Opdivo and CABOMETYX Exhibit Long-Term Survival in 4-Year CheckMate-9ER Trial for Advanced RCC
Bristol Myers Squibb alongside Exelixis, Inc. has revealed outcomes with a four-year monitoring duration from the clinical study known as CheckMate -9ER. This study assessed the efficacy of the drug combination comprising Opdivo® (nivolumab) and CABOMETYX® (cabozantinib) in contrast to sunitinib monotherapy. The patient group under investigation consisted of individuals who had not received prior treatment for their advanced or metastatic renal cell carcinoma.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
Continually updated findings indicate that the combination of Opdivo and CABOMETYX surpasses sunitinib in sustaining progression-free survival and enhancing the rates of objective responses among patients, transcending the prognostic categorization determined by International Metastatic Renal Cell Carcinoma Database Consortium scores. Patients receiving this therapeutic duo also exhibited superior overall survival.
Addressing the complexity of managing renal cell carcinoma, particularly in its advanced and metastatic stages, Maria Teresa Bourlon from the Urologic Oncology Clinic at the Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán in Mexico highlighted the dire prognoses typically faced by these patients.
With updated data from the CheckMate -9ER trial, the continued advantage of using nivolumab conjoined with cabozantinib as a premier line of attack against this formidable condition has been substantiated. The robust benefits of this regimen include sustained performance in all key trial markers and a notable 23% diminution in mortality risk, as further articulated by Maria Teresa Bourlon.
Expounding on the pivotal advances in treatment options, Dana Walker, M.D., M.S.C.E., vice president at Bristol Myers Squibb, remarked on the establishment of a novel standard of care through the employment of Opdivo in conjunction with CABOMETYX. This approach not only holds promise for a broad spectrum of advanced malignancies, especially those of the genitourinary system but also showcases the enduring impact of Opdivo-based combinations. These strategies are at the forefront of therapeutic modalities for advanced RCC, offering hope for extended survival independent of risk stratification.
👇Please click on the picture link below for free registration or login directly if you have freemium accounts, you can browse the latest research progress on drugs, indications, organizations, clinical trials, clinical results, and drug patents related to this target.
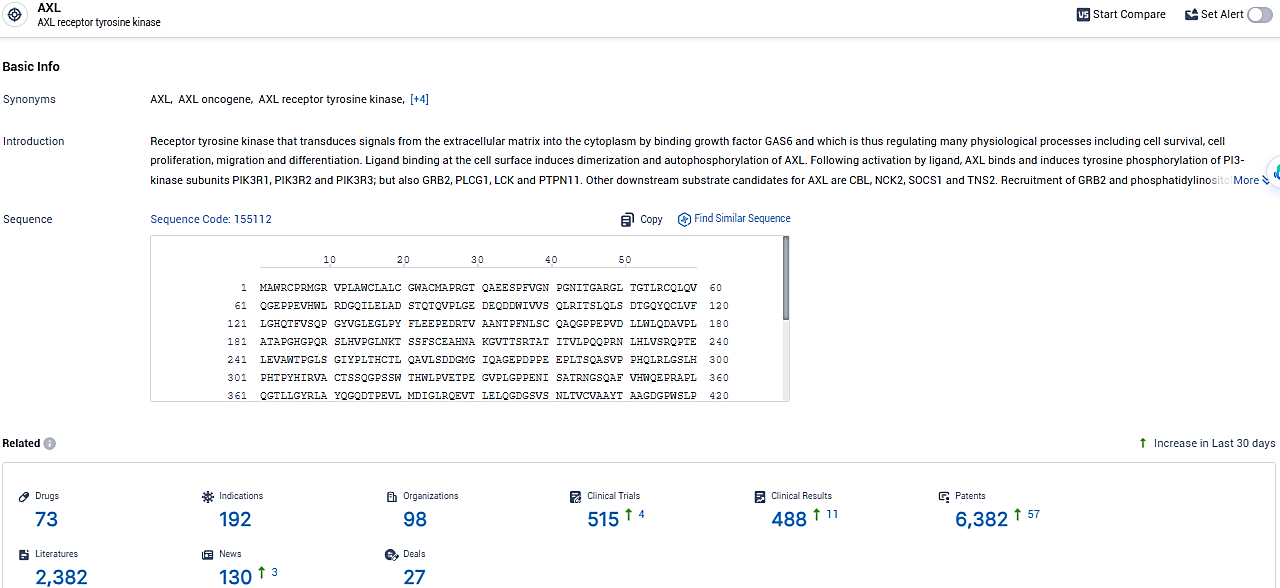
According to the data provided by the Synapse Database, As of January 29, 2024, there are 73 investigational drugs for the AXL target, including 192 indications, 98 R&D institutions involved, with related clinical trials reaching 515, and as many as 6382 patents.
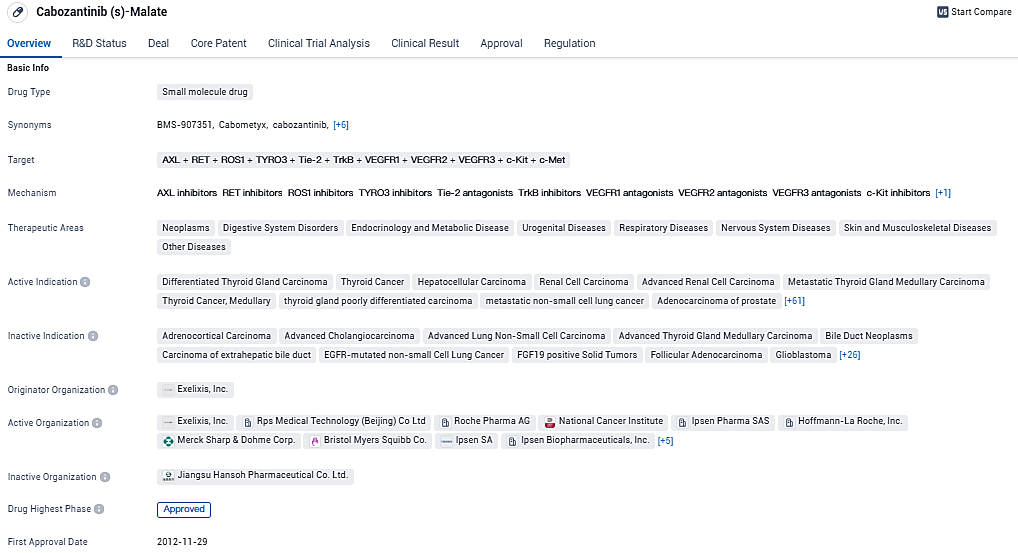
Cabozantinib is a promising drug with a wide range of potential therapeutic applications. The drug's approval in multiple countries and its regulatory designations further support its potential as an important treatment option in the pharmaceutical industry.