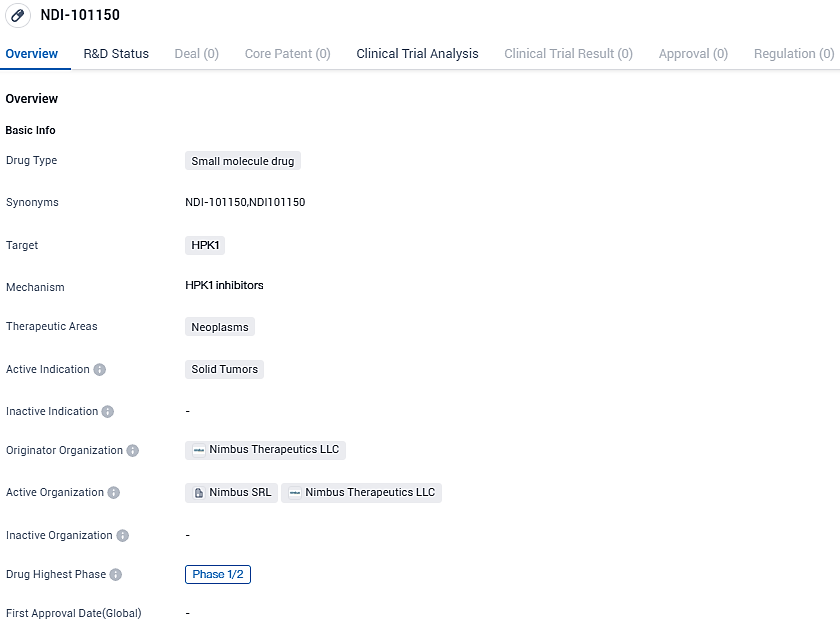
At the annual SITC meeting, Nimbus Therapeutics unveiled encouraging initial results from a clinical study on an HPK1 inhibitor for solid tumors
Nimbus Therapeutics, LLC, a clinical-stage organization that crafts and advances cutting-edge drugs via its robust computerized drug discovery platform, has now shared preliminary findings from its ongoing Phase 1/2 trial of NDI-101150, an HPK1 small-molecule inhibitor. These findings will be showcased in a poster presentation at the 38th Annual Meeting of the Society for Immunotherapy of Cancer held in San Diego, CA.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
The information shared at SITC comprises initial findings on the safety and efficacy of individual therapy from the dose escalation section of the trial. NDI-101150, used as monotherapy, provided clinical benefits to 3 patients. One patient diagnosed with renal cell carcinoma showed a CR - complete disappearance of any detectable cancer post-therapy - whereas two patients suffering from pancreatic cancer and renal cell carcinoma respectively showed an extended stable disease.
Significantly, the patient with renal cell carcinoma who attained a CR and clinical advantage with NDI-101150 monotherapy had previously undergone treatment with a regimen of nivolumab, an immune checkpoint inhibitor antibody, and had seen disease progression. Also, NDI-101150 displayed a tolerable safety profile below 200 mg/day, which is observed to be the non-tolerated dose.
“We are gratified to communicate the primary clinical results of our clinical study on NDI-101150. The tentative effectiveness we have observed thus far is quite inspiring and attests to the potentiality of NDI-101150 in treating patients with solid tumors,” stated Nathalie Franchimont, M.D., Ph.D., the Chief Medical Officer at Nimbus.
“HPK1 inhibition is a promising method due to its ability to stimulate not just T cells, similar to checkpoint inhibitors, but also B cells and dendritic cells. We are keen on disclosing further updates on NDI-101150, including information from combination cohorts ongoing and dose expansion cohorts,” Nathalie Franchimont further remarked.
The Phase 1/2 trial involves a multicenter, open-label study determining the safety, tolerability, pharmacokinetics, and preliminary anti-cancer activity of NDI-101150 administered as monotherapy or combined with pembrolizumab in adults suffering from advanced or metastatic solid tumors.
“The core mission driving Nimbus is to apply our expertise and technology to develop path-breaking drugs for patients,” expressed Jeb Keiper, M.S., MBA, Chief Executive Officer at Nimbus. “The findings shared at SITC underscore the potential unique and supreme profile of our HPK1 inhibitor, and we are steadfast in furthering its potential advantages for cancer patients.”
👇Please click on the picture link below for free registration or login directly if you have freemium accounts, you can browse the latest research progress on drugs, indications, organizations, clinical trials, clinical results, and drug patents related to this target.
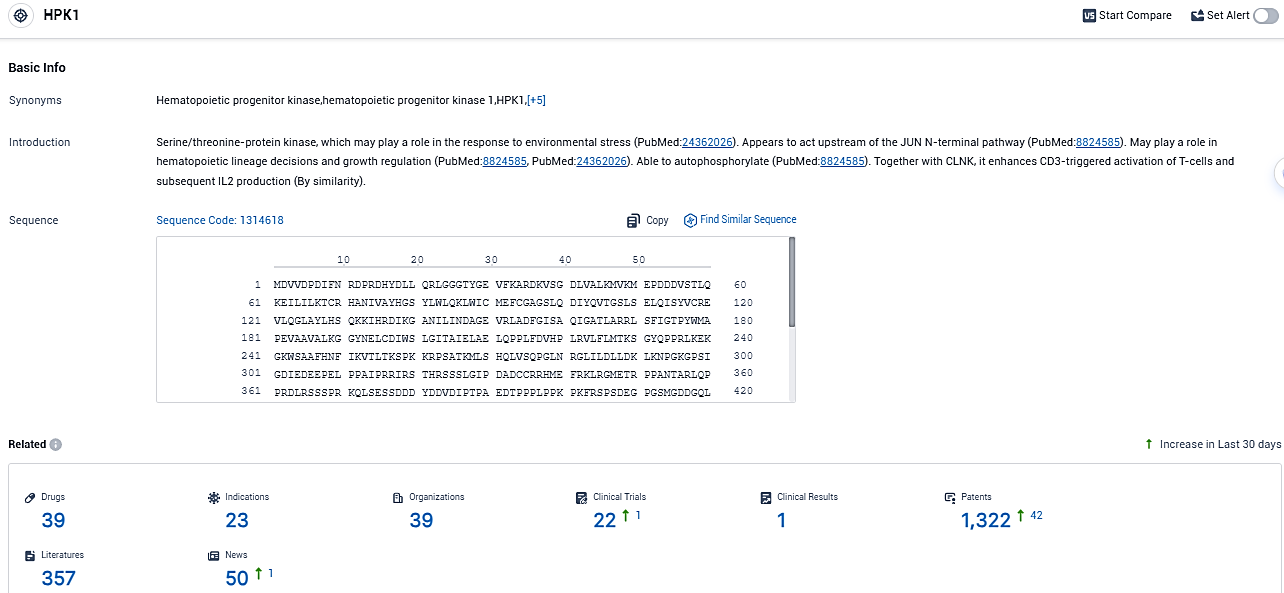
According to the data provided by the Synapse Database, As of November 3, 2023, there are 39 investigational drugs for the HPK1 target, including 23 indications, 39 R&D institutions involved, with related clinical trials reaching 22, and as many as 357 patents.
NDI-101150 is in the highest phase of clinical development, which is Phase 1/2. Clinical development is the process of testing a drug in humans to evaluate its safety and efficacy. It is important to note that this summary is based solely on the given information and does not include any additional subjective interpretations or fictional data.