Conformal Medical's CLAAS® System Demonstrates Low Thrombogenicity Compared to Commercially Available LAAO Devices
13 Mar 2024
Clinical StudyAHA
Novel left atrial appendage occlusion (LAAO) design aims to reduce the risk of stroke without the need for anticoagulants in patients with atrial fibrillation (Afib)
NASHUA, N.H., March 13, 2024 /PRNewswire/ -- Conformal Medical, Inc. announced that the CLAAS® System was featured in a podium presentation at the Cardiovascular Research Technologies (CRT) 2024 conference. Dr. William Gray, Professor of Medicine, Thomas Jefferson University, System Chief, Cardiovascular Diseases at Main Line Health, and Co-Director Lankenau Heart Institute presented "Comparative Acute Thrombogenicity of The CLAAS Foam Implant and Watchman FLX In An In Vitro Blood Loop Model" during the conference's Best Abstracts, March 10th.
Continue Reading

Preview
Source: PRNewswire
Conformal Medical's CLAAS® Implant
Preview
Source: PRNewswire
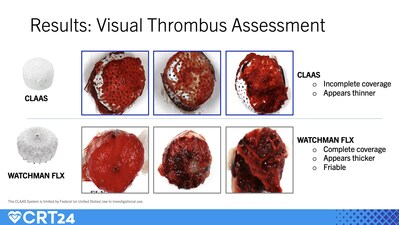
Visual Thrombus Assessment as presented by Dr. William Gray at the Cardiovascular Research Technologies (CRT) 2024 conference.
An in vitro study compared the relative thrombogenicity of the CLAAS implant to the Watchman FLX device. The CLAAS design features a foam cup with an embedded nitinol skeleton covered by a fluoropolymer (ePTFE) cover. The nitinol frame of the Watchman FLX is covered by polyethylene terephthalate (PET). Three of each device (n=3) were inserted into an acute radiolabeled in vitro blood loop system. After 90-120 minutes of exposure, the implants were visually assessed and radiolabels measured. When compared to the Watchman FLX, the CLASS device demonstrated:
Incomplete coverage with seemingly thinner thrombus
44% lower platelet deposition
"These results are very encouraging, indicating the CLAAS implant appeared less thrombotic than the Watchman FLX in this in vitro blood loop model," commented Dr. Gray. "Device Related Thrombus (DRT) remains a concern in left atrial appendage closure and advances in device design may help mitigate this in the clinical setting. The ePTFE fluoropolymer component in the CLAAS device appears to be less thrombogenic which may reduce DRT; still, more studies are required to further validate these initial results."
Additionally, the study was recently published online in JACC: Cardiovascular Interventions.
Conformal Medical is actively enrolling patients in the CONFORM pivotal trial, evaluating the safety and efficacy of the CLAAS System compared to other commercially available LAAO devices. The prospective, multicenter, randomized controlled study will enroll approximately 1,600 patients.
About Conformal Medical
Conformal Medical, Inc. is a medical device company developing devices to prevent stroke in patients with non-valvular atrial fibrillation. The company's proprietary technology is intended to make left atrial appendage closure a same day, single operator procedure. For more information, visit https://conformalmedical.com/.
About Left Atrial Appendage Closure
More than six million people in the United States suffer from Afib, placing them at an increased risk of stroke.1 Current standard of care for stroke prevention is chronic oral anticoagulants, which are not well accepted by patients due to concern about associated risk of bleeding. Left Atrial Appendage Occlusion (LAAO) is emerging as an important alternative to blood thinners for preventing strokes in patients with non-valvular Afib.
Kornej J, Börschel CS, Benjamin EJ, Schnabel RB. Epidemiology of Atrial Fibrillation in the 21st Century: Novel Methods and New Insights. Circ Res. 2020 Jun 19;127(1):4-20.
CAUTION: Investigational Device. The CLAAS System is limited by Federal (or United States) law to investigational use.
SOURCE Conformal Medical, Inc.
For more details,please visit the original website
The content of the article does not represent any opinions of Synapse and its affiliated companies. If there is any copyright infringement or error, please contact us, and we will deal with it within 24 hours.
Organizations
Targets
-Drugs
Hot reports
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Leverages most recent intelligence information, enabling fullest potential.





