Request Demo
Last update 08 May 2025
GR x M-CSF
Last update 08 May 2025
Related
1
Drugs associated with GR x M-CSF100 Clinical Results associated with GR x M-CSF
Login to view more data
100 Translational Medicine associated with GR x M-CSF
Login to view more data
0 Patents (Medical) associated with GR x M-CSF
Login to view more data
11
Literatures (Medical) associated with GR x M-CSF06 Jun 2023·Endocrinology
Glucocorticoid Receptors Drive Breast Cancer Cell Migration and Metabolic Reprogramming via PDK4
Article
Author: Oakley, Robert H ; Cidlowski, John A ; Dwyer, Amy R ; Hagen, Kyla M ; Krutilina, Raisa I ; Liddle, Christopher ; Perez Kerkvliet, Carlos ; Parke, Deanna N ; Seagroves, Tiffany N ; Truong, Thu H ; Lange, Carol A
01 Oct 2019·Brain, Behavior, and ImmunityQ1 · MEDICINE
Glucocorticoid receptor antagonism prevents microglia-mediated neuronal remodeling and behavioral despair following chronic unpredictable stress
Q1 · MEDICINE
Article
Author: Horchar, Matthew J ; Wohleb, Eric S
19 Sep 2013·Journal of Leukocyte BiologyQ3 · MEDICINE
CSF-1 receptor-mediated differentiation of a new type of monocytic cell with B cell-stimulating activity: its selective dependence on IL-34
Q3 · MEDICINE
Article
Author: Sano, Hiroki ; Ohmori, Hitoshi ; Yamane, Fumihiro ; Watanabe, Koji ; Asakura, Miki ; Magari, Masaki ; Tanimoto, Hikaru ; Kanayama, Naoki ; Mabbott, Neil A ; Nishikawa, Yumiko ; Iwasaki, Eriko ; Stanley, E Richard ; Fujiwara, Yuki ; Matsui, Kazue
Analysis
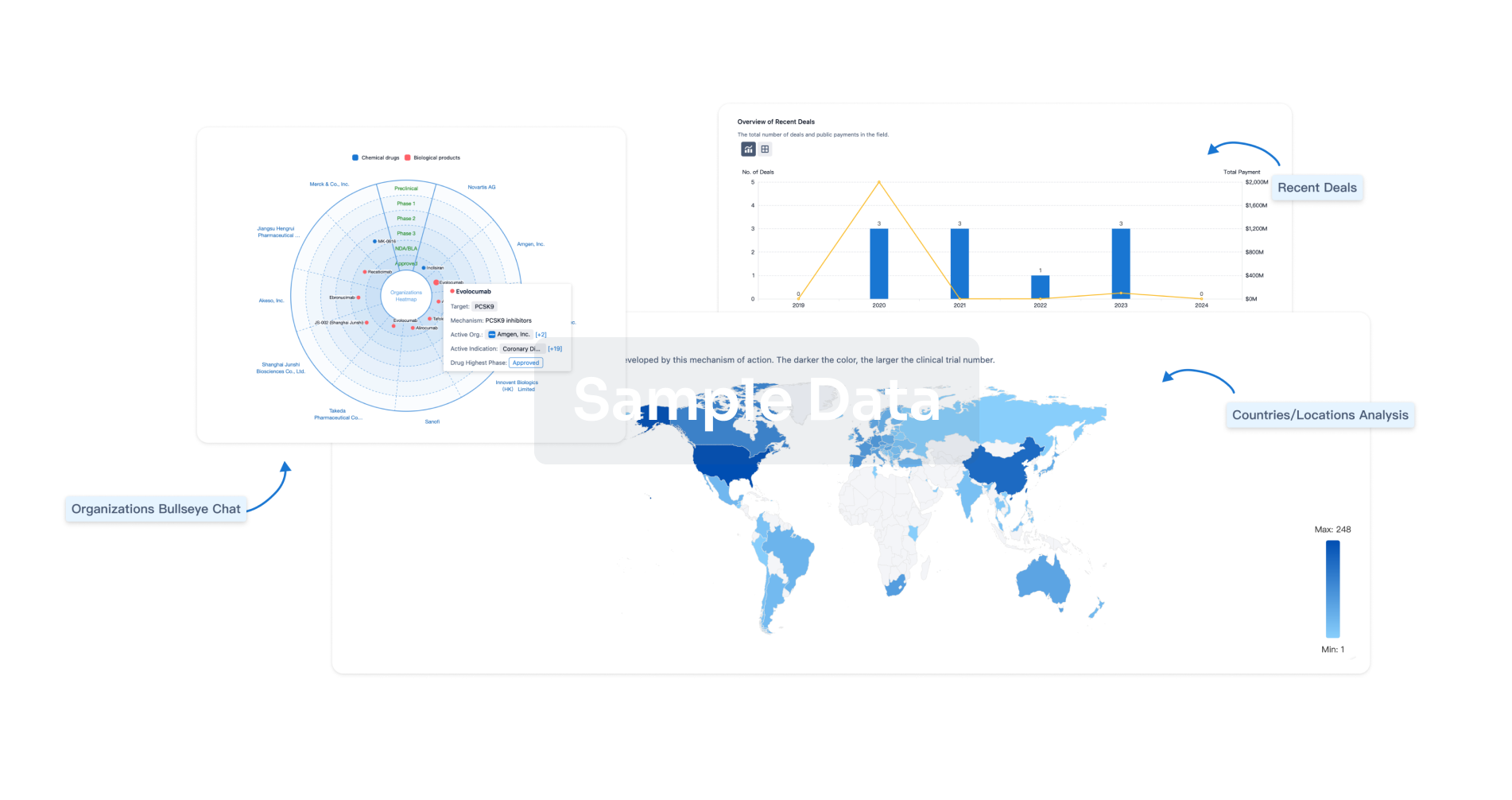
Perform a panoramic analysis of this field.
login
or
AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free
