[Translation] Human Bioequivalence Test of Vildagliptin Tablets under Postprandial Conditions
比较餐后条件下,由湖南普道医药技术有限公司委托湖南九典制药股份有限公司生产的维格列汀片(规格:50 mg)与Novartis Europharm Ltd.持证的维格列汀片(规格:50 mg,商品名:Galvus®/佳维乐®)在中国健康人群中吸收程度和吸收速度的差异,并评价湖南九典制药股份有限公司生产的维格列汀片(规格:50 mg)的安全性。
[Translation] Under postprandial conditions, vildagliptin tablets (specification: 50 mg) commissioned by Hunan Pudao Pharmaceutical Technology Co., Ltd. and produced by Hunan Jiudian Pharmaceutical Co., Ltd. and vildagliptin tablets (specification: : 50 mg, trade name: Galvus®/ Jia Wei Le ®) in the difference of absorption degree and absorption speed in Chinese healthy population, and evaluate vildagliptin tablets (specification: 50 mg) produced by Hunan Jiudian Pharmaceutical Co., Ltd. security.
[Translation] Bioequivalence test of loxoprofen sodium gel patch
健康受试者在空腹状态下,以LEAD CHEMICAL CO.,LTD持证的洛索洛芬钠凝胶贴膏(商品名:Loxonin®PAP,规格:每贴10cm×14cm,含洛索洛芬钠100mg)为参比制剂,研究湖南九典制药股份有限公司生产的洛索洛芬钠凝胶贴膏(受试制剂T,规格:每贴10cm×14cm,含洛索洛芬钠100mg)的吸收速度与吸收程度,评价受试制剂与参比制剂是否具有生物等效性。
[Translation] Healthy subjects were fasted, with loxoprofen sodium gel patch (trade name: Loxonin®PAP, 10cm×14cm per patch, containing loxoprofen sodium) licensed by LEAD CHEMICAL CO., LTD. 100mg) as a reference preparation, to study the absorption of loxoprofen sodium gel plaster (test preparation T, specification: each paste 10cm×14cm, containing loxoprofen sodium 100mg) produced by Hunan Jiudian Pharmaceutical Co., Ltd. Speed and degree of absorption, to evaluate whether the test preparation and the reference preparation have bioequivalence.
洛索洛芬钠凝胶贴膏治疗膝骨关节炎疼痛的有效性和安全性的多中心、随机、单盲、阳性药/安慰剂平行对照临床试验
[Translation] A multicenter, randomized, single-blind, active drug/placebo parallel controlled clinical trial of the efficacy and safety of loxoprofen sodium gel patch in the treatment of knee osteoarthritis pain
主要目的:
以阳性药为对照,评价洛索洛芬钠凝胶贴膏治疗膝骨关节炎的临床疗效一致性。
以安慰剂为对照,评价洛索洛芬钠凝胶贴膏治疗膝骨关节炎的优效性。
次要目的:
评价洛索洛芬钠凝胶贴膏在膝骨关节炎患者中的安全性。
[Translation] main purpose:
Taking the positive drug as the control, the clinical efficacy of Loxoprofen sodium gel patch in the treatment of knee osteoarthritis was evaluated.
To evaluate the efficacy of loxoprofen sodium gel patch in the treatment of knee osteoarthritis with placebo as control.
Secondary purpose:
To evaluate the safety of loxoprofen sodium gel patch in patients with knee osteoarthritis.
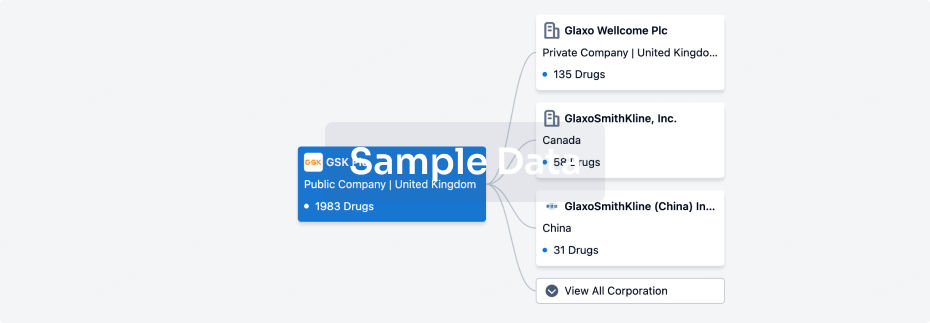
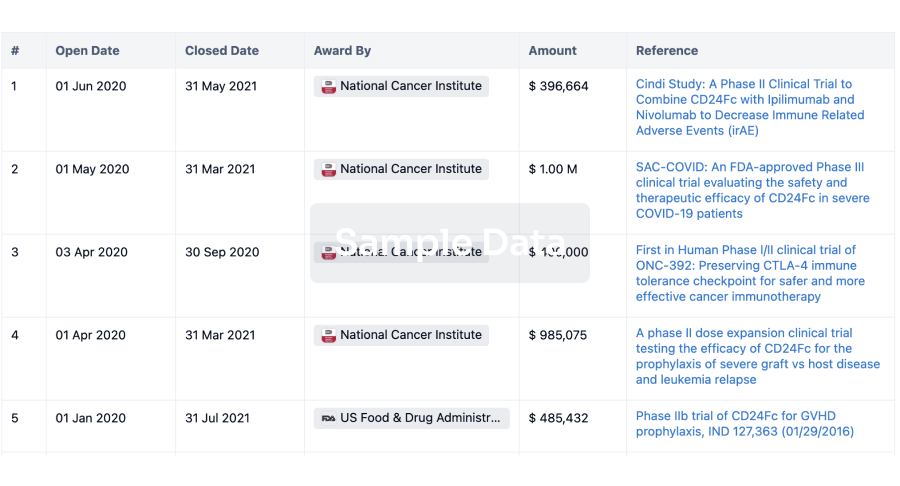
100 Clinical Results associated with Hunan Pudao Pharmaceutical Technology Co., Ltd.
0 Patents (Medical) associated with Hunan Pudao Pharmaceutical Technology Co., Ltd.
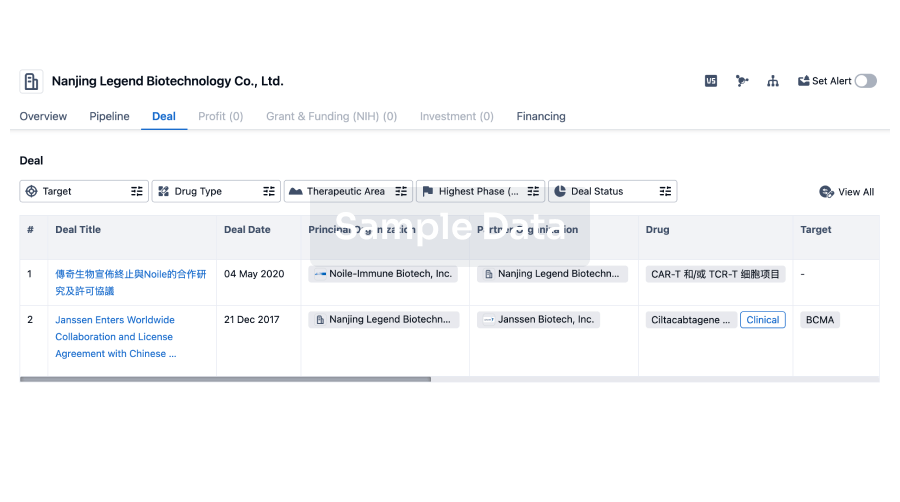
100 Deals associated with Hunan Pudao Pharmaceutical Technology Co., Ltd.
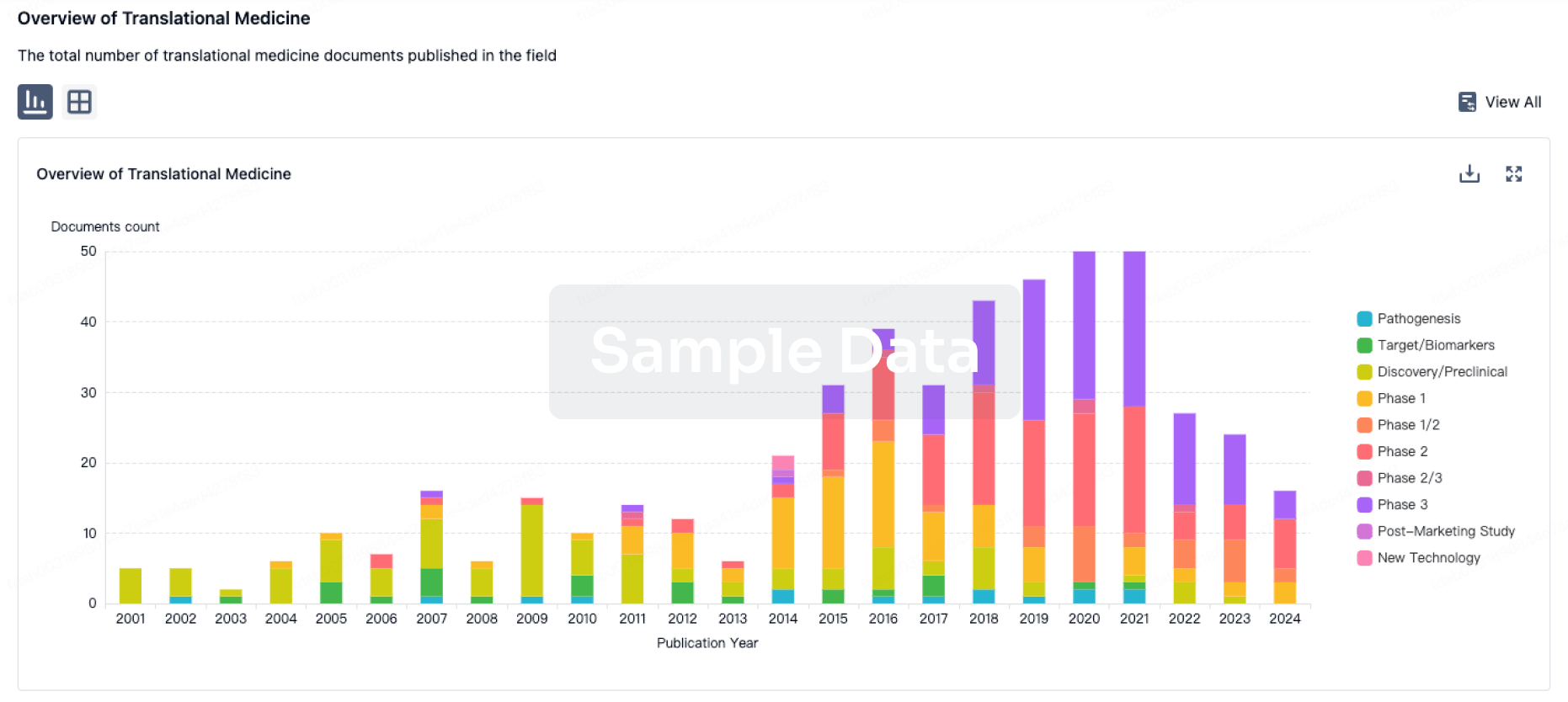
100 Translational Medicine associated with Hunan Pudao Pharmaceutical Technology Co., Ltd.