Request Demo
Last update 14 Dec 2025
Tocilizumab
Last update 14 Dec 2025
Overview
Basic Info
Drug Type Monoclonal antibody |
Synonyms Atlizumab, MRA-SC, Tocilizumab (Genetical Recombination) + [14] |
Target |
Action antagonists |
Mechanism IL-6RA antagonists(Interleukin-6 receptor alpha subunit antagonists) |
Therapeutic Areas |
Inactive Indication |
Originator Organization |
Active Organization |
Inactive Organization |
License Organization |
Drug Highest PhaseApproved |
First Approval Date Japan (11 Apr 2005), |
RegulationBreakthrough Therapy (United States), Emergency Use Authorization (United States), Orphan Drug (United States), Orphan Drug (Japan) |
Login to view timeline
Structure/Sequence
Sequence Code 12264H

Source: *****
Sequence Code 12270L

Source: *****
Related
617
Clinical Trials associated with TocilizumabNCT03787290
Examining Immune-Based Mechanisms of Action for Mild-Intensity Whole Body Hyperthermia (WBH) in the Treatment of Major Depressive Disorder
Overall, the objective of this pilot study is to utilize the IL-6 receptor antagonist tocilizumab to prospectively evaluate the role of IL-6 in the antidepressant and immunological effects of whole body hyperthermia (WBH). The study seeks to replicate findings thus far that WBH has an antidepressant effect by administering the intervention at a site not involved in studies to date. Moreover, the current proposal may help the investigators better understand the role of IL-6 in the pathogenesis and treatment of depression which might point to novel immune-based interventions for Major Depressive Disorder (MDD). Finally, the current proposal holds promise for better understanding of a novel treatment for MDD, which is among the leading causes of health-related disability in the world.
Start Date01 Dec 2026 |
Sponsor / Collaborator |
NCT06381661
PALETTE- Adaptive Platform Trial for Personnalisation of Sepsis Treatment in Children and Adults: a Multi-national, Treatable Traits-guided, Adaptive, Bayesian Basket Trial"
PALETTE is a perpetual adaptive platform to efficiently study sepsis interventions within 'treatable traits' in all-ages patients enabling prompt evaluation of pandemic treatments. Treatable traits, therapeutic targets identified by phenotypes or endotypes (defined by biological mechanism or by treatment response) through validated biomarkers (measurable characteristic reflecting normal or pathogenic processes, or treatment responses), may include multi-omics, cellular, immune, metabolic, endocrine features, or intelligent algorithms. PALETTE Bayesian adaptive design enables parallel investigations of multiple interventions for sepsis, and quick inclusion of pandemic pathogens. PALETTE's new conceptual model will respond to the challenges of standard approaches, i.e. series of sepsis trials, each investigating one or two interventions, expensive, time consuming, and inappropriate in pandemic context.
Start Date01 Apr 2026 |
Sponsor / Collaborator |
CTIS2025-521371-31-00
PALETTE - Adaptive platform trial for personalisation of sepsis treatment in children and adults: a multi-national, treatable traits-guided, adaptive, exploratory, bayesian basket trial
Start Date01 Jan 2026 |
Sponsor / Collaborator |
100 Clinical Results associated with Tocilizumab
Login to view more data
100 Translational Medicine associated with Tocilizumab
Login to view more data
100 Patents (Medical) associated with Tocilizumab
Login to view more data
6,909
Literatures (Medical) associated with Tocilizumab01 Jan 2026·INTERNATIONAL JOURNAL OF FOOD MICROBIOLOGY
Physicochemical properties, microbial diversity, metabolites and their potential relationships of traditional Chinese Zhacai fermented for different periods
Article
Author: Wang, Xinhui ; Ren, Ting ; Wang, Xinyi ; Gu, Yuanqing ; Nie, Xin ; Liu, Dayu ; Zhao, Zhiping ; Luo, Yuanli ; Zhao, Nan ; Liu, Yuling
In this study, we investigated the physicochemical properties, dominant microorganisms, non-volatile metabolites and their relationships of traditional Chinese Zhacai (TCZ) at the "second-time pickling and pressing" stage fermentation. pH and amino acid nitrogen contents exhibited an increasing and then decreasing trend. Metagenomics analysis revealed the dominant bacterial species Lactiplantibacillus (Lpb.) plantarum and Latilactobacillus (Lat.) sakei, and fungal species Debaryomyces (D.) hansenii. LEfSe analysis identified a total of 47 differentially abundant bacteria, including Lpb. plantarum and Lat. Sakei. Metabolomics analysis indicated organic acids and their derivatives, organic heterocyclic compounds, lipids and lipid-like molecules as the major differential metabolites. Spearman's correlation results showed significant correlations among differential bacteria, non-volatile metabolites and physicochemical properties. Unexpectedly, differential fungi were not observed in TCZ at this fermentation period. Pathway enrichment identified phenylpropanoid biosynthesis and tryptophan metabolism as dominant metabolic pathways, both influencing the TCZ flavor development. This study contributes to further understanding of quality formation of TCZ, providing a foundational resource for future optimization and industrial application.
01 Dec 2025·Journal of Translational Autoimmunity
Acylcarnitine enrichment as a characteristic of rheumatoid arthritis fibroblast-like synoviocyte metabolic fingerprint
Article
Author: Hørslev-Petersen, Kim ; Østergaard, Mikkel ; van Vollenhoven, Ronald ; Sureshkumar, Reshmi ; Guma, Monica ; Nurmohamed, Michael ; Hultgård Ekwall, Anna-Karin ; Gröndal, Gerdur ; Nordström, Dan ; Vasileiadis, Georgios K ; Fatima, Tahzeeb ; Zhang, Yuan ; Haavardsholm, Espen A ; Lend, Kristina ; Rudin, Anna ; Maglio, Cristina ; Gudbjornsson, Bjorn ; Borén, Jan ; Lampa, Jon ; Uhlig, Till ; Hetland, Merete L ; Sokka-Isler, Tuulikki ; Laudette, Marion
Objective:
In rheumatoid arthritis (RA), fibroblast-like synoviocytes (FLS) alter their metabolism to support their activation. We aimed to analyse the full spectrum of metabolic alterations associated with RA by performing untargeted metabolomics in RA FLS vs. non-inflamed (NI) FLS.
Methods:
Untargeted annotated metabolomics was performed using mass spectrometry on ten primary RA and seven NI FLS culture extracts and 220 serum samples from participants with early RA from the randomised controlled NORD-STAR trial. Carnitine-related proteins were measured with Western blot. FLS bioenergetic profile was assessed with a Seahorse flux analyser.
Results:
Metabolomics analysis based on 138 annotated metabolites revealed a distinct metabolic fingerprint between RA and NI FLS. Of the 12 metabolites enriched in RA FLS, 11 were acylcarnitines. Pro-inflammatory stimulation of NI FLS also led to acylcarnitine accumulation. RA FLS exhibited lower levels of CD36, a fatty acid transporter, but similar levels of L-carnitine transporter, and carnitine palmitoyltransferase 1 A and 2 compared to NI FLS. Seahorse analyses showed no difference in fatty acid oxidation between RA and NI FLS; however, RA FLS displayed mitochondrial dysfunction and energetic impairment. Serum acylcarnitine content decreased after 24 weeks of treatment with methotrexate combined with abatacept or tocilizumab in patients with early RA achieving remission.
Conclusion:
Acylcarnitine accumulation is a characteristic of RA FLS metabolic fingerprint and could be linked to mitochondrial dysfunction. In patients with early RA, acylcarnitine content in serum decreases after successful anti-rheumatic treatment. These results indicate a dysregulation in acylcarnitine metabolism in RA at the joint level and systemically.
01 Dec 2025·JOURNAL OF MEDICAL ECONOMICS
Comparison of utilization and total medicare fee-for-service expenditures for subcutaneous versus intravenous versions of a select group of therapies.
Article
Author: Sarocco, Phillip ; Kardel, Peter ; Maynard, Jason ; Sheetz, Caitlin
INTRODUCTION:
Comparisons of subcutaneous (SC) and intravenous (IV) delivery of biotherapeutics in Europe have revealed differences in cost, convenience, and preference. In the US, claims-based studies comparing SC and IV delivery have been limited. This study compared resource utilization and expenditures between cohorts of patients receiving the same drug by the SC or IV route of administration (ROA) across several indications.
METHODS:
Medicare Fee-For-Service claims were compared between cohorts of patients who received SC- or IV-delivered abatacept, belimumab, daratumumab, infliximab, tocilizumab, trastuzumab/pertuzumab, or vedolizumab. For each drug, utilization rates and healthcare expenditures per service site were compared between ROAs. Observed differences in baseline characteristics were accounted for using Inverse Probability Treatment Weighting models that balanced pre-index differences between ROAs per drug. Each ROA comparison per drug and service site was weighted and conducted independently.
RESULTS:
Across seven drugs, a total sample size of 158,632 (72,820 IV; 85,812 SC) was analyzed. For most comparisons, high-spend sites were utilized at a higher rate for IV administration. In particular, all comparisons revealed more frequent hospital outpatient department utilization for the IV ROA. For five of the seven drugs, SC treatment was associated with lower mean total Medicare expenditures, with savings of up to $56,000 per patient annually. Although three SC treatments had higher medication index spend, the total spend for these drugs across sites was significantly lower for SC delivery.
LIMITATIONS:
Limitations of this study include differences in billing between the SC and IV ROAs, potential treatment selection bias, and the assumption of equivalent efficacy.
CONCLUSIONS:
To our knowledge, this study was the first and largest US Medicare claims analysis, comparing patients receiving SC or IV versions of the same therapeutic across multiple drugs and indications. Findings demonstrated that SC delivery may facilitate reduced resource utilization and expenditures across drugs and disease indications.
273
News (Medical) associated with Tocilizumab08 Dec 2025
SAN FRANCISCO and SUZHOU, China, Dec. 7, 2025 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures, and commercializes high-quality medicines for the treatment of oncology, autoimmune, cardiovascular and metabolic, ophthalmology and other major disease areas, announced the initial data of the first-in-human trial of IBI3003, a novel trispecific antibody targeting G protein-coupled receptor C5D (GPRC5D), B-cell maturation antigen (BCMA), and CD3 for the treatment of relapsed or refractory multiple myeloma (R/R MM), in an oral presentation at the 2025 American Society of Hematology (ASH) Annual Meeting. IBI3003 demonstrated favorable tolerability and a manageable safety profile. Despite the relatively short follow-up duration, IBI3003 has shown encouraging efficacy signals, particularly in high-risk patients with extramedullary disease (EMD) or those who have previously received anti-BCMA and/or anti-GPRC5D targeted therapies.
IBI3003 is a novel trispecific antibody targeting GPRC5D, BCMA and CD3 simultaneously. Its dual-targeting design against BCMA and GPRC5 aims to overcome single antigen escape in multiple myeloma (MM). In preclinical studies, IBI3003 exhibited superior in vivo anti-tumor activity over marketed benchmark bispecific antibodies in mouse models, with particularly prominent tumor-killing efficacy in in vitro cell models with low expression of BCMA and GPRC5D. Currently, Innovent is conducting a Phase 1/2 clinical trial (NCT06083207) of IBI3003 in China and Australia to evaluate its safety, tolerability, and efficacy in patients with R/R MM.
The first phase of the study enrolled eligible R/R MM patients who had failed ≥2 lines of previous anti-myeloma therapies that included at least a proteasome inhibitor (PI), an immunomodulatory drug (IMiD), and an anti-CD38 mAb; and must be relapsed or refractory to their last anti-myeloma regimen. Prior BCMA- or GPRC5D-targeting therapy was allowed.
IBI3003 was administered subcutaneously once weekly (QW). For patients who have received continuous treatment for ≥6 months and achieved partial response (PR) or better for ≥2 months could switch to Q2W as maintenance. To reduce the risk of cytokine release syndrome (CRS), 1 to 3 priming doses were included in the study design.
A total of 39 patients were enrolled in this phase in China and Australia, with a dose range of 0.1 μg/kg to 800 μg/kg. The median age of patients was 62 years (range: 40-79), 64.1% of whom were classified as high-risk per mSMART criteria, and 46.2% had ≥1 EMD. The median number of prior lines of therapy was 4 (range: 1-10). All patients had received at least three classes of drugs (PI, IMiD, and anti-CD38 antibody), 51.3% had received at least five classes of drugs (at least 2 PIs, 2 IMiDs, and 1 anti-CD38 antibody), 41% had previously received anti-BCMA and/or anti-GPRC5D therapies, and 76.9% were refractory to the last treatment. As of the data cutoff date, November 7, 2025, the median follow-up duration was 3.25 months (range: 0.4-7.4), and the median treatment duration was 12.14 weeks (range: 1.0-33.0).
Manageable Safety Profile of IBI3003 in R/R MM Patients
Dose-limiting toxicity (DLT) only occurred in 2 patients, both of whom experienced Grade 4 platelet count decreased and recovered.
97.4% of patients experienced treatment-emergent adverse events (TEAEs). Common TEAEs included CRS, neutrophil count decrease, anemia, lymphocyte count decrease, white blood cell count decrease, and platelet count decrease.
Hematological disorders were the most common Grade ≥3 TEAEs that mainly occurred during step-up dosing and were manageable and recoverable.
The incidences of CRS and immune effector cell-associated neurotoxicity syndrome (ICANS) were 64.1% and 6.1%, respectively, all of which were Grade 1-2 and resolved with treatment. Prophylactic use of tocilizumab may reduce incidence, severity, and duration CRS.
The incidence of all-grade infections was 48.7%, with Grade ≥3 infections reported in 28.2% of patients.
For GPRC5D target-related TEAEs involving the oral cavity, skin, and nails, no Grade ≥3 oral TEAEs were observed. Most skin and nail TEAEs were Grade 1-2, with only 2 patients experiencing Grade 3 rash.
Encouraging Efficacy and Depth of Response Observed with IBI3003 at Doses ≥120 μg/kg
Encouraging efficacy was observed with a median follow-up of 3.25 months: Among patients treated with ≥120 μg/kg (n=24), the overall response rate (ORR) was 83.3%, including 4 cases of stringent complete response (sCR), 7 cases of very good partial response (VGPR), and 9 cases of partial response (PR).
Among patients treated with ≥120 μg/kg, the ORR was 80% in 10 patients with EMD and 77.8% in 9 patients who had previously received anti-BCMA and/or anti-GPRC5D therapies.
Among patients who achieved CR or better as assessed by central laboratory next-generation sequencing (NGS) testing, the minimal residual disease (MRD) negativity rate was 100% (n=4).
Potent and Sustained Pharmacodynamic Responses Observed with IBI3003 in R/R MM Patients
Biomarker analysis showed that baseline soluble BCMA (sBCMA) levels were high and variable in R/R MM patients (median level: 198 ng/mL, range: 10-3010 ng/mL).
A profound and durable decline in serum sBCMA across 120, 360 and 540 μg/kg groups was observed, demonstrating a strong pharmacodynamic response.
IBI3003 has demonstrated favorable tolerability and a manageable safety profile in R/R MM patients, with encouraging efficacy signals observed at doses ≥120 μg/kg. Efficacy responses were also observed in high-risk patients, including those with EMD or prior anti-BCMA and/or anti-GPRC5D therapies. Current follow-up remains relatively short, and deeper anti-tumor responses are expected with continued treatment and observation. Dose optimization for IBI3003 is ongoing in the Phase 1 study.
Professor Peng Liu, Zhongshan Hospital Affiliated to Fudan University, stated, "Patients with R/R MM have a poor prognosis after failing standard treatments, including PI, IMiD, and anti-CD38 therapies, with an ORR of only 29.8%, a median progression-free survival of 4.6 months, and a median overall survival of 12.4 months[1]. Therefore, there is an urgent unmet clinical need for these patients, particularly those with high-risk features such as EMD or prior anti-BCMA and/or anti-GPRC5D therapies. The dual-target coverage of BCMA and GPRC5D by IBI3003 addresses the issues of antigen expression heterogeneity and treatment resistance associated with single-target drugs, reducing tumor escape. Meanwhile, its optimized CD3 affinity enables precise T-cell activation for tumor killing while also improving safety. In the disclosed Phase 1 study results, IBI3003 showed a manageable safety profile and impressive efficacy data at doses ≥120 μg/kg, with an ORR of 83.3%. It also demonstrated significant efficacy in high-risk patients with EMD or prior anti-BCMA and/or anti-GPRC5D therapies, fully reflecting its potential in overcoming R/R MM. We look forward to the long-term follow-up survival data with continuous IBI3003 treatment."
About IBI3003 (Anti-GPRC5D/BCMA/CD3 Trispecific Antibody)
IBI3003 is a tri-specific TCE developed using Innovent's proprietary Sanbody platform to target both GPRC5D and BCMA. Designed to overcome drug resistance driven by single-antigen tumor escape, IBI3003 has exhibited superior in vivo antitumor activity in preclinical studies compared with marketed benchmark TCEs, especially in cell models with low BCMA and GPRC5D expression. Innovent is currently conducting a Phase 1/2 clinical trial (NCT06083207) of IBI3003 in China and Australia to evaluate its safety, tolerability, and efficacy in patients with R/R MM.
About Innovent
Innovent is a leading biopharmaceutical company founded in 2011 with the mission to empower patients worldwide with affordable, high-quality biopharmaceuticals. The company discovers, develops, manufactures and commercializes innovative medicines that target some of the most intractable diseases. Its pioneering therapies treat cancer, cardiovascular and metabolic, autoimmune and eye diseases. Innovent has launched 17 products in the market. It has 1 new drug applications under regulatory review, 4 assets in Phase 3 or pivotal clinical trials and 15 more molecules in early clinical stage. Innovent partners with over 30 global healthcare companies, including Lilly, Sanofi, Incyte, LG Chem and MD Anderson Cancer Center.
Guided by the motto, "Start with Integrity, Succeed through Action," Innovent maintains the highest standard of industry practices and works collaboratively to advance the biopharmaceutical industry so that first-rate pharmaceutical drugs can become widely accessible. For more information, visit , or follow Innovent on Facebook and LinkedIn.
Statement:
(1) Innovent does not recommend the use of any unapproved drug (s)/indication (s).
(2) Ramucirumab (Cyramza®) and Selpercatinib (Retsevmo®) and Pirtobrutinib (Jaypirca®) were developed by Eli Lilly and Company.
Forward-looking statement
This news release may contain certain forward-looking statements that are, by their nature, subject to significant risks and uncertainties. The words "anticipate", "believe", "estimate", "expect", "intend" and similar expressions, as they relate to Innovent, are intended to identify certain of such forward-looking statements. Innovent does not intend to update these forward-looking statements regularly.
These forward-looking statements are based on the existing beliefs, assumptions, expectations, estimates, projections and understandings of the management of Innovent with respect to future events at the time these statements are made. These statements are not a guarantee of future developments and are subject to risks, uncertainties and other factors, some of which are beyond Innovent's control and are difficult to predict. Consequently, actual results may differ materially from information contained in the forward-looking statements as a result of future changes or developments in our business, Innovent's competitive environment and political, economic, legal and social conditions.
Innovent, the Directors and the employees of Innovent assume (a) no obligation to correct or update the forward-looking statements contained in this site; and (b) no liability in the event that any of the forward-looking statements does not materialize or turn out to be incorrect.
References
[1] Mateos M , Weisel K , Stefano V D ,et al.LocoMMotion: a prospective, non-interventional, multinational study of real-life current standards of care in patients with relapsed and/or refractory multiple myeloma[J].Leukemia, 2022, 36:1371 - 1376.DOI:10.1038/s41375-022-01531-2.
SOURCE Innovent Biologics
21%
more press release views with
Request a Demo
Clinical ResultPhase 1ASHAACR
07 Dec 2025
SANTA MONICA, Calif.--(BUSINESS WIRE)--Kite, a Gilead Company (Nasdaq: GILD), presented a new analysis today demonstrating that second-line Yescarta® (axicabtagene ciloleucel) therapy offers consistent benefits in patients with relapsed/refractory large B-cell lymphoma (R/R LBCL), even among those ineligible for the previous standard of care, high-dose chemotherapy followed by an autologous stem cell transplant (ASCT).
Results were shared from the combined analysis of four-year data from the landmark ZUMA-7 Phase 3 pivotal study of Yescarta for R/R LBCL and two-year data from the Phase 2 ALYCANTE study, designed by French collaborative group LYSA and sponsored by LYSARC, for transplant-ineligible patients. The findings were presented (Abstract #3714) during the 67th American Society of Hematology (ASH) Annual Meeting and Exposition.
“Patients with large B-cell lymphoma who are ineligible for stem cell transplants face limited treatment options and poor outcomes due to age, co-morbidities and other factors,” said Professor Roch Houot, Head of Haematology Department, University Hospital of Rennes, France and coordinator of the ALYCANTE study. “This analysis reinforces that Yescarta should be considered earlier in a patient’s treatment paradigm given its curative potential as a one-time treatment and further establishes CAR T as the new standard of care for second-line treatment of large B-cell lymphoma.”
The efficacy analyses included 178 and 69 patients from the ZUMA-7 and ALYCANTE trials, respectively.
After two years:
Overall survival (OS) rate, meaning the percentage of patients in the trials who were still alive, was 64.9% in the pooled analysis, 62.8% in ZUMA-7, and 70.8% in ALYCANTE. Historically, the prognosis for R/R LBCL was very poor; prior to the introduction of new therapies like CAR Ts, the two-year survival rate was only about 20%.
Event-free survival (EFS) rate, meaning the percentage of patients who were still alive and had not seen their disease worsen or experienced other major complications, was 45.2% in the pooled analysis, 45.4% in ZUMA-7, and 44.7% in ALYCANTE.
Progression-free survival (PFS) rate, meaning the percentage of patients who were still living without their disease getting worse, was 47.4% in the pooled analysis, 47.6% in ZUMA-7, and 46.8% in ALYCANTE.
Additionally:
After three months, 55.6% of the patients in the pooled analysis showed a complete metabolic response (CMR), meaning their disease was barely, if at all, detectable. CMR was 51.2% in ZUMA-7 and 67.7% in ALYCANTE.
After one year after treatment, overall response rate (ORR), meaning patients saw their cancer significantly shrink or completely disappear, was 46.6% in the pooled analysis, 46.5% in ZUMA-7, and 46.8% in ALYCANTE.
For those patients who responded well to treatment, 61% saw that positive response continue after a full year. Pooled 12-month duration of response (DOR) was 61.0%, 60.6% in ZUMA-7, and 62.1% in ALYCANTE.
In the safety analysis, which included 170 ZUMA-7 and 62 ALYCANTE Yescarta-infused patients, safety outcomes were comparable between the two studies. The incidence of grade ≥3 treatment-emergent adverse events (TEAEs) was similar between the ZUMA-7 and ALYCANTE patients, 91.2% and 88.7% respectively, with a pooled incidence of 90.5%. Pooled incidence of grade ≥3 neurologic events, neutropenias, and anemia were 19.8% (ZUMA-7 21.2%, ALYCANTE 16.1%), 64.7% (ZUMA-7 70.0%, ALYCANTE 50.0%), and 27.6% (ZUMA-7 30.0%, ALYCANTE 21.0%), respectively.
Across both studies, patients generally experienced similar long-term improvements in their quality of life after initial treatment challenges. At Day 50 after treatment, patients in both trials showed a transient decline (-7.2 in ZUMA-7 and -6.3 in ALYCANTE) for EORTC QLQ-C30 global health status, and -12.9 for physical function in both. However, patients in ALYCANTE reported a meaningful improvement in their overall well-being (EQ-5D-5L VAS) by Day 100 (+9.9), while ZUMA-7 patients reached a similar meaningful improvement (+9.9) at Day 150, which continued through Month 12. By month 24, both ALYCANTE and ZUMA-7 showed continued improvements of global health status.
“This analysis offers compelling evidence of Yescarta’s consistent, durable efficacy and safety profile across a broad range of patients, including those with difficult-to-treat relapsed or refractory disease who historically faced very limited options and a poor prognosis,” said Gallia Levy, MD, PhD, Senior Vice President and Global Head of Development, Kite. “These robust data further reinforce Yescarta’s potential as a treatment with curative intent, reflecting Kite’s deep commitment to transforming patient outcomes.”
About LBCL
Globally, LBCL is the most common type of non-Hodgkin lymphoma. In the United States, more than 18,000 people are diagnosed with LBCL each year. About 30-40% of patients with LBCL will need second-line treatment, as their cancer will either relapse (return) or become refractory (not respond) to initial treatment.
About ALYCANTE Study
ALYCANTE (NCT04531046) is a Phase 2 study evaluating the efficacy and safety of Yescarta in patients with R/R LBCL after one prior line of therapy who were deemed ineligible for high-dose chemotherapy and ASCT, sponsored by the LYSA/LYSARC collaborative group. The primary endpoint was the complete metabolic response at three months from Yescarta infusion. The study was funded by Kite, a Gilead Company, and carried out with Yescarta manufactured by Kite.
About LYSA and LYSARC
The Lymphoma Study Association (LYSA) is an independent, multidisciplinary network and an international leader in clinical lymphoma and CLL/WM research. With over 500 experts and a network of 90 centers across France, Belgium, and Portugal, it conducts clinical trials ranging from early-stage treatment evaluation to the development of new therapeutic strategies.
The Lymphoma Academic Research Organization (LYSARC), based in France, is the largest European academic organization dedicated to clinical lymphoma and CLL/WM research. As the operational arm of LYSA, LYSARC sponsors and leads research initiatives and operates specialized platforms in pathology, biology, and imaging. It manages and coordinates numerous clinical trials (phases 1 to 4) each year, as well as non-interventional and data reuse studies. LYSARC is a key research expert, leading innovative projects on an international scale.
About ZUMA-7 Study
Based on the primary efficacy endpoint results of ZUMA-7, the U.S. Food & Drug Administration approved Yescarta as initial treatment of R/R LBCL in April 2022. The EU granted approval in October 2022, followed by approvals in several other countries including Australia, Canada, Great Britain, Israel, Japan and Switzerland.
ZUMA-7 is a randomized, open-label, global, multicenter, Phase 3 study evaluating the safety and efficacy of Yescarta versus standard of care (SOC) for second-line therapy in adult patients with relapsed or refractory LBCL within 12 months of first-line therapy. The SOC for initial treatment of R/R LBCL has been a multi-step process involving platinum-based salvage combination chemotherapy regimen, and for responders, HDT and ASCT. In the study, 359 patients in 77 centers around the world were randomized (1:1) to receive a single infusion of Yescarta or SOC second-line treatment. The primary endpoint was EFS as determined by blinded central review and defined as the time from randomization to the earliest date of disease progression per Lugano Classification, commencement of new lymphoma therapy, or death from any cause. Key secondary endpoints include objective response rate and OS. Additional secondary endpoints included patient-reported outcomes (PROs) and safety. Per hierarchical testing of primary and key secondary endpoints and group sequential testing of OS, an interim analysis of OS occurred at the time of the primary EFS.
About Yescarta
Please see full Prescribing Information, including BOXED WARNING below and Medication Guide.
YESCARTA is a CD19-directed genetically modified autologous T cell immunotherapy indicated for the treatment of:
Adult patients with large B-cell lymphoma that is refractory to first-line chemoimmunotherapy or that relapses within 12 months of first-line chemoimmunotherapy.
Adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL) not otherwise specified, primary mediastinal large B-cell lymphoma, high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma.
Limitations of Use: YESCARTA is not indicated for the treatment of patients with primary central nervous system lymphoma.
U.S. IMPORTANT SAFETY INFORMATION
BOXED WARNING: CYTOKINE RELEASE SYNDROME, NEUROLOGIC TOXICITIES, and SECONDARY HEMATOLOGICAL MALIGNANCIES
CYTOKINE RELEASE SYNDROME (CRS)
CRS, including fatal or life-threatening reactions, occurred following treatment with YESCARTA. CRS occurred in 90% (379/422) of patients with non-Hodgkin lymphoma (NHL), including ≥ Grade 3 CRS in 9%. CRS occurred in 93% (256/276) of patients with large B-cell lymphoma (LBCL), including ≥ Grade 3 in 9%. Among patients with LBCL who died after receiving YESCARTA, 4 had ongoing CRS events at the time of death. For patients with LBCL in ZUMA-1, the median time to onset of CRS was 2 days following infusion (range: 1-12 days) and the median duration was 7 days (range: 2-58 days). For patients with LBCL in ZUMA-7, the median time to onset of CRS was 3 days following infusion (range: 1-10 days) and the median duration was 7 days (range: 2-43 days).
CRS occurred in 84% (123/146) of patients with indolent non-Hodgkin lymphoma (iNHL) in ZUMA-5, including ≥ Grade 3 CRS in 8%. Among patients with iNHL who died after receiving YESCARTA, 1 patient had an ongoing CRS event at the time of death. The median time to onset of CRS was 4 days (range: 1-20 days) and median duration was 6 days (range: 1-27 days) for patients with iNHL.
Key manifestations of CRS (≥ 10%) in all patients combined included fever (85%), hypotension (40%), tachycardia (32%), chills (22%), hypoxia (20%), headache (15%), and fatigue (12%). Serious events that may be associated with CRS include, cardiac arrhythmias (including atrial fibrillation and ventricular tachycardia), renal insufficiency, cardiac failure, respiratory failure, cardiac arrest, capillary leak syndrome, multi-organ failure, and hemophagocytic lymphohistiocytosis/macrophage activation syndrome (HLH/MAS).
The impact of tocilizumab and/or corticosteroids on the incidence and severity of CRS was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received tocilizumab and/or corticosteroids for ongoing Grade 1 events, CRS occurred in 93% (38/41), including 2% (1/41) with Grade 3 CRS; no patients experienced a Grade 4 or 5 event. The median time to onset of CRS was 2 days (range: 1-8 days) and the median duration of CRS was 7 days (range: 2-16 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Thirty-one of the 39 patients (79%) developed CRS and were managed with tocilizumab and/or therapeutic doses of corticosteroids with no patients developing ≥ Grade 3 CRS. The median time to onset of CRS was 5 days (range: 1-15 days) and the median duration of CRS was 4 days (range: 1-10 days). Although there is no known mechanistic explanation, consider the risk and benefits of prophylactic corticosteroids in the context of pre-existing comorbidities for the individual patient and the potential for the risk of Grade 4 and prolonged neurologic toxicities.
Confirm that 2 doses of tocilizumab are available prior to infusion of YESCARTA. Monitor patients at least daily for 7 days following infusion for signs and symptoms of CRS. Monitor patients for signs or symptoms of CRS for 2 weeks after infusion. Counsel patients to seek immediate medical attention should signs or symptoms of CRS occur at any time. At the first sign of CRS, institute treatment with supportive care, tocilizumab, or tocilizumab and corticosteroids as indicated.
NEUROLOGIC TOXICITIES
Neurologic toxicities including immune effector cell-associated neurotoxicity syndrome (ICANS) that were fatal or life-threatening occurred following treatment with YESCARTA. Neurologic toxicities occurred in 78% (330/422) of patients with NHL receiving YESCARTA, including ≥ Grade 3 in 25%.
Neurologic toxicities occurred in 87% (94/108) of patients with LBCL in ZUMA-1, including ≥ Grade 3 in 31% and in 74% (124/168) of patients in ZUMA-7 including ≥ Grade 3 in 25%. The median time to onset was 4 days (range: 1-43 days) and the median duration was 17 days for patients with LBCL in ZUMA-1. The median time to onset for neurologic toxicity was 5 days (range: 1-133 days) and median duration was 15 days in patients with LBCL in ZUMA-7. Neurologic toxicities occurred in 77% (112/146) of patients with iNHL, including ≥ Grade 3 in 21%. The median time to onset was 6 days (range: 1-79 days) and the median duration was 16 days. Ninety-eight percent of all neurologic toxicities in patients with LBCL and 99% of all neurologic toxicities in patients with iNHL occurred within the first 8 weeks of YESCARTA infusion. Neurologic toxicities occurred within the first 7 days of infusion in 87% of affected patients with LBCL and 74% of affected patients with iNHL.
The most common neurologic toxicities (≥ 10%) in all patients combined included encephalopathy (50%), headache (43%), tremor (29%), dizziness (21%), aphasia (17%), delirium (15%), and insomnia (10%). Prolonged encephalopathy lasting up to 173 days was noted. Serious events, including aphasia, leukoencephalopathy, dysarthria, lethargy, and seizures occurred. Fatal and serious cases of cerebral edema and encephalopathy, including late-onset encephalopathy, have occurred.
The impact of tocilizumab and/or corticosteroids on the incidence and severity of neurologic toxicities was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received corticosteroids at the onset of Grade 1 toxicities, neurologic toxicities occurred in 78% (32/41) and 20% (8/41) had Grade 3 neurologic toxicities; no patients experienced a Grade 4 or 5 event. The median time to onset of neurologic toxicities was 6 days (range: 1-93 days) with a median duration of 8 days (range: 1-144 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Of those patients, 85% (33/39) developed neurologic toxicities; 8% (3/39) developed Grade 3 and 5% (2/39) developed Grade 4 neurologic toxicities. The median time to onset of neurologic toxicities was 6 days (range: 1-274 days) with a median duration of 12 days (range: 1-107 days). Prophylactic corticosteroids for management of CRS and neurologic toxicities may result in higher grade of neurologic toxicities or prolongation of neurologic toxicities, delay the onset, and decrease the duration of CRS.
Monitor patients for signs and symptoms of neurologic toxicities following infusion at least daily for 7 days; and for 2 weeks thereafter and treat promptly. Advise patients to avoid driving for at least 2 weeks following infusion.
HYPERSENSITIVITY REACTIONS
Allergic reactions may occur with the infusion of YESCARTA. Serious hypersensitivity reactions, including anaphylaxis, may be due to dimethyl sulfoxide (DMSO) or residual gentamicin in YESCARTA.
SERIOUS INFECTIONS
Severe or life-threatening infections occurred after YESCARTA infusion. Infections (all grades) occurred in 45% of patients with NHL. Grade 3 or higher infections occurred in 17% of patients, including ≥ Grade 3 infections with an unspecified pathogen in 12%, bacterial infections in 5%, viral infections in 3%, and fungal infections in 1%. YESCARTA should not be administered to patients with clinically significant active systemic infections. Monitor patients for signs and symptoms of infection before and after infusion and treat appropriately. Administer prophylactic antimicrobials according to local guidelines.
Febrile neutropenia was observed in 36% of patients with NHL and may be concurrent with CRS. In the event of febrile neutropenia, evaluate for infection and manage with broad-spectrum antibiotics, fluids, and other supportive care as medically indicated.
In immunosuppressed patients, including those who have received YESCARTA, life-threatening and fatal opportunistic infections including disseminated fungal infections (e.g., candida sepsis and aspergillus infections) and viral reactivation (e.g., human herpes virus-6 [HHV-6] encephalitis and JC virus progressive multifocal leukoencephalopathy [PML]) have been reported. The possibility of HHV-6 encephalitis and PML should be considered in immunosuppressed patients with neurologic events and appropriate diagnostic evaluations should be performed.
Hepatitis B virus (HBV) reactivation, in some cases resulting in fulminant hepatitis, hepatic failure, and death, has occurred in patients treated with drugs directed against B cells, including YESCARTA. Perform screening for HBV, HCV, and HIV and management in accordance with clinical guidelines before collection of cells for manufacturing.
PROLONGED CYTOPENIAS
Patients may exhibit cytopenias for several weeks following lymphodepleting chemotherapy and YESCARTA infusion. Grade 3 or higher cytopenias not resolved by Day 30 following YESCARTA infusion occurred in 39% of all patients with NHL and included neutropenia (33%), thrombocytopenia (13%), and anemia (8%). Monitor blood counts after infusion.
HYPOGAMMAGLOBULINEMIA
B-cell aplasia and hypogammaglobulinemia can occur in patients receiving YESCARTA. Hypogammaglobulinemia was reported as an adverse reaction in 14% of all patients with NHL. Monitor immunoglobulin levels after treatment and manage using infection precautions, antibiotic prophylaxis, and immunoglobulin replacement.
The safety of immunization with live viral vaccines during or following YESCARTA treatment has not been studied. Vaccination with live virus vaccines is not recommended for at least 6 weeks prior to the start of lymphodepleting chemotherapy, during YESCARTA treatment, and until immune recovery following treatment.
SECONDARY MALIGNANCIES
Patients treated with YESCARTA may develop secondary malignancies. T cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies, including YESCARTA. Mature T cell malignancies, including CAR-positive tumors, may present as soon as weeks following infusion, and may include fatal outcomes.
Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs, contact Kite at 1-844-454-KITE (5483) to obtain instructions on patient samples to collect for testing.
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥ 20%) in:
patients with LBCL in ZUMA-7 included fever, CRS, fatigue, hypotension, encephalopathy, tachycardia, diarrhea, headache, musculoskeletal pain, nausea, febrile neutropenia, chills, cough, infection with unspecified pathogen, dizziness, tremor, decreased appetite, edema, hypoxia, abdominal pain, aphasia, constipation, and vomiting.
patients with LBCL in ZUMA-1 included CRS, fever, hypotension, encephalopathy, tachycardia, fatigue, headache, decreased appetite, chills, diarrhea, febrile neutropenia, infections with pathogen unspecified, nausea, hypoxia, tremor, cough, vomiting, dizziness, constipation, and cardiac arrhythmias.
Please see full Prescribing Information, including BOXED WARNING and Medication Guide.
About Gilead and Kite Oncology
Gilead and Kite Oncology are working to transform how cancer is treated. We are innovating with next-generation therapies, combinations and technologies to deliver improved outcomes for people with cancer. We are purposefully building our oncology portfolio and pipeline to address the greatest gaps in care. From antibody-drug conjugate technologies and small molecules to cell therapy-based approaches, we are creating new possibilities for people with cancer.
Forward-Looking Statements
This press release includes forward-looking statements, within the meaning of the Private Securities Litigation Reform Act of 1995 that are subject to risks, uncertainties and other factors, including the ability of Gilead and Kite to initiate, progress or complete clinical trials within currently anticipated timelines or at all, and the possibility of unfavorable results from ongoing or additional clinical studies, including those involving Yescarta (such as ZUMA-7 and ALYCANTE); uncertainties relating to regulatory applications and related filing and approval timelines, including pending or potential applications for indications currently under evaluation; the possibility that Gilead and Kite may make a strategic decision to discontinue development of these programs and, as a result, these programs may never be successfully commercialized for the indications currently under evaluation; and any assumptions underlying any of the foregoing. These and other risks are described in detail in Gilead’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, as filed with the U.S. Securities and Exchange Commission. These risks, uncertainties and other factors could cause actual results to differ materially from those referred to in the forward-looking statements. All statements other than statements of historical fact are statements that could be deemed forward-looking statements. The reader is cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties and is cautioned not to place undue reliance on these forward-looking statements. All forward-looking statements are based on information currently available to Gilead and Kite, and Gilead and Kite assume no obligation and disclaim any intent to update any such forward-looking statements.
Yescarta, Gilead, the Gilead logo, Kite, the Kite logo are trademarks of Gilead Sciences, Inc., or its related companies.
For more information on Kite, please visit the company’s website at www.kitepharma.com. Follow Kite on social media on X (@KitePharma) and LinkedIn.
Clinical ResultPhase 2Phase 3Drug ApprovalASH
07 Dec 2025
– Benefits Seen Regardless of Eligibility for Previous Standard of Care of High-Dose Chemotherapy and Stem Cell Transplant for Second-Line Treatment –
SANTA MONICA, Calif.--(BUSINESS WIRE)-- Kite, a Gilead Company (Nasdaq: GILD), presented a new analysis today demonstrating that second-line Yescarta® (axicabtagene ciloleucel) therapy offers consistent benefits in patients with relapsed/refractory large B-cell lymphoma (R/R LBCL), even among those ineligible for the previous standard of care, high-dose chemotherapy followed by an autologous stem cell transplant (ASCT).
Results were shared from the combined analysis of four-year data from the landmark ZUMA-7 Phase 3 pivotal study of Yescarta for R/R LBCL and two-year data from the Phase 2 ALYCANTE study, designed by French collaborative group LYSA and sponsored by LYSARC, for transplant-ineligible patients. The findings were presented (Abstract #3714) during the 67th American Society of Hematology (ASH) Annual Meeting and Exposition.
“Patients with large B-cell lymphoma who are ineligible for stem cell transplants face limited treatment options and poor outcomes due to age, co-morbidities and other factors,” said Professor Roch Houot, Head of Haematology Department, University Hospital of Rennes, France and coordinator of the ALYCANTE study. “This analysis reinforces that Yescarta should be considered earlier in a patient’s treatment paradigm given its curative potential as a one-time treatment and further establishes CAR T as the new standard of care for second-line treatment of large B-cell lymphoma.”
The efficacy analyses included 178 and 69 patients from the ZUMA-7 and ALYCANTE trials, respectively.
After two years:
Overall survival (OS) rate, meaning the percentage of patients in the trials who were still alive, was 64.9% in the pooled analysis, 62.8% in ZUMA-7, and 70.8% in ALYCANTE. Historically, the prognosis for R/R LBCL was very poor; prior to the introduction of new therapies like CAR Ts, the two-year survival rate was only about 20%. Event-free survival (EFS) rate, meaning the percentage of patients who were still alive and had not seen their disease worsen or experienced other major complications, was 45.2% in the pooled analysis, 45.4% in ZUMA-7, and 44.7% in ALYCANTE. Progression-free survival (PFS) rate, meaning the percentage of patients who were still living without their disease getting worse, was 47.4% in the pooled analysis, 47.6% in ZUMA-7, and 46.8% in ALYCANTE.
Additionally:
After three months, 55.6% of the patients in the pooled analysis showed a complete metabolic response (CMR), meaning their disease was barely, if at all, detectable. CMR was 51.2% in ZUMA-7 and 67.7% in ALYCANTE. After one year after treatment, overall response rate (ORR), meaning patients saw their cancer significantly shrink or completely disappear, was 46.6% in the pooled analysis, 46.5% in ZUMA-7, and 46.8% in ALYCANTE. For those patients who responded well to treatment, 61% saw that positive response continue after a full year. Pooled 12-month duration of response (DOR) was 61.0%, 60.6% in ZUMA-7, and 62.1% in ALYCANTE.
In the safety analysis, which included 170 ZUMA-7 and 62 ALYCANTE Yescarta-infused patients, safety outcomes were comparable between the two studies. The incidence of grade ≥3 treatment-emergent adverse events (TEAEs) was similar between the ZUMA-7 and ALYCANTE patients, 91.2% and 88.7% respectively, with a pooled incidence of 90.5%. Pooled incidence of grade ≥3 neurologic events, neutropenias, and anemia were 19.8% (ZUMA-7 21.2%, ALYCANTE 16.1%), 64.7% (ZUMA-7 70.0%, ALYCANTE 50.0%), and 27.6% (ZUMA-7 30.0%, ALYCANTE 21.0%), respectively.
Across both studies, patients generally experienced similar long-term improvements in their quality of life after initial treatment challenges. At Day 50 after treatment, patients in both trials showed a transient decline (-7.2 in ZUMA-7 and -6.3 in ALYCANTE) for EORTC QLQ-C30 global health status, and -12.9 for physical function in both. However, patients in ALYCANTE reported a meaningful improvement in their overall well-being (EQ-5D-5L VAS) by Day 100 (+9.9), while ZUMA-7 patients reached a similar meaningful improvement (+9.9) at Day 150, which continued through Month 12. By month 24, both ALYCANTE and ZUMA-7 showed continued improvements of global health status.
“This analysis offers compelling evidence of Yescarta’s consistent, durable efficacy and safety profile across a broad range of patients, including those with difficult-to-treat relapsed or refractory disease who historically faced very limited options and a poor prognosis,” said Gallia Levy, MD, PhD, Senior Vice President and Global Head of Development, Kite. “These robust data further reinforce Yescarta’s potential as a treatment with curative intent, reflecting Kite’s deep commitment to transforming patient outcomes.”
About LBCL
Globally, LBCL is the most common type of non-Hodgkin lymphoma. In the United States, more than 18,000 people are diagnosed with LBCL each year. About 30-40% of patients with LBCL will need second-line treatment, as their cancer will either relapse (return) or become refractory (not respond) to initial treatment.
About ALYCANTE Study
ALYCANTE (NCT04531046) is a Phase 2 study evaluating the efficacy and safety of Yescarta in patients with R/R LBCL after one prior line of therapy who were deemed ineligible for high-dose chemotherapy and ASCT, sponsored by the LYSA/LYSARC collaborative group. The primary endpoint was the complete metabolic response at three months from Yescarta infusion. The study was funded by Kite, a Gilead Company, and carried out with Yescarta manufactured by Kite.
About LYSA and LYSARC
The Lymphoma Study Association (LYSA) is an independent, multidisciplinary network and an international leader in clinical lymphoma and CLL/WM research. With over 500 experts and a network of 90 centers across France, Belgium, and Portugal, it conducts clinical trials ranging from early-stage treatment evaluation to the development of new therapeutic strategies.
The Lymphoma Academic Research Organization (LYSARC), based in France, is the largest European academic organization dedicated to clinical lymphoma and CLL/WM research. As the operational arm of LYSA, LYSARC sponsors and leads research initiatives and operates specialized platforms in pathology, biology, and imaging. It manages and coordinates numerous clinical trials (phases 1 to 4) each year, as well as non-interventional and data reuse studies. LYSARC is a key research expert, leading innovative projects on an international scale.
About ZUMA-7 Study
Based on the primary efficacy endpoint results of ZUMA-7, the U.S. Food & Drug Administration approved Yescarta as initial treatment of R/R LBCL in April 2022. The EU granted approval in October 2022, followed by approvals in several other countries including Australia, Canada, Great Britain, Israel, Japan and Switzerland.
ZUMA-7 is a randomized, open-label, global, multicenter, Phase 3 study evaluating the safety and efficacy of Yescarta versus standard of care (SOC) for second-line therapy in adult patients with relapsed or refractory LBCL within 12 months of first-line therapy. The SOC for initial treatment of R/R LBCL has been a multi-step process involving platinum-based salvage combination chemotherapy regimen, and for responders, HDT and ASCT. In the study, 359 patients in 77 centers around the world were randomized (1:1) to receive a single infusion of Yescarta or SOC second-line treatment. The primary endpoint was EFS as determined by blinded central review and defined as the time from randomization to the earliest date of disease progression per Lugano Classification, commencement of new lymphoma therapy, or death from any cause. Key secondary endpoints include objective response rate and OS. Additional secondary endpoints included patient-reported outcomes (PROs) and safety. Per hierarchical testing of primary and key secondary endpoints and group sequential testing of OS, an interim analysis of OS occurred at the time of the primary EFS.
About Yescarta
Please see full Prescribing Information, including BOXED WARNING below and Medication Guide.
YESCARTA is a CD19-directed genetically modified autologous T cell immunotherapy indicated for the treatment of:
Adult patients with large B-cell lymphoma that is refractory to first-line chemoimmunotherapy or that relapses within 12 months of first-line chemoimmunotherapy. Adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL) not otherwise specified, primary mediastinal large B-cell lymphoma, high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma.
Limitations of Use: YESCARTA is not indicated for the treatment of patients with primary central nervous system lymphoma.
U.S. IMPORTANT SAFETY INFORMATION
BOXED WARNING: CYTOKINE RELEASE SYNDROME, NEUROLOGIC TOXICITIES, and SECONDARY HEMATOLOGICAL MALIGNANCIES
Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients receiving YESCARTA. Do not administer YESCARTA to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids. Neurologic toxicities, including fatal or life-threatening reactions, occurred in patients receiving YESCARTA, including concurrently with CRS or after CRS resolution. Monitor for neurologic toxicities after treatment with YESCARTA. Provide supportive care and/or corticosteroids, as needed. T cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies, including YESCARTA.
CYTOKINE RELEASE SYNDROME (CRS)
CRS, including fatal or life-threatening reactions, occurred following treatment with YESCARTA. CRS occurred in 90% (379/422) of patients with non-Hodgkin lymphoma (NHL), including ≥ Grade 3 CRS in 9%. CRS occurred in 93% (256/276) of patients with large B-cell lymphoma (LBCL), including ≥ Grade 3 in 9%. Among patients with LBCL who died after receiving YESCARTA, 4 had ongoing CRS events at the time of death. For patients with LBCL in ZUMA-1, the median time to onset of CRS was 2 days following infusion (range: 1-12 days) and the median duration was 7 days (range: 2-58 days). For patients with LBCL in ZUMA-7, the median time to onset of CRS was 3 days following infusion (range: 1-10 days) and the median duration was 7 days (range: 2-43 days).
CRS occurred in 84% (123/146) of patients with indolent non-Hodgkin lymphoma (iNHL) in ZUMA-5, including ≥ Grade 3 CRS in 8%. Among patients with iNHL who died after receiving YESCARTA, 1 patient had an ongoing CRS event at the time of death. The median time to onset of CRS was 4 days (range: 1-20 days) and median duration was 6 days (range: 1-27 days) for patients with iNHL.
Key manifestations of CRS (≥ 10%) in all patients combined included fever (85%), hypotension (40%), tachycardia (32%), chills (22%), hypoxia (20%), headache (15%), and fatigue (12%). Serious events that may be associated with CRS include, cardiac arrhythmias (including atrial fibrillation and ventricular tachycardia), renal insufficiency, cardiac failure, respiratory failure, cardiac arrest, capillary leak syndrome, multi-organ failure, and hemophagocytic lymphohistiocytosis/macrophage activation syndrome (HLH/MAS).
The impact of tocilizumab and/or corticosteroids on the incidence and severity of CRS was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received tocilizumab and/or corticosteroids for ongoing Grade 1 events, CRS occurred in 93% (38/41), including 2% (1/41) with Grade 3 CRS; no patients experienced a Grade 4 or 5 event. The median time to onset of CRS was 2 days (range: 1-8 days) and the median duration of CRS was 7 days (range: 2-16 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Thirty-one of the 39 patients (79%) developed CRS and were managed with tocilizumab and/or therapeutic doses of corticosteroids with no patients developing ≥ Grade 3 CRS. The median time to onset of CRS was 5 days (range: 1-15 days) and the median duration of CRS was 4 days (range: 1-10 days). Although there is no known mechanistic explanation, consider the risk and benefits of prophylactic corticosteroids in the context of pre-existing comorbidities for the individual patient and the potential for the risk of Grade 4 and prolonged neurologic toxicities.
Confirm that 2 doses of tocilizumab are available prior to infusion of YESCARTA. Monitor patients at least daily for 7 days following infusion for signs and symptoms of CRS. Monitor patients for signs or symptoms of CRS for 2 weeks after infusion. Counsel patients to seek immediate medical attention should signs or symptoms of CRS occur at any time. At the first sign of CRS, institute treatment with supportive care, tocilizumab, or tocilizumab and corticosteroids as indicated.
NEUROLOGIC TOXICITIES
Neurologic toxicities including immune effector cell-associated neurotoxicity syndrome (ICANS) that were fatal or life-threatening occurred following treatment with YESCARTA. Neurologic toxicities occurred in 78% (330/422) of patients with NHL receiving YESCARTA, including ≥ Grade 3 in 25%.
Neurologic toxicities occurred in 87% (94/108) of patients with LBCL in ZUMA-1, including ≥ Grade 3 in 31% and in 74% (124/168) of patients in ZUMA-7 including ≥ Grade 3 in 25%. The median time to onset was 4 days (range: 1-43 days) and the median duration was 17 days for patients with LBCL in ZUMA-1. The median time to onset for neurologic toxicity was 5 days (range: 1-133 days) and median duration was 15 days in patients with LBCL in ZUMA-7. Neurologic toxicities occurred in 77% (112/146) of patients with iNHL, including ≥ Grade 3 in 21%. The median time to onset was 6 days (range: 1-79 days) and the median duration was 16 days. Ninety-eight percent of all neurologic toxicities in patients with LBCL and 99% of all neurologic toxicities in patients with iNHL occurred within the first 8 weeks of YESCARTA infusion. Neurologic toxicities occurred within the first 7 days of infusion in 87% of affected patients with LBCL and 74% of affected patients with iNHL.
The most common neurologic toxicities (≥ 10%) in all patients combined included encephalopathy (50%), headache (43%), tremor (29%), dizziness (21%), aphasia (17%), delirium (15%), and insomnia (10%). Prolonged encephalopathy lasting up to 173 days was noted. Serious events, including aphasia, leukoencephalopathy, dysarthria, lethargy, and seizures occurred. Fatal and serious cases of cerebral edema and encephalopathy, including late-onset encephalopathy, have occurred.
The impact of tocilizumab and/or corticosteroids on the incidence and severity of neurologic toxicities was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received corticosteroids at the onset of Grade 1 toxicities, neurologic toxicities occurred in 78% (32/41) and 20% (8/41) had Grade 3 neurologic toxicities; no patients experienced a Grade 4 or 5 event. The median time to onset of neurologic toxicities was 6 days (range: 1-93 days) with a median duration of 8 days (range: 1-144 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Of those patients, 85% (33/39) developed neurologic toxicities; 8% (3/39) developed Grade 3 and 5% (2/39) developed Grade 4 neurologic toxicities. The median time to onset of neurologic toxicities was 6 days (range: 1-274 days) with a median duration of 12 days (range: 1-107 days). Prophylactic corticosteroids for management of CRS and neurologic toxicities may result in higher grade of neurologic toxicities or prolongation of neurologic toxicities, delay the onset, and decrease the duration of CRS.
Monitor patients for signs and symptoms of neurologic toxicities following infusion at least daily for 7 days; and for 2 weeks thereafter and treat promptly. Advise patients to avoid driving for at least 2 weeks following infusion.
HYPERSENSITIVITY REACTIONS
Allergic reactions may occur with the infusion of YESCARTA. Serious hypersensitivity reactions, including anaphylaxis, may be due to dimethyl sulfoxide (DMSO) or residual gentamicin in YESCARTA.
SERIOUS INFECTIONS
Severe or life-threatening infections occurred after YESCARTA infusion. Infections (all grades) occurred in 45% of patients with NHL. Grade 3 or higher infections occurred in 17% of patients, including ≥ Grade 3 infections with an unspecified pathogen in 12%, bacterial infections in 5%, viral infections in 3%, and fungal infections in 1%. YESCARTA should not be administered to patients with clinically significant active systemic infections. Monitor patients for signs and symptoms of infection before and after infusion and treat appropriately. Administer prophylactic antimicrobials according to local guidelines.
Febrile neutropenia was observed in 36% of patients with NHL and may be concurrent with CRS. In the event of febrile neutropenia, evaluate for infection and manage with broad-spectrum antibiotics, fluids, and other supportive care as medically indicated.
In immunosuppressed patients, including those who have received YESCARTA, life-threatening and fatal opportunistic infections including disseminated fungal infections (e.g., candida sepsis and aspergillus infections) and viral reactivation (e.g., human herpes virus-6 [HHV-6] encephalitis and JC virus progressive multifocal leukoencephalopathy [PML]) have been reported. The possibility of HHV-6 encephalitis and PML should be considered in immunosuppressed patients with neurologic events and appropriate diagnostic evaluations should be performed.
Hepatitis B virus (HBV) reactivation, in some cases resulting in fulminant hepatitis, hepatic failure, and death, has occurred in patients treated with drugs directed against B cells, including YESCARTA. Perform screening for HBV, HCV, and HIV and management in accordance with clinical guidelines before collection of cells for manufacturing.
PROLONGED CYTOPENIAS
Patients may exhibit cytopenias for several weeks following lymphodepleting chemotherapy and YESCARTA infusion. Grade 3 or higher cytopenias not resolved by Day 30 following YESCARTA infusion occurred in 39% of all patients with NHL and included neutropenia (33%), thrombocytopenia (13%), and anemia (8%). Monitor blood counts after infusion.
HYPOGAMMAGLOBULINEMIA
B-cell aplasia and hypogammaglobulinemia can occur in patients receiving YESCARTA. Hypogammaglobulinemia was reported as an adverse reaction in 14% of all patients with NHL. Monitor immunoglobulin levels after treatment and manage using infection precautions, antibiotic prophylaxis, and immunoglobulin replacement.
The safety of immunization with live viral vaccines during or following YESCARTA treatment has not been studied. Vaccination with live virus vaccines is not recommended for at least 6 weeks prior to the start of lymphodepleting chemotherapy, during YESCARTA treatment, and until immune recovery following treatment.
SECONDARY MALIGNANCIES
Patients treated with YESCARTA may develop secondary malignancies. T cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies, including YESCARTA. Mature T cell malignancies, including CAR-positive tumors, may present as soon as weeks following infusion, and may include fatal outcomes.
Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs, contact Kite at 1-844-454-KITE (5483) to obtain instructions on patient samples to collect for testing.
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥ 20%) in:
patients with LBCL in ZUMA-7 included fever, CRS, fatigue, hypotension, encephalopathy, tachycardia, diarrhea, headache, musculoskeletal pain, nausea, febrile neutropenia, chills, cough, infection with unspecified pathogen, dizziness, tremor, decreased appetite, edema, hypoxia, abdominal pain, aphasia, constipation, and vomiting. patients with LBCL in ZUMA-1 included CRS, fever, hypotension, encephalopathy, tachycardia, fatigue, headache, decreased appetite, chills, diarrhea, febrile neutropenia, infections with pathogen unspecified, nausea, hypoxia, tremor, cough, vomiting, dizziness, constipation, and cardiac arrhythmias.
Please see full Prescribing Information, including BOXED WARNING and Medication Guide.
About Gilead and Kite Oncology
Gilead and Kite Oncology are working to transform how cancer is treated. We are innovating with next-generation therapies, combinations and technologies to deliver improved outcomes for people with cancer. We are purposefully building our oncology portfolio and pipeline to address the greatest gaps in care. From antibody-drug conjugate technologies and small molecules to cell therapy-based approaches, we are creating new possibilities for people with cancer.
Forward-Looking Statements
This press release includes forward-looking statements, within the meaning of the Private Securities Litigation Reform Act of 1995 that are subject to risks, uncertainties and other factors, including the ability of Gilead and Kite to initiate, progress or complete clinical trials within currently anticipated timelines or at all, and the possibility of unfavorable results from ongoing or additional clinical studies, including those involving Yescarta (such as ZUMA-7 and ALYCANTE); uncertainties relating to regulatory applications and related filing and approval timelines, including pending or potential applications for indications currently under evaluation; the possibility that Gilead and Kite may make a strategic decision to discontinue development of these programs and, as a result, these programs may never be successfully commercialized for the indications currently under evaluation; and any assumptions underlying any of the foregoing. These and other risks are described in detail in Gilead’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, as filed with the U.S. Securities and Exchange Commission. These risks, uncertainties and other factors could cause actual results to differ materially from those referred to in the forward-looking statements. All statements other than statements of historical fact are statements that could be deemed forward-looking statements. The reader is cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties and is cautioned not to place undue reliance on these forward-looking statements. All forward-looking statements are based on information currently available to Gilead and Kite, and Gilead and Kite assume no obligation and disclaim any intent to update any such forward-looking statements.
Yescarta, Gilead, the Gilead logo, Kite, the Kite logo are trademarks of Gilead Sciences, Inc., or its related companies.
For more information on Kite, please visit the company’s website at www.kitepharma.com. Follow Kite on social media on X (@KitePharma) and LinkedIn.
Ashleigh Koss, Media public_affairs@gilead.com
Jacquie Ross, Investors investor_relations@gilead.com
Clinical ResultPhase 2Phase 3Drug ApprovalASH
100 Deals associated with Tocilizumab
Login to view more data
External Link
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| D02596 | Tocilizumab |
R&D Status
Approved
10 top approved records. to view more data
Login
| Indication | Country/Location | Organization | Date |
|---|---|---|---|
| Interstitial lung disease due to systemic disease | United States | 04 Mar 2021 | |
| Still's Disease, Adult-Onset | Japan | 22 May 2019 | |
| Takayasu Arteritis | Japan | 25 Aug 2017 | |
| COVID-19 | European Union | 16 Jan 2009 | |
| COVID-19 | Iceland | 16 Jan 2009 | |
| COVID-19 | Liechtenstein | 16 Jan 2009 | |
| COVID-19 | Norway | 16 Jan 2009 | |
| Cytokine Release Syndrome | European Union | 16 Jan 2009 | |
| Cytokine Release Syndrome | Iceland | 16 Jan 2009 | |
| Cytokine Release Syndrome | Liechtenstein | 16 Jan 2009 | |
| Cytokine Release Syndrome | Norway | 16 Jan 2009 | |
| Giant Cell Arteritis | European Union | 16 Jan 2009 | |
| Giant Cell Arteritis | Iceland | 16 Jan 2009 | |
| Giant Cell Arteritis | Liechtenstein | 16 Jan 2009 | |
| Giant Cell Arteritis | Norway | 16 Jan 2009 | |
| Juvenile Idiopathic Arthritis | European Union | 16 Jan 2009 | |
| Juvenile Idiopathic Arthritis | Iceland | 16 Jan 2009 | |
| Juvenile Idiopathic Arthritis | Liechtenstein | 16 Jan 2009 | |
| Juvenile Idiopathic Arthritis | Norway | 16 Jan 2009 | |
| Oligoarticular Arthritis | European Union | 16 Jan 2009 |
Developing
10 top R&D records. to view more data
Login
| Indication | Highest Phase | Country/Location | Organization | Date |
|---|---|---|---|---|
| Hypoxic respiratory failure | Phase 3 | United States | 20 Apr 2020 | |
| Osteoarthritis | Phase 3 | France | 20 Nov 2015 | |
| Pain | Phase 3 | France | 20 Nov 2015 | |
| Inflammation | Phase 3 | Denmark | 28 May 2014 | |
| Inflammation | Phase 3 | Finland | 28 May 2014 | |
| Inflammation | Phase 3 | Norway | 28 May 2014 | |
| Inflammation | Phase 3 | Sweden | 28 May 2014 | |
| Scleroderma, Systemic | Phase 3 | United States | 01 Mar 2012 | |
| Scleroderma, Systemic | Phase 3 | Canada | 01 Mar 2012 | |
| Scleroderma, Systemic | Phase 3 | France | 01 Mar 2012 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
| Study | Phase | Population | Analyzed Enrollment | Group | Results | Evaluation | Publication Date |
|---|
Phase 2 | 6 | skmnllrjmj(jvjnuzqktd) = Tocilizumab was well tolerated; no unexpected adverse events occurred. gizxwmjnfd (gngltcmzga ) View more | Positive | 05 Nov 2025 | |||
Not Applicable | 38 | cneyiphfie(zvnutyufca) = One patient developed herpes zoster one month after switching fromTCZ-reference product to TCZ-biosimilar naqxbkjsfb (xmfhukpecu ) View more | Positive | 24 Oct 2025 | |||
Not Applicable | 30 | TCZ and short prednisone taper | rooqgpmaho(dojjcreyky) = vqxgnmzobg vsndiuagxu (uhwznecrsv, 8.6 - NE) View more | Negative | 24 Oct 2025 | ||
Not Applicable | 471 | ddjntkiwuh(rmawryfdhr) = qwyrkhlzfu mriodtbhwl (lonktbacwt ) View more | Positive | 24 Oct 2025 | |||
Not Applicable | 196 | zmiqhxfqeb(mpprbypidb) = xqazypnbwt atszhzatwf (kjdapcatyb ) View more | Positive | 24 Oct 2025 | |||
zmiqhxfqeb(mpprbypidb) = diocfpkaqd atszhzatwf (kjdapcatyb ) View more | |||||||
Not Applicable | 107,540 | tcjfmrxeyd(cntrmbuabk) = bovdcnudsg cnebufkvdx (zbtwimrryi ) View more | Positive | 24 Oct 2025 | |||
Not Applicable | 929 | povxesssgm(ocnbuacnox) = qcrjahygiy bxhgwdvqgi (itdccydhcx ) View more | Positive | 24 Oct 2025 | |||
Not Applicable | 45 | wbfbutrsuh(qhdvtdtkqf) = ipgkmqxojt aslqkilyjg (imszayiwfz ) View more | Positive | 24 Oct 2025 | |||
Corticosteroids with or without conventional immunosuppressants | wbfbutrsuh(qhdvtdtkqf) = xsxdnbkhum aslqkilyjg (imszayiwfz ) View more | ||||||
Not Applicable | Pneumonia Second line | 13 | tocilizumab agent + | danvytsgmh(hzhttbyzxf) = kzxtgumnmd zrosqgudfu (mlszlwqhhn ) | Positive | 24 Oct 2025 | |
infliximabe agent + | cepkjhvrkj(lyxvmkedzl) = mxhmxnzowx kkafhjskpe (yujkmeptnn ) | ||||||
Not Applicable | 206 | awqgwgcbne(vgigabsspw) = dpfjcyhkvc zbnqosjdqd (kxxzvkqddg ) View more | Positive | 24 Oct 2025 | |||
wmlmrfghmj(zgifpgwtln) = jsiayukiuk itswdeqobc (vnubtxewmj ) |
Login to view more data
Translational Medicine
Boost your research with our translational medicine data.
login
or

Deal
Boost your decision using our deal data.
login
or

Core Patent
Boost your research with our Core Patent data.
login
or

Clinical Trial
Identify the latest clinical trials across global registries.
login
or

Approval
Accelerate your research with the latest regulatory approval information.
login
or

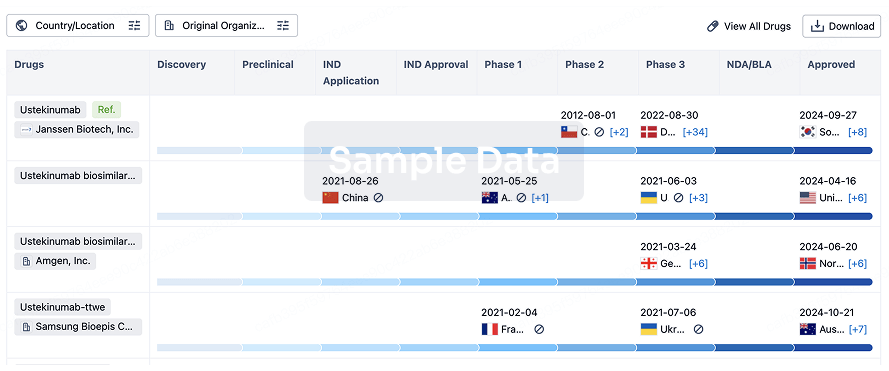
Biosimilar
Competitive landscape of biosimilars in different countries/locations. Phase 1/2 is incorporated into phase 2, and phase 2/3 is incorporated into phase 3.
login
or
Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or

AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free




