EXS-21546: A Quick Look at Its R&D Progress and Clinical Results from the 2023 AACR
On 14 Apr 2023, data from first-in-human study of EXS21546, an A2A receptor antagonist, was reported at the AACR Congress.
EXS-21546's R&D Progress
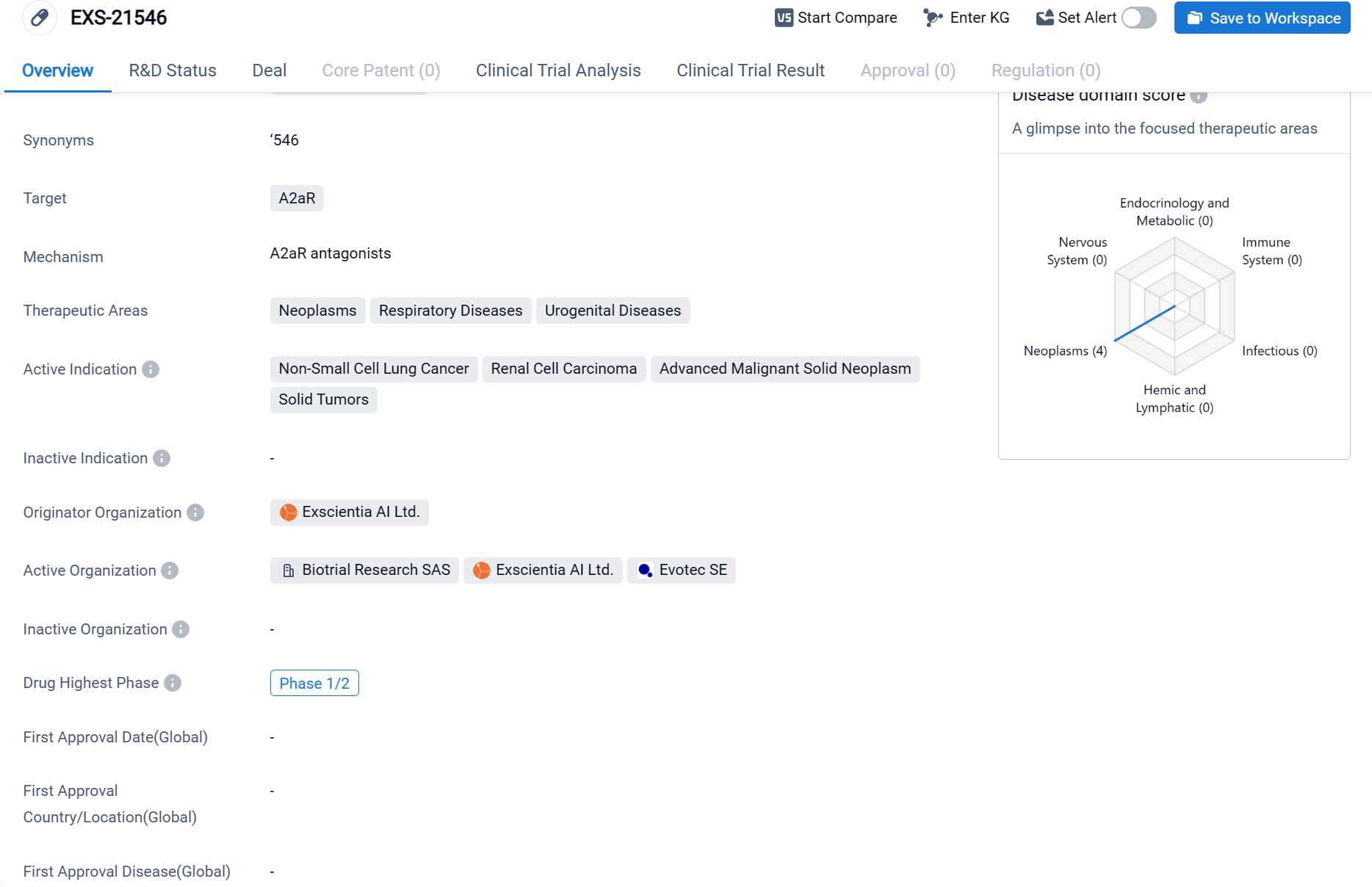
EXS-21546 is a small molecule drug that targets the A2aR receptor. It is being developed by Exscientia AI Ltd., an organization specializing in artificial intelligence-driven drug discovery. The therapeutic areas that EXS-21546 aims to address are neoplasms (abnormal growth of cells), respiratory diseases, and urogenital diseases. Within these therapeutic areas, the drug is specifically indicated for the treatment of non-small cell lung cancer, renal cell carcinoma, advanced malignant solid neoplasms, and solid tumors.
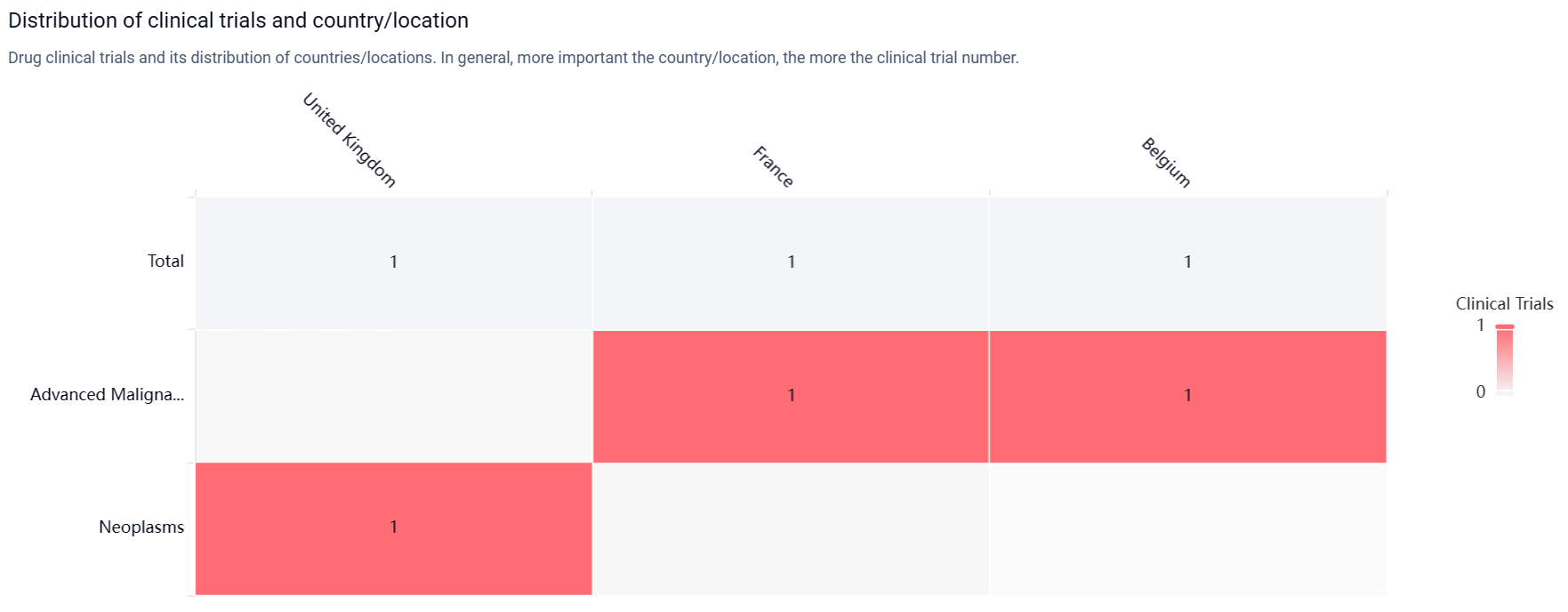
According to the Patsnap Synapse, EXS-21546 is currently in the highest phase of clinical development, Phase 1/2. And the clinical trial areas for EXS-21546 are primarily in the United Kingdom, France, and Belgium. The key indication is Advanced Malignant Solid Neoplasm.
Detailed Clinical Result of EXS-21546
The randomized, sequential assignment, quadruple masking clinical trial (NCT04727138) was conducted in RCC/NSCLC patients.
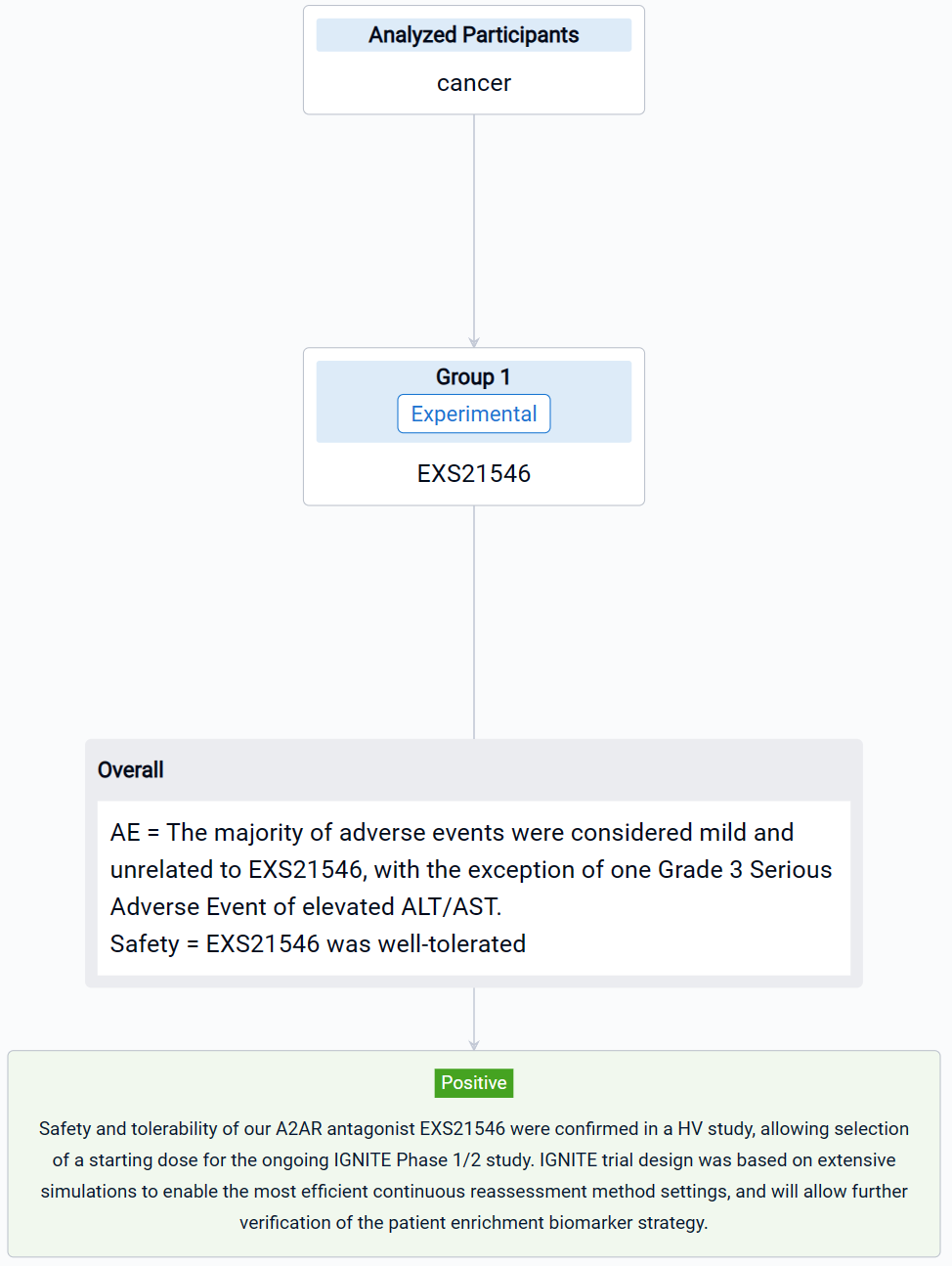
In this study, a 3-part Phase 1 FIH study in 60 HV was completed. Parts 1 and 2 were double-blind, placebo-controlled single ascending dose (SAD) and multiple ascending dose (MAD) studies investigating EXS21546 safety, tolerability, PK and PD. Part 3 was a 3-period, open label, randomized, sequential study evaluating bioavailability of oral EXS21546 as a granule in capsule vs reference powder suspension. The ongoing Phase 1/2 IGNITE trial will study EXS21546 in patients with immunotherapy relapsed or refractory RCC or NSCLC directly in combination with a PD-1 inhibitor. This combination aims to leverage the properties of A2AR antagonism in the tumor microenvironment, to drive the immune system through PD-1 inhibition. IGNITE will also provide clinical data to support the validation of our adenosine signature to identify patients with adenosine rich tumor microenvironments who may benefit from treatment (Alt et al, 2022). Data will be continually reassessed to determine the recommended Phase 2 dose. A dose escalation phase will be followed by an expansion phase.
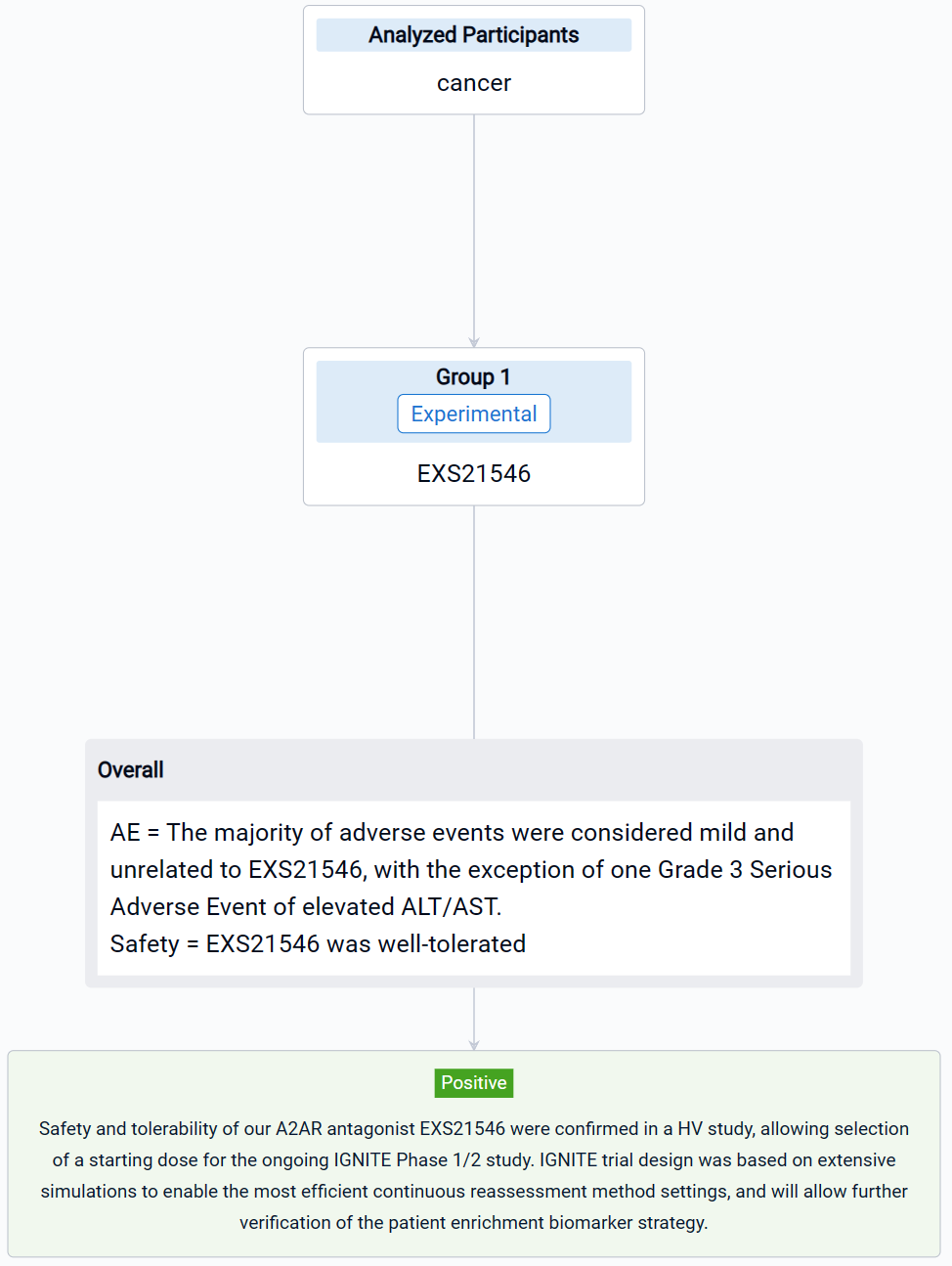
The result showed that PK/PD modeling of Phase 1 study results, along with the EXS21546 safety profile, allowed identification of a potential therapeutic starting dose. PK results aligned with design specifications, based upon predictive preclinical modeling, and support twice-daily (BID) dosing for continuous A2AR antagonism. EXS21546 induced dose-dependent inhibition of CREB phosphorylation in CD8-positive cells, with the PD profile mirroring plasma exposure. Inhibition of A2AR signaling was sustained over the BID dosing period, demonstrating a level of lasting target engagement. PoP PK/PD simulations informed the starting dose and dosing regimen for the Phase 1/2 trial. EXS21546 was well-tolerated with no CNS adverse events reported in the SAD at all doses and in the MAD at 150 mg BID. The majority of adverse events were considered mild and unrelated to EXS21546, with the exception of one Grade 3 Serious Adverse Event of elevated ALT/AST.
It can be concluded that safety and tolerability of our A2AR antagonist EXS21546 were confirmed in a HV study, allowing selection of a starting dose for the ongoing IGNITE Phase 1/2 study. IGNITE trial design was based on extensive simulations to enable the most efficient continuous reassessment method settings, and will allow further verification of the patient enrichment biomarker strategy.
How to Easily View the Clinical Results Using Synapse Database?
If you want to know the other clinical results of popular conferences, please lick on the “Clinical Results” on the homepage of Patsnap Synapse, which provides multi-dimensional screening and filtering of drugs, indications, targets, companies, result evaluation, release date, popular conferences, etc. to help you quickly locate the data you need.
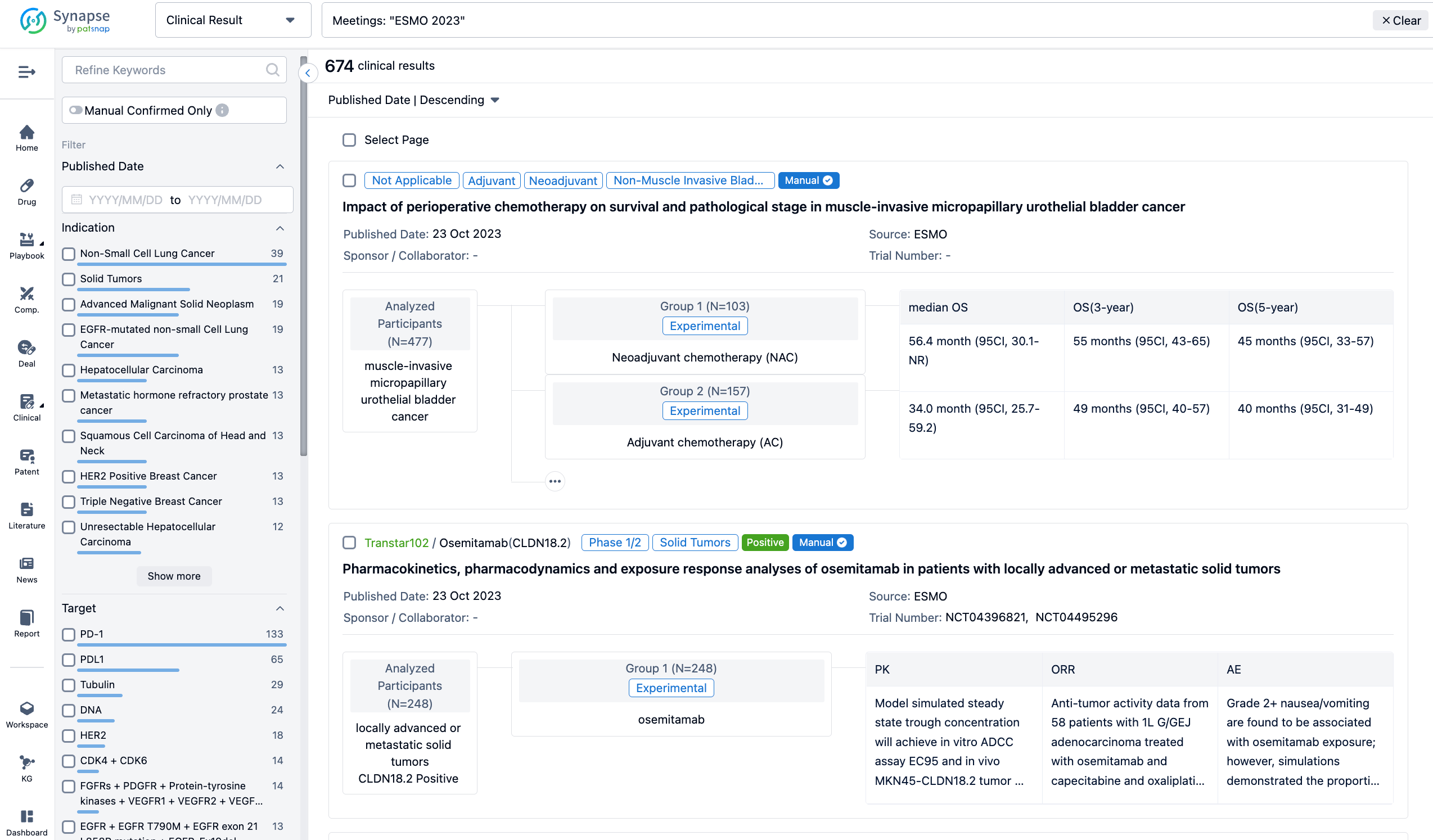
Select the clinical meeting you are interested in, such as ESMO. In the results, you can quickly locate the data you want to view by indication, phase and drug name.
A single result clearly shows important information such as registration number, phase, indication, Sponsor/Collaborator, biomarker, Trial number, dosing regimen and more.
If you would like to view more information about this result, you can go to the result detail page by clicking on the title.
Above the headings, we provide the original source of the outcome data. The basic information is supplemented with more information beyond the list, such as company, study. design, etc.

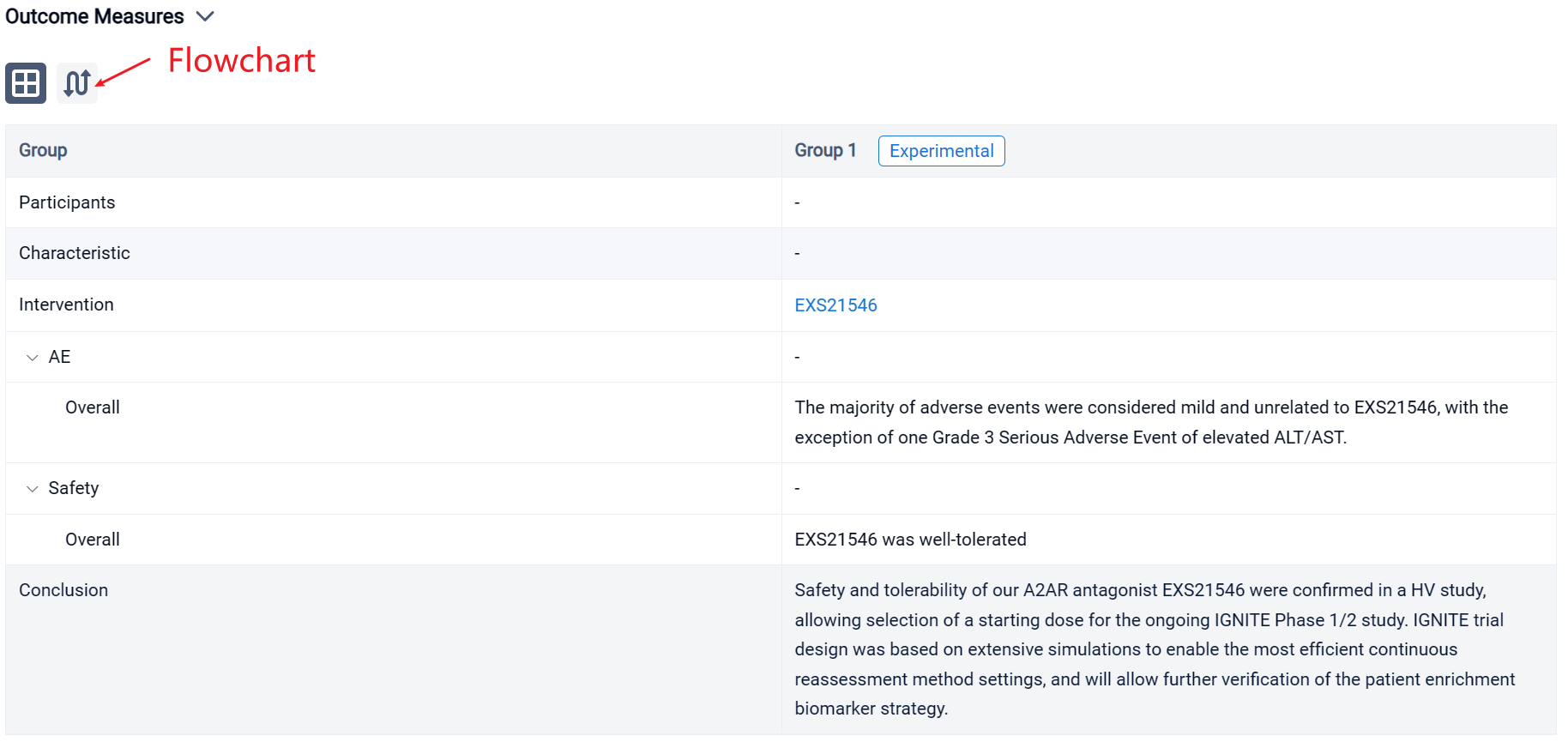
In the important Outcome Measures section, we provide both list and flowchart forms, which are convenient for you to overview the comparison group information and core indicator data.
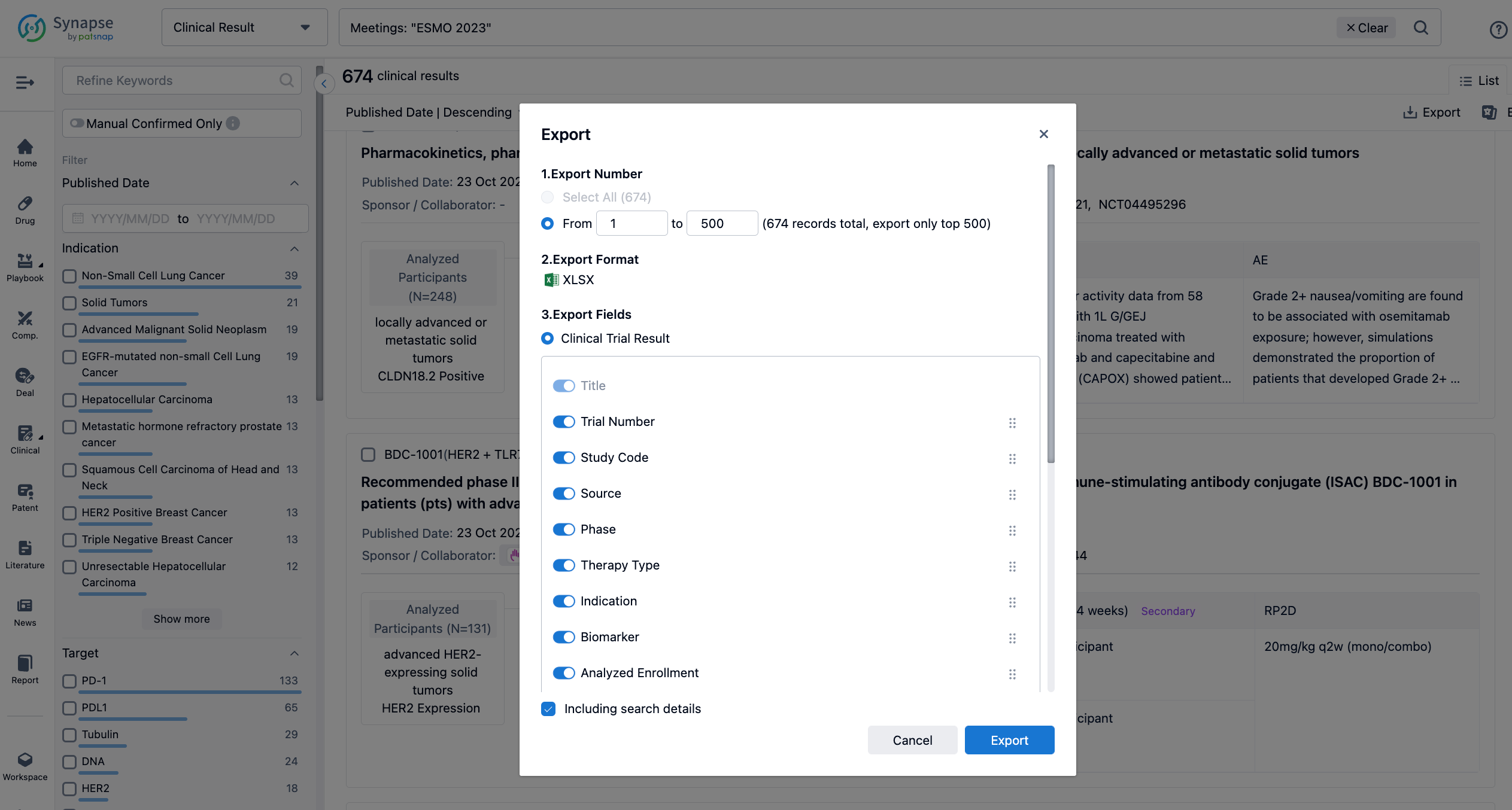
Finally, if you need to download these results, you can conveniently check the check boxes on the left side of the list, or directly click the "Export" button to download the data for personalized analysis and file sharing.
Click on the image below to embark on a brand new journey of drug discovery!