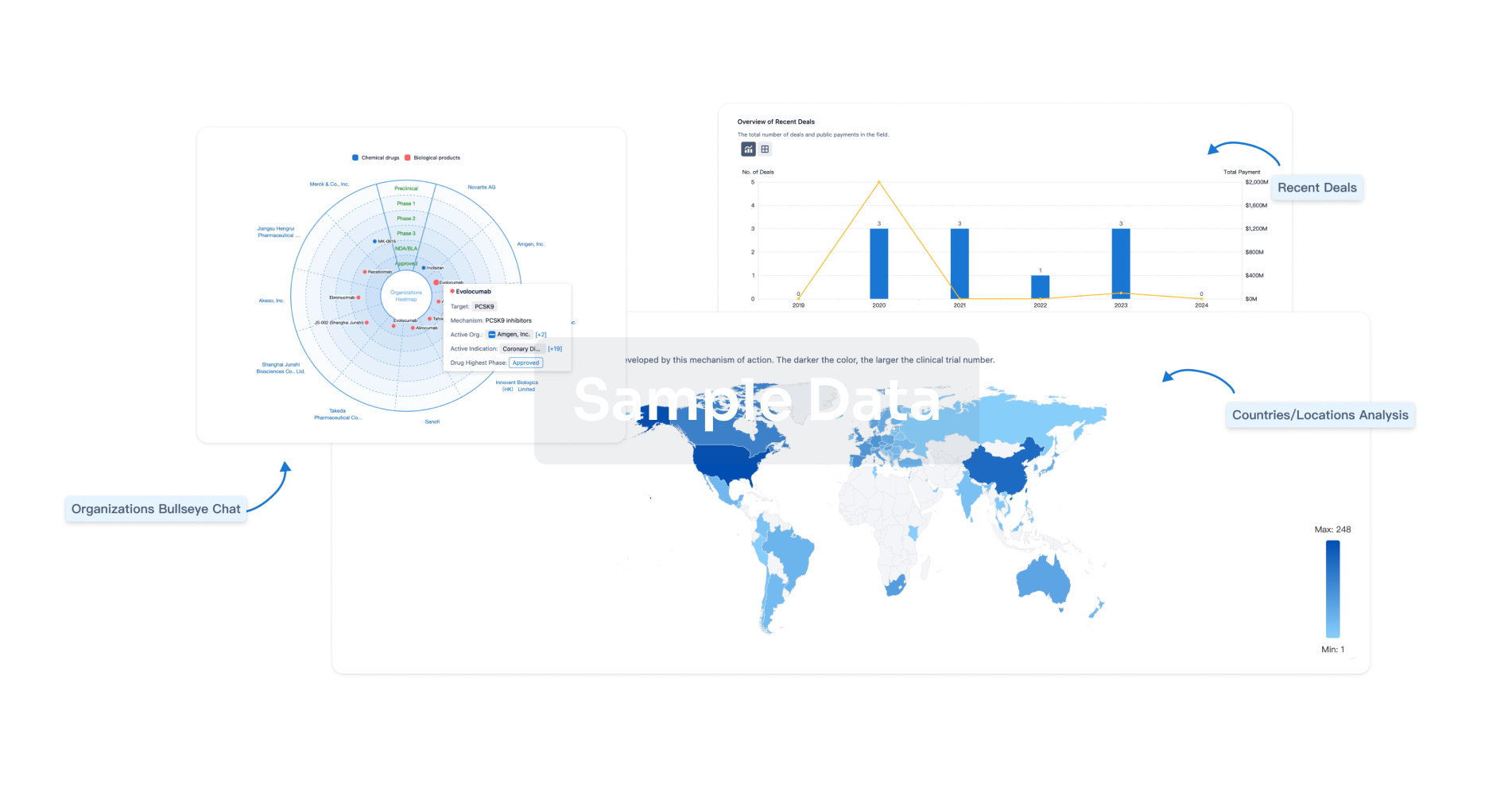
|
MechanismFree radicals inhibitors [+1] |
|
|
|
|
Drug Highest PhasePhase 2 |
First Approval Ctry. / Loc.- |
First Approval Date20 Jan 1800 |
A Phase 2b, Randomized, Placebo-Controlled, Double-Blind, Parallel Group Dose-Finding Study to Evaluate the Efficacy on Renal Function and Safety of RMC-035 in Participants At High Risk for Kidney Injury Following Open-Chest Cardiac Surgery
The goal of this clinical trial is to identify the optimal dose of RMC-035 for protection of long-term renal function in adult patients undergoing cardiac surgery who are at high risk of kidney injury. It will also learn about the safety of RMC-035. The main question it aims to answer is:
* Does RMC-035 protect the function of kidneys after surgery?
* Is RMC-035 safe?
Researchers will compare RMC-035 in high dose, RMC-035 in low dose and placebo to see if
* Kidney function better for participants treated with any of the RMC-035 doses?
* What medical problems do participants have when receiving RMC-035?
Participants will
* Receive 3 doses of RMC-035 or placebo: at the beginning of surgery, end of surgery and 24h after surgery
* Have extra checkups and tests during their hospital stay
* Visit the clinic at two extra occasions at 60 days and 90 days after surgery for checkups and tests
A Phase 2, Randomized, Placebo-Controlled, Double-Blind, Adaptive, Parallel Group Clinical Study to Evaluate the Efficacy and Safety of RMC-035 in Subjects at High Risk for Acute Kidney Injury Following Open-Chest Cardiac Surgery
This study evaluates RMC-035 compared to placebo for the prevention of acute kidney injury (AKI) in subjects who are at high risk for AKI following cardiac surgery. Half of the subjects will receive RMC-035 and the other half will receive placebo.
A Phase 1b, Randomised, Double-Blind, Parallel Treatment Group Clinical Study to Evaluate the Safety, Tolerability and Pharmacokinetics of RMC-035 in Subjects Undergoing Non-Emergent On-Pump Coronary Artery Bypass Graft and/or Valve Surgery
The purpose of the clinical study is to assess safety, tolerability and pharmacokinetics of RMC-035 for the prevention and treatment of acute kidney injury (AKI) in patients undergoing cardiac surgery.
100 Clinical Results associated with Hemoglobins x Free radicals
100 Translational Medicine associated with Hemoglobins x Free radicals
0 Patents (Medical) associated with Hemoglobins x Free radicals