TOKYO, Oct. 1, 2024 /PRNewswire/ -- PeptiGrowth Inc. (Headquarters: Chiyoda-ku, Tokyo; President: Jiro Sugimoto) has successfully developed a novel synthetic peptide called "FGF2 alternative peptide (FGFR1c agonist)", which has equivalent function to recombinant FGF2 (Fibroblast Growth Factor 2, basic FGF (bFGF)). This product will be on the market at the beginning of October 2024.
Development of Synthetic Peptide Growth Factors by PeptiGrowth
Continue Reading
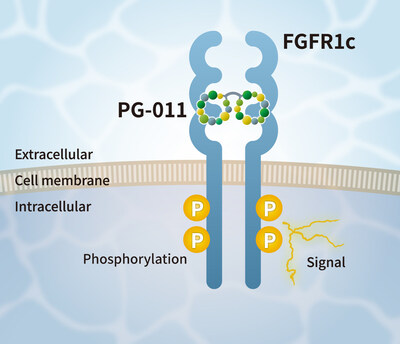
Conceptual Diagram of the Mechanism of Action of PG-011
Proliferation of human MSCs by PG-011 and observations of cell morphology of iPSCs after 3 passages of culturing in the presence of PG-011 or FGFs, as well as the measurement of undifferentiated markers on Day 7 at P3.
Traditional growth factors and cytokines used in regenerative medicine and cell therapy manufacturing face several quality issues, including lot-to-lot variability, potential contamination with biological impurities, low stability, and high costs. PeptiGrowth has focused on developing a series of synthetic peptides that address these challenges while maintaining functionality equivalent to conventional growth factors and cytokines. Their peptides are completely chemically synthesized and free from animal components, enabling the formulation of xeno-free and chemically defined cell culture media.
About FGF2 alternative peptide (FGFR1c agonist) [Product code: PG-011]
The FGF2 Alternative Peptide (
PG-011) is a dimeric peptide composed of cyclic peptides that bind to human FGFR1c. PG-011 exhibits agonist activity toward FGFR1c by binding to it, thereby activating signaling pathways in various cell types.
PeptiGrowth has confirmed that PG-011 promotes the proliferation of human mesenchymal stem cells (MSCs) at approximately one-third the concentration of FGF2 when compared on a ng/mL basis. Additionally, in collaboration with the CiRA Foundation, they evaluated PG-011's ability to maintain the undifferentiated state of human iPSCs. Three iPSC lines were cultured for 7 days in media containing either 100 ng/mL (~5.9 nM) of FGF2 or the same molar concentration (equivalent to ~30 ng/mL) of PG-011, followed by three passages. Afterward, cell morphology observations and undifferentiated marker measurements were conducted. Following three passages, iPSCs cultured in both PG-011 and FGF2 exhibited nearly identical morphology. Furthermore, analysis of undifferentiated markers (SSEA4 and TRA-1-60) indicated that iPSCs cultured with PG-011 displayed a similar positivity rate to those cultured with FGF2. Cell proliferation rates during the culture period were also equivalent under both conditions. These results suggest that PG-011 has the ability to maintain iPSCs in an undifferentiated state, comparable to FGF2.
Recombinant FGF2 is known for its low stability, as it degrades and loses activity easily under culture conditions. This necessitates higher concentrations and frequent medium changes, significantly increasing production costs and workload. PeptiGrowth has confirmed that PG-011 maintained its full activity even after 4 days of incubation at 37°C, whereas FGF2's activity significantly declined under the same conditions, demonstrating the superior stability of PG-011.
Moreover, they assessed the reactivity of PG-011 with bovine cells, as FGF2 is a key growth factor in cultivated meat production. PG-011 efficiently promoted the proliferation of bovine muscle satellite cells, suggesting that PG-011 may demonstrate cross-reactivity across various cell types, including those of non-human origin, making it a promising candidate for use in cultivated meat production.
PG-011 provides cell proliferation and undifferentiated state maintenance comparable to FGF2 but with superior stability, reducing both usage costs and workload. It also shows excellent proliferation activity in both human and bovine cells, making it a compelling choice for regenerative medicine and cultivated meat production.
Product Overview
Product name: FGF2 alternative peptide (FGFR1c agonist)
Product code: PG-011
Product form: Lyophilized
Storage conditions: -20°C or less
Purity: ≥95% by HPLC
Molecular weight: 5127.81(Acetate)
Size: 10 µg, 50 μg (volume per glass vial)*This product can be provided with an Animal Component Free (ACF) certificate.
*This product is for testing and research use only (RUO).
*There is a possibility that the specifications can change.
Purchase Our Product
If you would like to purchase our products, please contact us using the information below.
For customers who would like to purchase GMP compliant products as well as the above PG products in bulk scale, please contact us using the information below.
Development of other peptides from PeptiGrowth
We have developed alternative peptides targeting numerous growth factors and cytokines. To date, we have launched 11 products and plan to launch several additional growth factor alternative peptides targeting PDGF-AA, KGF, and IL-15 between late 2024 and mid- 2025. Details will be updated on our website () . If you are interested, please contact us using the following contact information.
Contacts for inquiries or additional information:
PeptiGrowth Inc. E-mail: [email protected] TEL:+81-70-4503-1497
Photo -
Photo -
Logo -
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED