铝镁匹林片(Ⅱ)在健康人体进行的单中心、随机、开放、单次给药、两制剂、自身交叉生物等效性试验
[Translation] A single-center, randomized, open-label, single-dose, two-preparation, self-crossover bioequivalence trial of aluminum-magnesium pirin tablets (Ⅱ) in healthy humans
本次试验以山东中健康桥制药有限公司研发的铝镁匹林片(II)(规格:每片含阿司匹林81mg,重质碳酸镁22mg,甘羟铝11mg)为受试制剂,按生物等效性研究的有关规定,以铝镁匹林片(II)(日本Lion Corporation生产,规格:每片含阿司匹林81mg,重质碳酸镁22mg,甘羟铝11mg,商品名:BUFFERIN)为参比制剂,考察两种制剂的相关药代动力学参数和相对生物利用度,评价两者在单次给药后(空腹和餐后)是否具有生物等效性。
[Translation] In this test, aluminum-magnesium pirin tablets (II) (specification: each tablet contains 81 mg of aspirin, 22 mg of heavy magnesium carbonate, and 11 mg of aluminum carboxylate) developed by Shandong Zhongjianjianqiao Pharmaceutical Co., Ltd. were used as the test preparation. According to the relevant regulations of sexual research, take aluminum magnesium pirin tablets (II) (produced by Lion Corporation of Japan, specifications: each tablet contains 81 mg of aspirin, 22 mg of heavy magnesium carbonate, 11 mg of aluminum carboxylate, trade name: BUFFERIN) as the reference preparation, The relevant pharmacokinetic parameters and relative bioavailability of the two preparations were investigated, and whether the two preparations were bioequivalent after a single dose (fasting and postprandial).
盐酸阿罗洛尔片在健康受试者中随机、开放、单剂量、两制剂、两序列、两周期交叉空腹状态下的生物等效性试验
[Translation] A randomized, open-label, single-dose, two-dose, two-sequence, two-cycle cross-over fasting bioequivalence trial of arololol hydrochloride tablets in healthy subjects
主要目的:健康受试者空腹状态下,口服单剂量盐酸阿罗洛尔片(受试制剂T,山东中健康桥制药有限公司生产,规格:10mg/片)与盐酸阿罗洛尔片(参比制剂R,Sumitomo Dainippon Pharma Co.,Ltd.生产,商品名:Almarl,规格:10mg/片)后,考察空腹条件下受试制剂与参比制剂的吸收速度和吸收程度,评价受试制剂与参比制剂是否具有生物等效性。
次要目的:观察受试制剂盐酸阿罗洛尔片和参比制剂盐酸阿罗洛尔片(Almarl®)在健康受试者中的安全性。
[Translation] Main purpose: Healthy subjects were given a single dose of arolol hydrochloride tablets (test preparation T, produced by Shandong Zhongjianjianqiao Pharmaceutical Co., Ltd., specification: 10 mg/tablet) and arolol hydrochloride tablets (refer to Comparative formulation R, produced by Sumitomo Dainippon Pharma Co., Ltd., trade name: Almarl, specification: 10 mg/tablet), the absorption rate and degree of absorption of the test preparation and the reference preparation under fasting conditions were investigated, and the test preparation and the reference preparation were evaluated. Whether the reference product is bioequivalent.
Secondary objective: To observe the safety of test preparation Arolol Hydrochloride Tablets and reference preparation Arololol Hydrochloride Tablets (Almarl®) in healthy subjects.
盐酸阿罗洛尔片在健康受试者中随机、开放、单剂量、两制剂、两序列、两周期交叉空腹/餐后状态下的生物等效性试验
[Translation] A randomized, open-label, single-dose, two-dose, two-sequence, two-cycle crossover fasting/postprandial bioequivalence trial of arolol hydrochloride tablets in healthy subjects
主要研究目的:健康受试者空腹/餐后口服单剂量盐酸阿罗洛尔片(受试制剂T,山东中健康桥制药有限公司生产,规格:10mg/片)与盐酸阿罗洛尔片(参比制剂R,Sumitomo Dainippon Pharma Co.,Ltd.生产,商品名:Almarl,规格:10mg/片)后,考察空腹和餐后条件下受试制剂与参比制剂的吸收速度和吸收程度,评价受试制剂与参比制剂是否具有生物等效性。次要研究目的:观察受试制剂盐酸阿罗洛尔片和参比制剂盐酸阿罗洛尔片(Almarl®)在健康受试者中的安全性。
[Translation] Main research purpose: Healthy subjects take a single dose of arolol hydrochloride tablets (test preparation T, produced by Shandong Zhongjianjianqiao Pharmaceutical Co., Ltd., specification: 10mg/tablet) and arololol hydrochloride tablets ( Reference preparation R, produced by Sumitomo Dainippon Pharma Co., Ltd., trade name: Almarl, specification: 10 mg/tablet), the absorption rate and degree of absorption of the test preparation and the reference preparation under fasting and postprandial conditions were investigated, and the evaluation Whether the test preparation is bioequivalent to the reference preparation. Secondary research objectives: To observe the safety of the test preparation arolol hydrochloride tablets and the reference preparation arolol hydrochloride tablets (Almarl®) in healthy subjects.
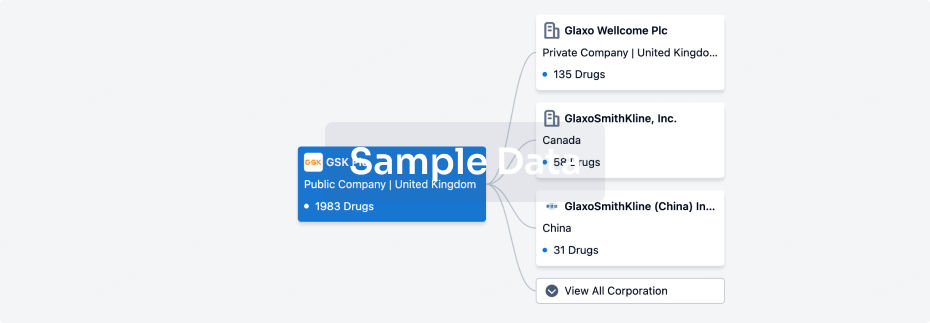
100 Clinical Results associated with Shandong Zhongjiankangqiao Pharmaceutical Co., Ltd.
0 Patents (Medical) associated with Shandong Zhongjiankangqiao Pharmaceutical Co., Ltd.
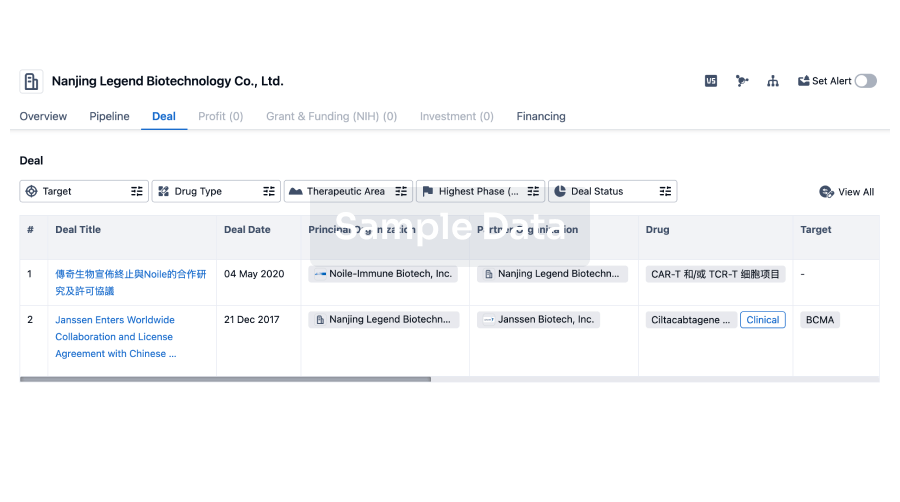
100 Deals associated with Shandong Zhongjiankangqiao Pharmaceutical Co., Ltd.
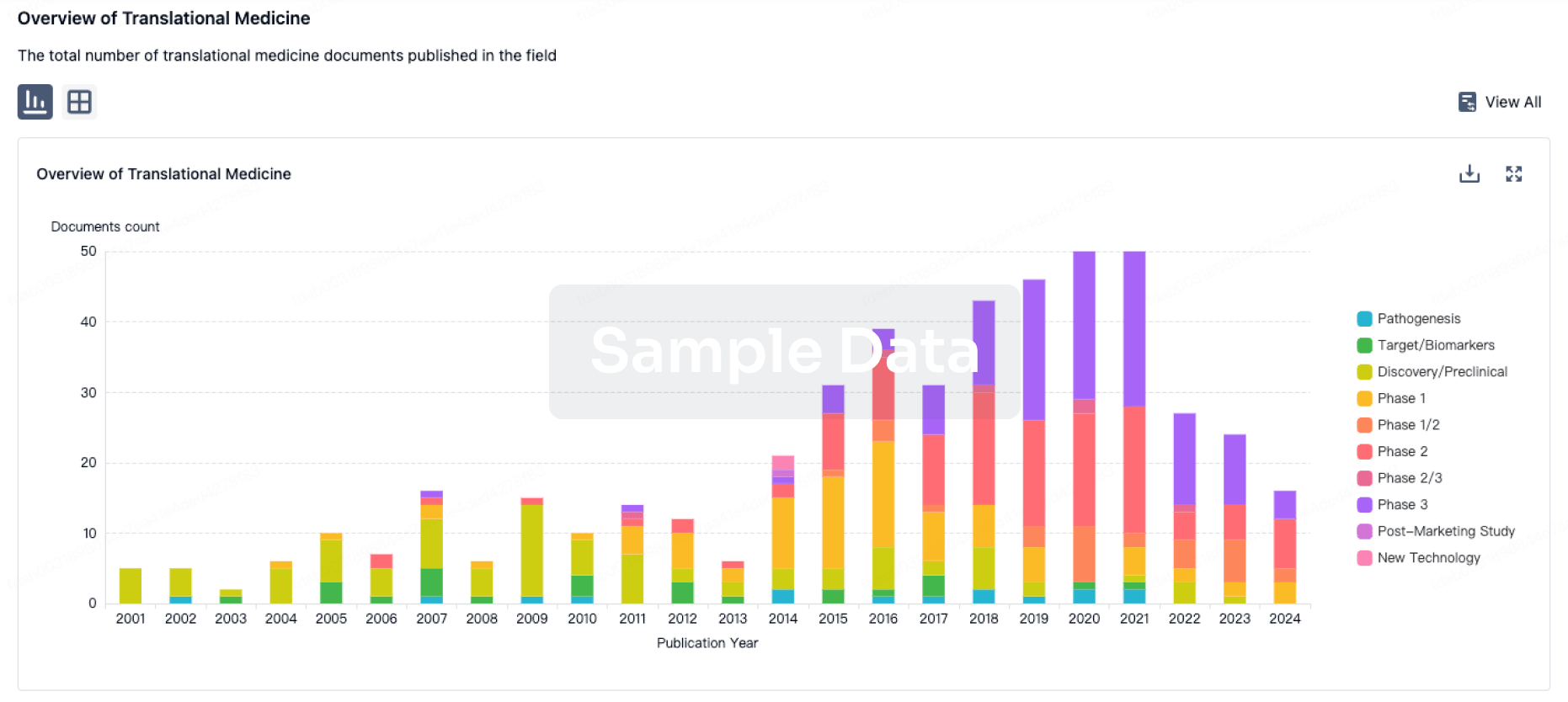
100 Translational Medicine associated with Shandong Zhongjiankangqiao Pharmaceutical Co., Ltd.