amgen, approved in 2015), Alirocumab (Sanofi, approved in 2015), Inclisiran (Novartis, approved in 2020).
Factor Xa inhibitors are a class of drugs that specifically inhibit the activity of coagulation factor Xa (FXa). FXa plays a crucial role in the blood coagulation process, as it acts as a key enzyme in the coagulation cascade, facilitating the conversion of prothrombin to thrombin, which in turn catalyzes the transformation of fibrinogen into fibrin, leading to the formation of a stable clot. By binding to FXa and preventing its activity, Factor Xa inhibitors can effectively suppress the formation of thrombi and are widely used in clinical practice to prevent and treat various thrombotic diseases, such as deep vein thrombosis, pulmonary embolism, and stroke prevention in patients with atrial fibrillation. Therapeutic candidates in this category include Rivaroxaban (Bayer, approved in 2008), Edoxaban Tosylate (Daiichi Sankyo, approved in 2012), Apixaban (Bristol-Myers Squibb, approved in 2011).
Additionally, SGLT2 inhibitors were initially developed for the treatment of type 2 diabetes by inhibiting the SGLT2 transporter protein in the renal proximal convoluted tubules, reducing the reabsorption of glucose and thereby promoting the excretion of glucose in the urine, leading to decreased blood glucose levels. In recent years, multiple large-scale clinical trials have revealed the significant therapeutic effect of SGLT2 inhibitors in reducing the risk of cardiovascular events, especially in patients with type 2 diabetes who have atherosclerotic cardiovascular diseases. Therapeutic candidates related to this include Tofogliflozin (Kowa, approved in 2014) and Ertugliflozin (Pfizer, approved in 2017).
Besides the classical P2Y12 receptor, potential therapeutic targets within the GPCR family include AT1R, PAR-1R, CXCR4, CCR2, NIACR1, TBXA2R, PGI2R, ADRA1, and EP3. However, the progress in the drug pipeline related to these targets is not promising. Only Vorapaxar, a tricyclic compound derived from camptothecin that acts as a selective inhibitor on thrombin-activated receptor (PAR-1), has been approved for reducing the incidence of thrombotic cardiovascular events in patients with a history of myocardial infarction (MI) or peripheral artery disease (PAD).
The first antibody drug candidate developed targeting an immunomodulatory factor associated with GPCRs for treating atherosclerosis is the humanized monoclonal antibody Plozalizumab, an antagonistic monoclonal antibody targeting CCR2 developed by Takeda Pharmaceutical in Japan. CCR2 plays a critical role in the migration of monocytes and macrophages to sites of inflammation. The recruitment of macrophages to the arterial wall is considered a key step in the development of atherosclerosis.
The first nanobody drug candidate is BI-655088, targeting the CX3CR1 receptor.