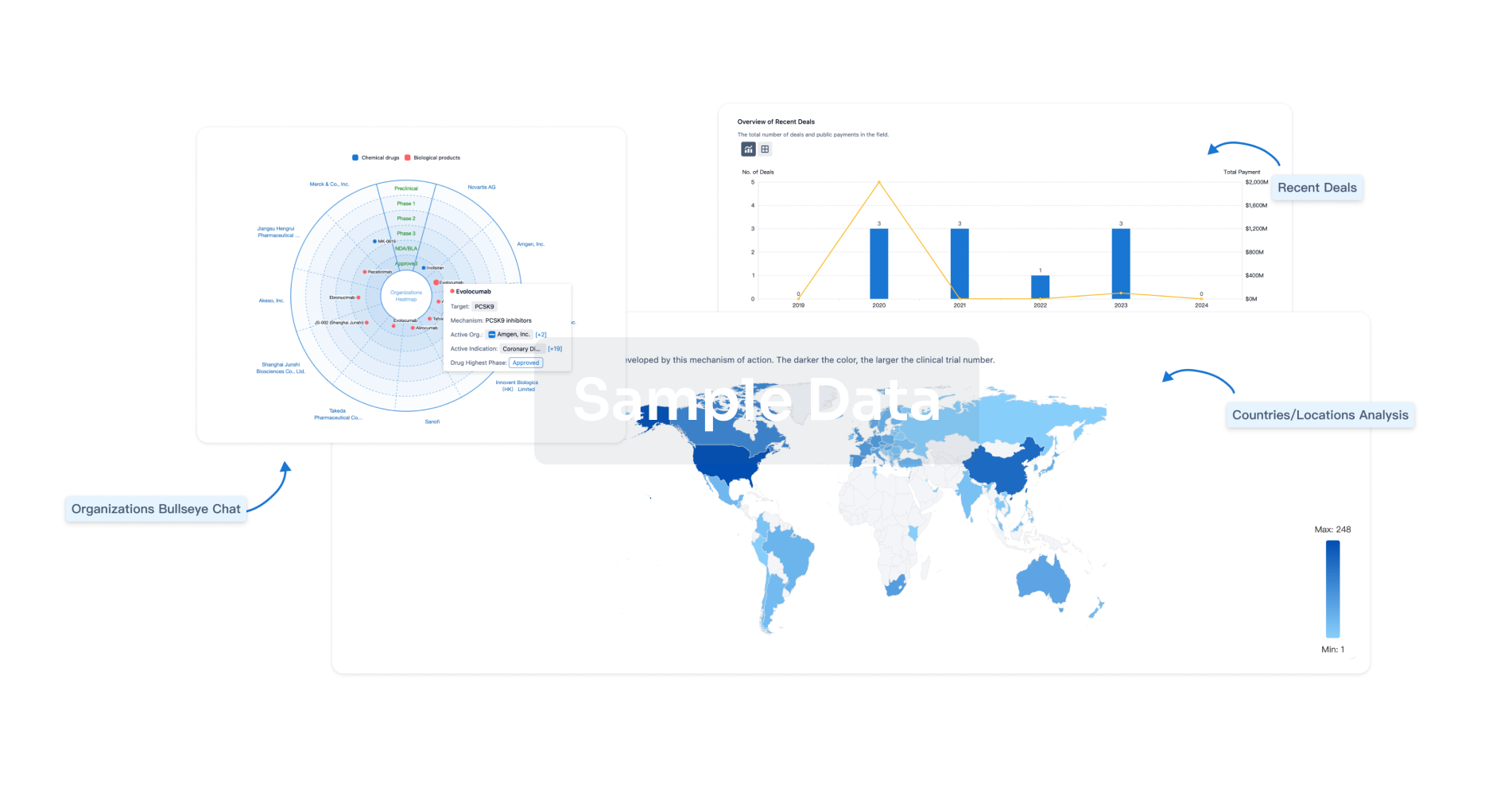
|
|
Active Org.- |
|
Active Indication- |
|
Drug Highest PhaseDiscontinued |
First Approval Ctry. / Loc.- |
First Approval Date20 Jan 1800 |
Phase I Biodistribution Study of 111-Indium-CMD-193 in Patients With Advanced Tumours Expressing the Lewis-Y Antigen
This was a Phase 1 dose-escalation study of CMD-193, a humanized monoclonal antibody linked to the toxin calicheamicin, in subjects with advanced tumors expressing the Lewis-Y antigen. The primary study objective was to determine the biodistribution and pharmacokinetics (PK) of 111-In-CMD-193 (i.e., CMD-193 tagged with a small amount of radioactive Indium [111-In]), with secondary objectives of determining changes in tumor metabolism and describing the antitumor responses to CMD-193.
Phase 1 Dose-Escalation Study of Intravenous CMD-193 in Subjects With Advanced Malignant Solid Tumors
The primary purpose of this study is to evaluate the safety, tolerability, and maximum tolerated dose (MTD) of CMD-193 administered intravenously (IV) to subjects with advanced malignant tumors. The secondary purpose is to obtain preliminary information on the pharmacokinetics and antitumor activity of IV CMD-193.
A Phase 1 Dose Escalation Study of CMD-193 in Subjects With Advanced Malignant Tumors.
The purpose of this study is to determine the safety, tolerability and maximum tolerated dose of CMD-193. Preliminary information about how a person's body processes CMD-193 and how CMD-193 affects tumors will also be collected.
100 Clinical Results associated with Lewis-Y antigen x DNA
100 Translational Medicine associated with Lewis-Y antigen x DNA
0 Patents (Medical) associated with Lewis-Y antigen x DNA