A Prospective, Single Arm, Cohort Study of Nocturnal Temperature-controlled Laminar Airflow (TLA) Treatment (Airsonett®) in Patients With Allergic Asthma to Determine the Effect of Long-term Treatment on Exacerbation Frequency.
Temperature-controlled Laminar Airflow (TLA) treatment delivered by the Airsonett treatment device is a new treatment for patients with allergic asthma.
The LASER Trial (temperature-controlled Laminar Airflow in Severe asthma for Exacerbation Reduction - LASER) is a trial, currently evaluating whether the Airsonett device is able to reduce the frequency of asthma attacks in patients with allergic asthma. (www.lasertrial.co.uk)
Participants who successfully complete at least 6 months of follow-up in the LASER trial are eligible for the post trial provision of 4 years of an active TLA treatment device.
To date there is no long term performance data available to show whether there is any sustained benefit from the long term use of the Airsonett device, whether any improvement in asthma control or quality of life is maintained and whether patients will continue to use the treatment device beyond 12 months (the length of the longest previous trials of TLA treatment.)
The investigators plan to assess the performance of the Airsonett device by recording the frequency of asthma attacks in patients over a 4-year period (48 months.) The investigators also wish to evaluate the effect of long term TLA treatment on asthma control, quality of life, healthcare resource use and patient acceptability of longer term use of the Airsonett device.
Participants will be sent and asked to complete a series of questionnaires on a 6 monthly basis over the 48 month study period. Questionnaires will be returned by post to a single study centre based in Portsmouth, UK. Participants will also be contacted by telephone on a 6 monthly basis by a member of the trial team to collect information about their medication, healthcare resource use and whether they have had any asthma attacks since their last contact.
Participants will not be expected to visit the study centre during the trial.
A single-blind placebo-controlled trial to determine the effect of the airsonett airshower on sleep quality
Airsonett Airshower in Allergic Asthma a Double-blind Randomized Multi-centre Trial
The purpose of this study is to determine whether nocturnal environmental control with Temperature controlled Laminar Airflow (TLA) is effective as add on treatment in patients with perennial allergic asthma.
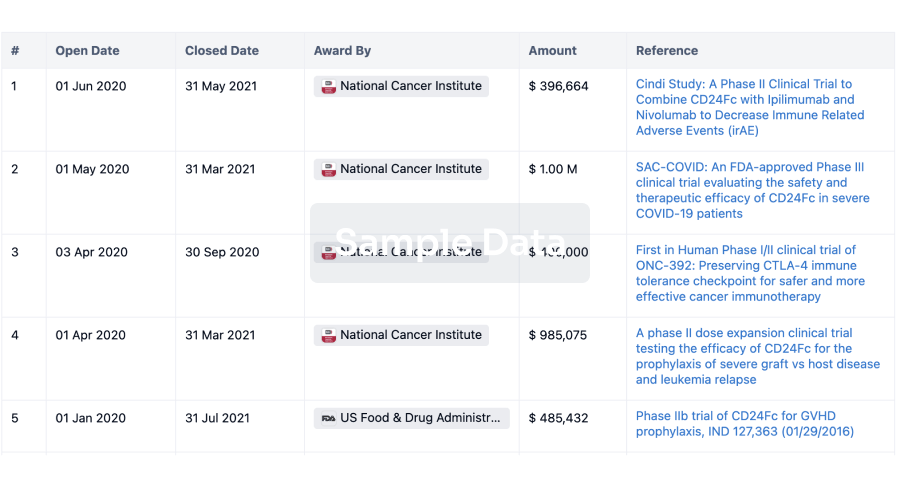
100 Clinical Results associated with Airsonett AB
0 Patents (Medical) associated with Airsonett AB
100 Deals associated with Airsonett AB
100 Translational Medicine associated with Airsonett AB