/ CompletedNot Applicable A Modified Draize Repeat Insult Patch Test in Healthy Volunteers, of Either Sex, to Investigate the Irritation and Allergenic Potential of One Test Article Following Repeated Cutaneous Patch Applications
The goal of this clinical trial is to assess whether a medical-grade Norway spruce resin-based topical product (Abilar) causes skin irritation or allergic sensitization in healthy adult volunteers. The main questions it aims to answer are:
Does the product cause skin irritation after repeated applications? Does the product induce sensitization (allergic contact dermatitis) after repeated exposure?
Participants will:
Have the test product applied to their skin via occlusive patches multiple times over three weeks.
Undergo dermatological assessments for signs of erythema (redness), swelling, or sensitization at scheduled time points.
Have a final challenge application on a new skin site after a rest period to check for delayed allergic reactions.
This study follows Good Clinical Practice (GCP) guidelines and the Declaration of Helsinki. It was conducted at Princeton Consumer Research (PCR Corp, UK) with approval from the East Anglia Ethics Committee, UK.
/ CompletedNot ApplicableIIT Comparison of Resin Salve and Conventional Octenidine in Patients With Neuropathic Diabetic Foot Ulcers - a Prospective, Randomized and Controlled Clinical Trial
Prevalence of diabetic foot ulcers are reported to be 15% in patients who suffer from diabetes and ulcerations are present in 84% of all diabetes-related amputations. Peripheral neuropathy leading to unperceived trauma seems to be the major cause of diabetic foot ulcers with 45-60% of ulcers to be considered merely neuropathic and 45% of mixed, neuropathic and ischemic etiology. Ulceration of lower limb is one of the most common complications related with diabetes and one of the major causes for hospitalization of diabetic patients. The most significant contributors to diabetic lower limb ulceration are neuropathy, deformity, uncontrolled elevated plantar pressure, poor glycemic status, peripheral vascular disease, male gender and duration of diabetes. Treatment of lower limb ulcers imposes an enormous burden on health care resources worldwide, and at least 33% of all expenses are spent to treat diabetic ulcers manifested as a complication of diabetes.
Although at least 170 topical wound care products are available, evidence of the superiority of one over another is tenuous, well-designed randomized, controlled trials are rare, and the number of case-control or observational studies is limited. In recent years, salve prepared from Norway spruce (Picea abies) resin has successfully been used in medical context to treat both acute and chronic wounds and ulcers of various origins. The objective of this prospective, randomized and controlled clinical trial is to investigate healing rate and healing time of neuropathic diabetic foot ulcer in patients, who are suffering from infected fore- or mid-foot ulceration (PEDIS-classification ≥ Grade II; 19) originated from Type I or II diabetes, and in patients whose diabetic ulcerations are candidates for topical treatment with resin (Study treatment) or octenidine (Control treatment). In addition, factors contributing with delayed healing of ulceration, antimicrobial properties, safety and cost-effectiveness of the resin salve treatment and control treatment will be analyzed.
Efficacy of Topical Resin Lacquer, Amorolfine, and Oral Terbinafine for Treating Toenail Onychomycosis: a Prospective, Randomized, Controlled, Investigator-blinded, Parallel-group Clinical Trial
The current study is conducted to corroborate the previous observational clinical trial with more valid methods and a more clinically relevant experimental design. This study aims to compare efficacy, safety, and cost between topically administered 30% resin lacquer for the treatment of dermatophyte toenail onychomycosis and the current "best practices": topical 5% amorolfine and systemic terbinafine.
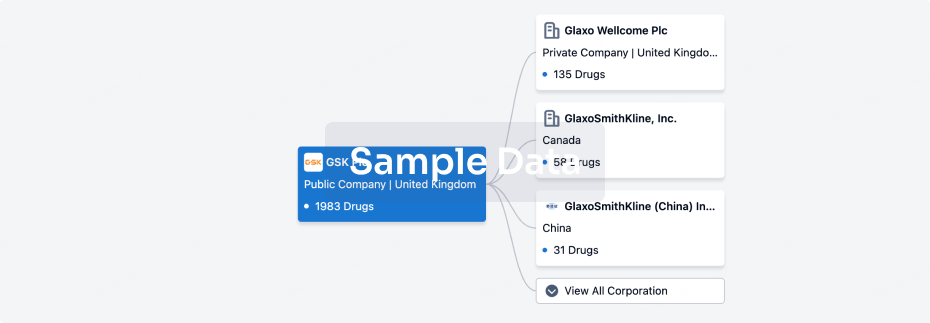
100 Clinical Results associated with Repolar Pharmaceuticals Oy
0 Patents (Medical) associated with Repolar Pharmaceuticals Oy
100 Deals associated with Repolar Pharmaceuticals Oy
100 Translational Medicine associated with Repolar Pharmaceuticals Oy