Assessing the Effectiveness of Continuous Glucose Monitoring Compared With Conventional Monitoring in Enhancing Surgical Outcomes for Diabetic Patients: A Multi-center, Pragmatic, Randomized Controlled Trial in China
The goal of this multi-center, pragmatic, randomized controlled trial is to assess the effectiveness of continuous glucose monitoring (CGM) compared with conventional monitoring in enhancing surgical outcomes for diabetic patients. The main questions it aims to answer are:
To assess the effectiveness of CGM compared with conventional monitoring in reducing the comprehensive complication index (CCI) for patients with diabetes or impaired glucose tolerance (IGT) within 30 days after surgery.
To analyze the impact of different age groups, types of surgery, preoperative levels of HbA1c, and preoperative 24h glycemic variations on the improvement of surgical outcomes under the CGM model.
Participants will receive CGM prescribed by the attending physician for at least 6 hours before surgery. Glucose monitoring should be continued until the 7th day after surgery or discharge.
The investigators will compare conventional monitoring group to see if CGM could reduce the CCIs for patients with diabetes or impaired glucose tolerance within 30 days after surgery.
Precision Vaginal Microbiome Transplantation in Women With Bacterial Vaginosis
The aim of this study was to explore the efficacy and safety of a new vaginal microbiome transplantation technique, using Lactobacillus cispatus from healthy donors as a treatment of bacterial vaginosis.
A Prospective Study to Evaluate the Diagnostic Accuracy of Computer-aided (CADx) System in Real-time Characterization of Colorectal Ulcerative Diseases
The goal of this observational study is to test the diagnostic accuracy of the newly developed CADx system in predicting the histopathology of colorectal ulcers when compared to expert endoscopists. The main question it aims to answer are to demonstrate whether the newly developed CADx system has a high-level diagnostic accuracy in predicting characterization of colorectal ulcerative diseases.
It is a multi-center study with two phases. The first retrospective phase is the development and validation of a CADx system by feature extraction from endoscopic photos and videos. The second prospective phase is the evaluation and comparison of the diagnostic accuracy between the CADx system, expert endoscopists and junior endoscopists.
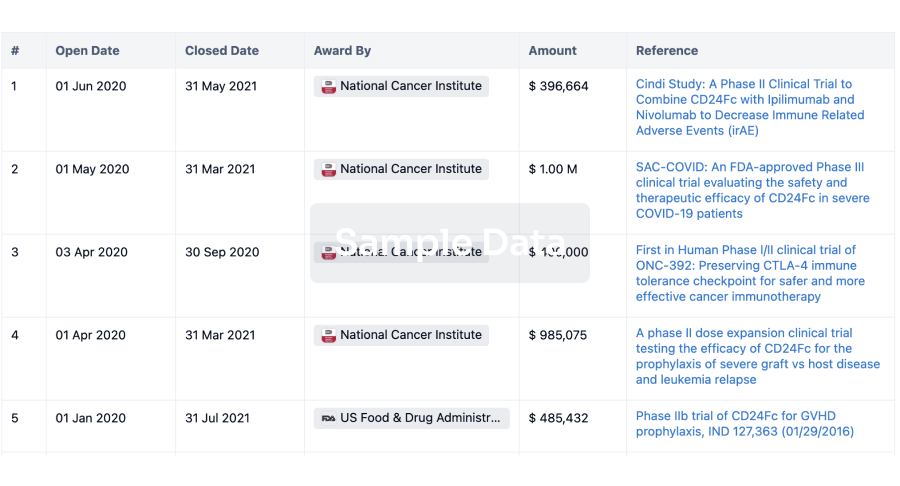
100 Clinical Results associated with
0 Patents (Medical) associated with
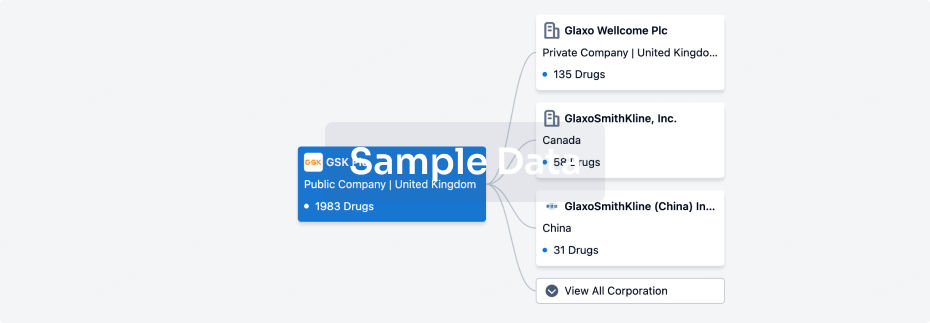
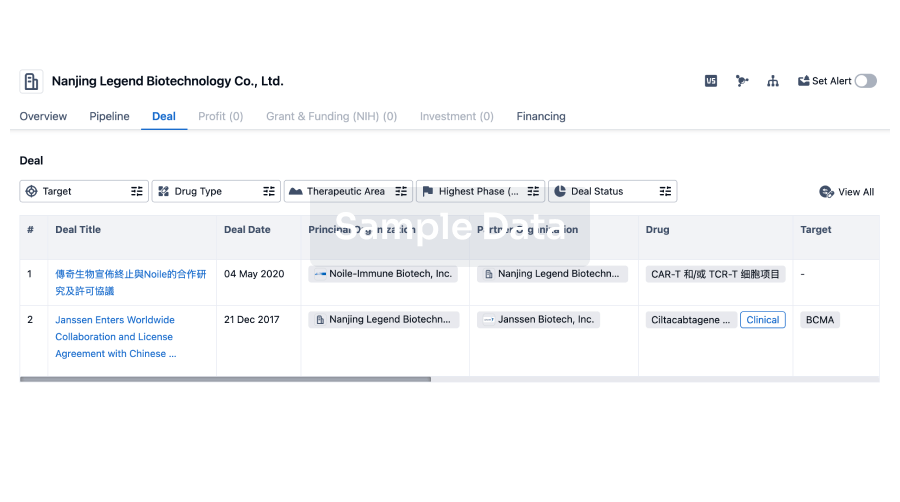
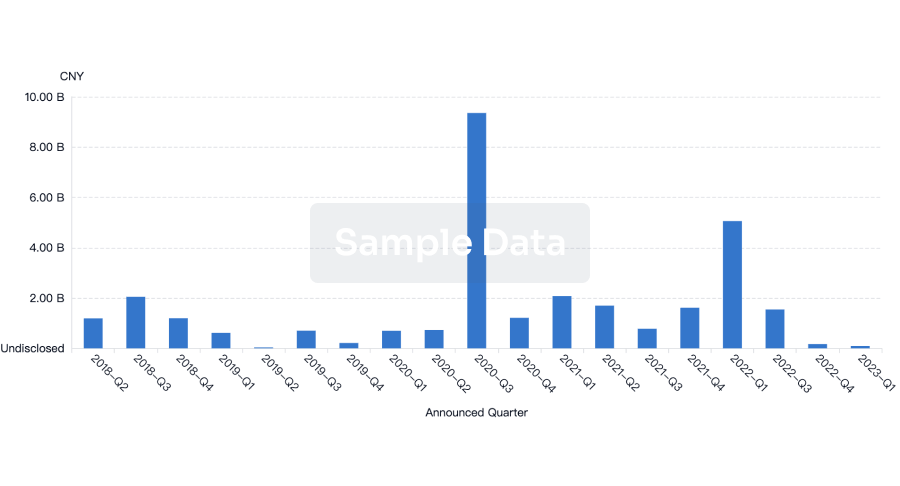
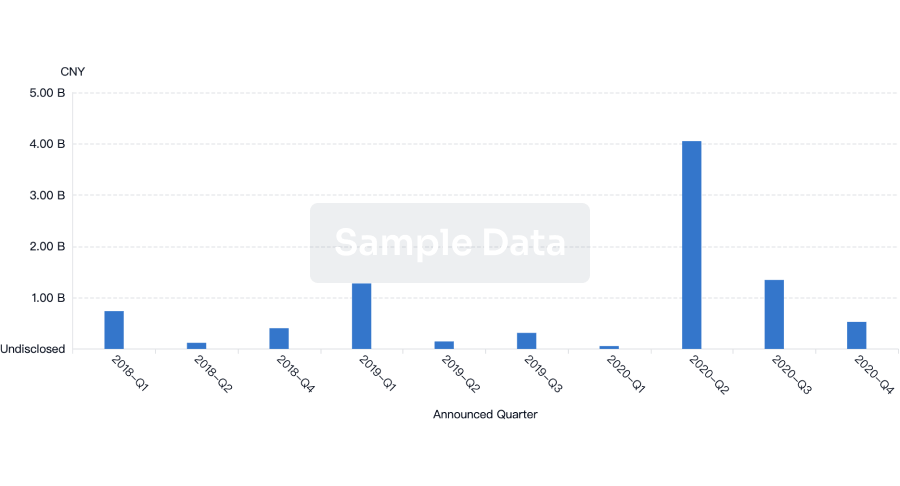
100 Deals associated with
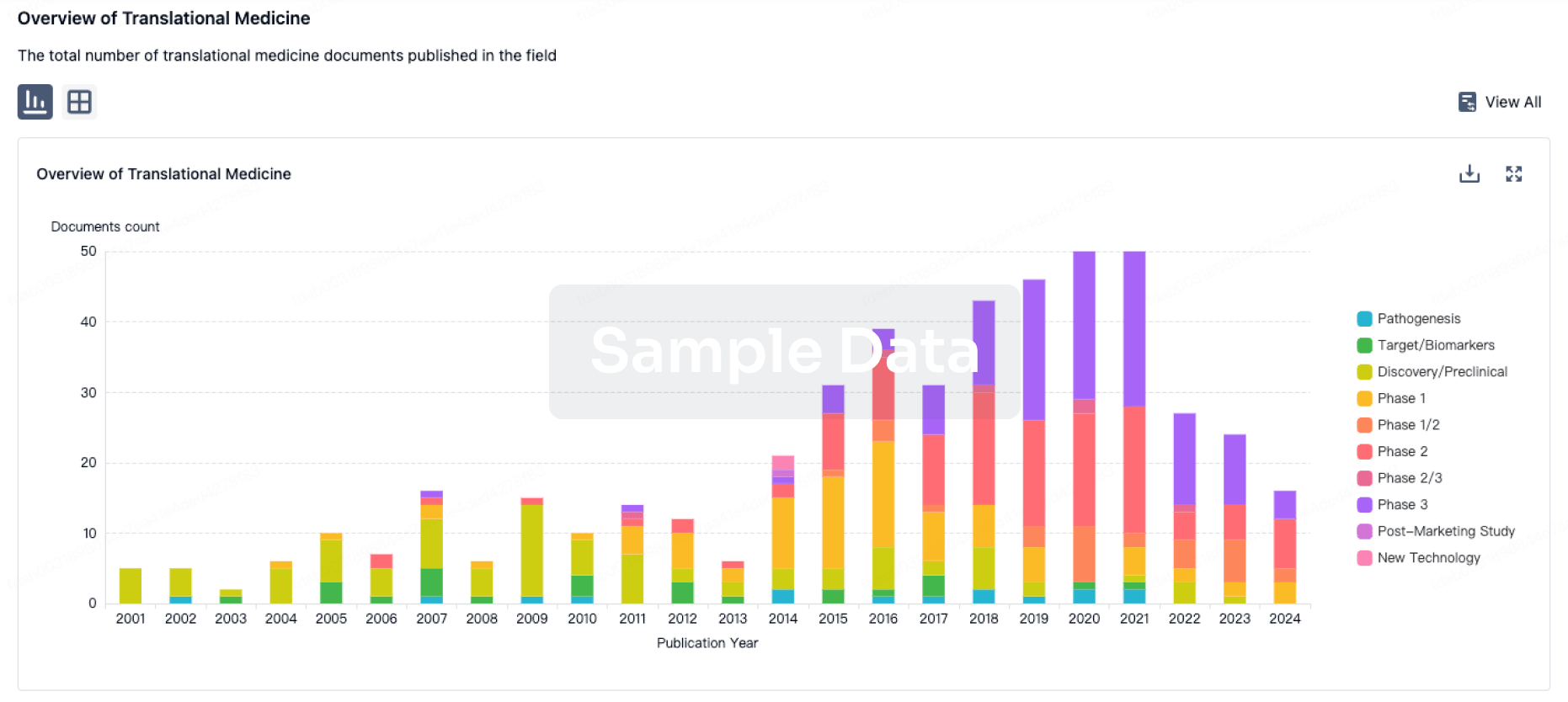
100 Translational Medicine associated with