[Translation] Human bioequivalence of diclofenac sodium sustained-release tablets under fasting conditions
主要目标:为了证明受试制剂(T):安若维他药业泰州有限公司生产的双氯芬酸钠缓释片75mg,与参比制剂(R):Novartis Pharma S.A.S, France的Voltarene® L.P(双氯芬酸钠缓释片)75mg之间,在空腹条件下作用于48名健康、成年、人体受试者的生物等效性。
次要目标:监测受试者口服单剂量研究产品(IPs)的安全性和有效性。
[Translation] Main objective: To prove that the test preparation (T): Diclofenac sodium sustained-release tablets 75 mg produced by Anruvita Pharmaceutical Taizhou Co., Ltd. is consistent with the reference preparation (R): Voltarene® L.P (diclofenac sodium) from Novartis Pharma S.A.S, France Sustained-release tablets) 75 mg, bioequivalence in 48 healthy, adult, human subjects under fasting conditions.
Secondary objectives: To monitor the safety and efficacy of oral single doses of investigational products (IPs) in subjects.
[Translation] Human bioequivalence of diclofenac sodium extended-release tablets under postprandial conditions
主要目标:为了证明受试制剂(T):安若维他药业泰州有限公司生产的双氯芬酸钠缓释片75mg,与参比制剂(R):Novartis Pharma S.A.S, France的Voltarene® L.P(双氯芬酸钠缓释片)75mg之间,在餐后条件下作用于48名健康、成年、人体受试者的生物等效性。
次要目标:监测受试者口服单剂量研究产品(IPs)的安全性和有效性。
[Translation] Main objective: To prove that the test preparation (T): Diclofenac sodium sustained-release tablets 75 mg produced by Anruvita Pharmaceutical Taizhou Co., Ltd. is consistent with the reference preparation (R): Voltarene® L.P (diclofenac sodium) from Novartis Pharma S.A.S, France Extended-release tablets) 75 mg, bioequivalence in 48 healthy, adult, human subjects under postprandial conditions.
Secondary objectives: To monitor the safety and efficacy of oral single doses of investigational products (IPs) in subjects.
[Translation] Human Bioequivalence Test of Ezetimibe Tablets under Fasting Condition
主要目标:为了证明受试制剂(T):安若维他药业泰州有限公司生产的依折麦布片10mg,与参比制剂(R):默沙东(中国)投资有限公司的Ezetrol片10mg之间,在空腹条件下作用于48名健康、成年、男性和女性、人体受试者的生物等效性。
次要目标:监测受试者口服单剂量研究产品(IPs)的安全性和有效性。
应报告不良事件以及与实验室检查、体格检查和生命体征的临床显著差异,以评估安全性。
[Translation] Main objective: To prove the difference between the test preparation (T): Ezetimibe Tablets 10mg produced by Enrovita Pharmaceutical Taizhou Co., Ltd., and the reference preparation (R): Ezetrol Tablets 10mg from Merck (China) Investment Co., Ltd. Bioequivalence in 48 healthy, adult, male and female, human subjects under fasting conditions.
Secondary objectives: To monitor the safety and efficacy of single oral doses of investigational products (IPs) in subjects.
Adverse events and clinically significant differences from laboratory tests, physical examination, and vital signs should be reported to assess safety.
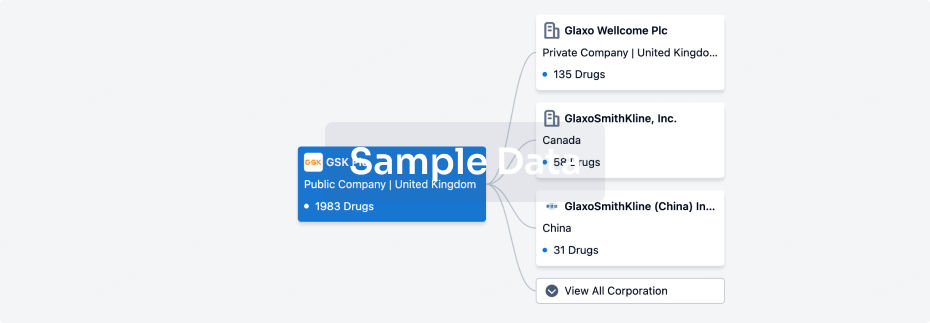
100 Clinical Results associated with Anruowita Pharmaceutical Taizhou Co., Ltd.
0 Patents (Medical) associated with Anruowita Pharmaceutical Taizhou Co., Ltd.
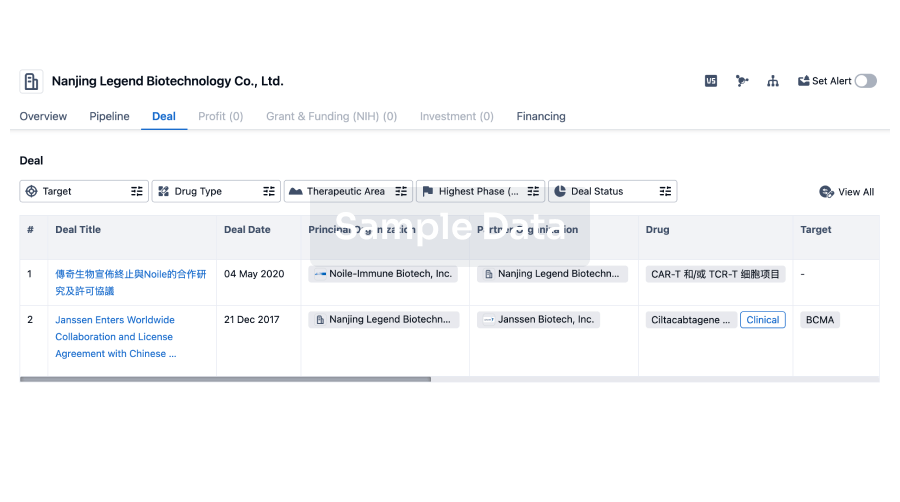
100 Deals associated with Anruowita Pharmaceutical Taizhou Co., Ltd.
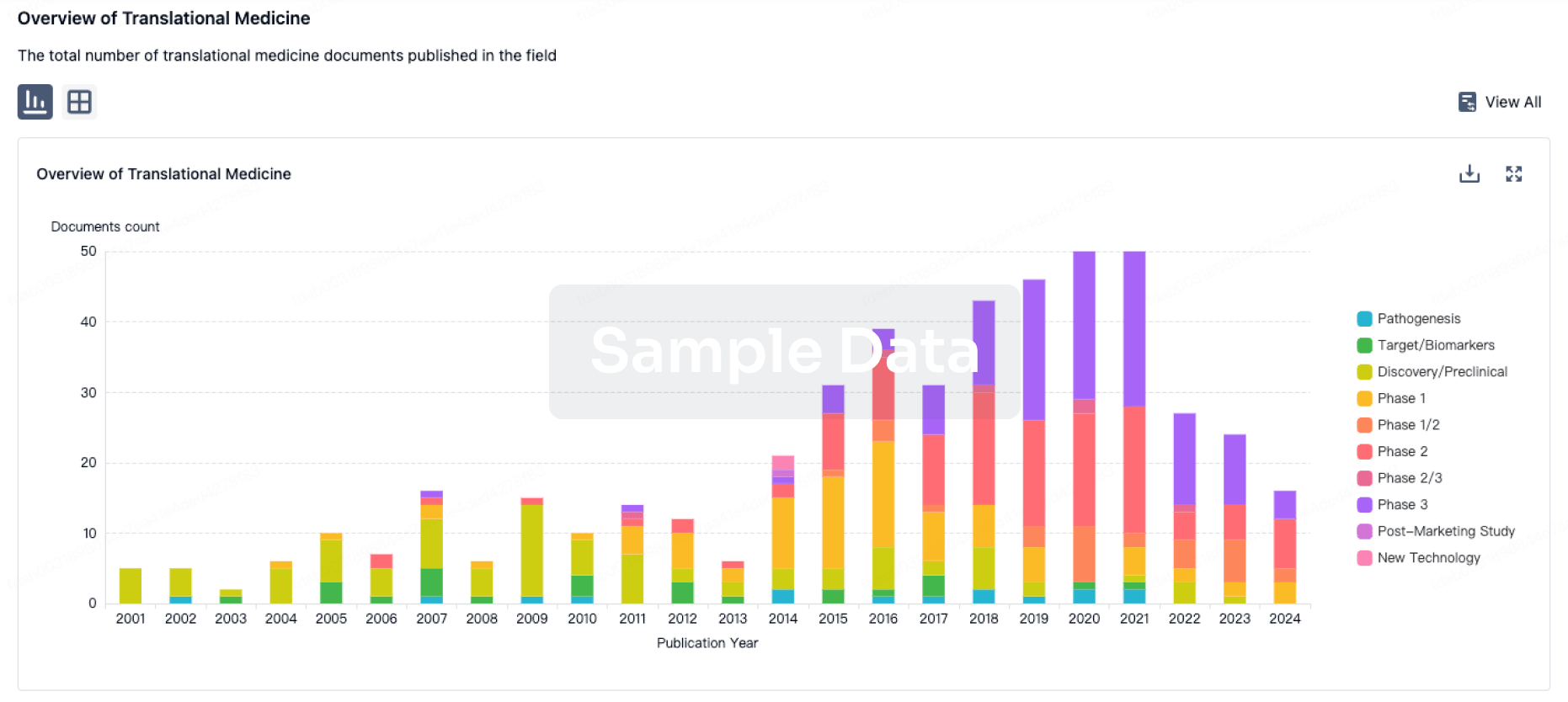
100 Translational Medicine associated with Anruowita Pharmaceutical Taizhou Co., Ltd.