Qilu Pharmaceutical's Three Clinical Studies on Cancer Immunotherapy Presented at ASCO 2024
07 Jun 2024
JINAN, China, June 6, 2024 /PRNewswire/ -- The 2024 American Society of Clinical Oncology (ASCO) Annual Meeting convened from May 31 to June 4, 2024 in Chicago, USA, adopting a hybrid format. Among the presented works, three clinical studies from Qilu Pharmaceutical were selected for poster sessions. These studies introduced novel immunotherapeutic agents, specifically QLF31907, a bispecific antibody targeting PD-L1/4-1BB; iparomlimab and tuvonralimab, a MabPair product targeting PD-1/CTLA-4; and iparomlimab, a monoclonal antibody targeting PD-1. The research involved treatments for advanced solid tumors and lymphoma, nasopharyngeal carcinoma, as well as solid tumors characterized by either DNA mismatch repair (dMMR) deficiency or high microsatellite instability (MSI-H).
Continue Reading
Preview
Source: PRNewswire
View PDF
Preview
Source: PRNewswire
View PDF
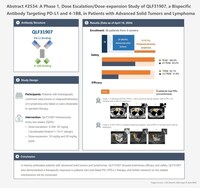
QLF31907, developed by Qilu Pharmaceutical, combines two mechanisms: blockade of PD-L1 to restore T-cell receptor (TCR) signaling, while binding to 4-1BB to provide costimulatory signals essential for T-cell activation. This dual-action mechanism fosters T-cell proliferation and activation, enhancing the anti-tumor immune response. The study, led by Professor Tongyu Lin from Sichuan Cancer HospitalCancer Hospital, is a phase I dose-escalation/expansion trial focusing on QLF31907 (PD-1/4-1BB dual antibody) in patients with advanced solid tumors and lymphoma (Abstract No. 2534).
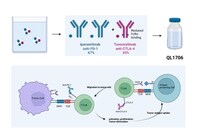
Iparomlimab and tuvonralimab, representing an innovative immunotherapeutic approach, comprises a unique combination of PD-1 antibody IgG4 and CTLA-4 antibody IgG1 in a predetermined ratio, with the latter engineered to have a reduced half-life. This allows sustaining normal PD-1 antibody levels in vivo while minimizing CTLA-4 antibodyCTLA-4 antibody exposure and is a potential therapy with lower toxicity and improved tolerability. In the phase 1 study, iparomlimab and tuvonralimab exhibited encouraging anti-tumor activity in patients with advanced nasopharyngeal carcinoma. The ongoing multicenter, single-arm, phase II trial (DUBHE-N-302, Abstract No. 6026) led by Professor Yan Huang from Sun Yat-sen University Cancer CenterCancer Center, explores iparomlimab and tuvonralimab in combination with gemcitabine and cisplatin for the first-line treatment of recurrent or metastatic nasopharyngeal carcinoma.
In this study, 29 patients were enrolled, with 7 (24%) having a baseline ECOG PS score of 1. After a median follow-up duration of 15.5 months, 18 (62%) patients reported grade 3-4 treatment-related adverse events (TRAEs), and the most common TRAE was neutrophil count decreased (41%). A total of 28 patients had at least 1 post-baseline tumor assessment. The objective response rate (ORR) was 82.1% (95% CI: 63.1%-93.9%). Median progression-free survival (mPFS) was 12.5 months (95% CI: 5.7-NE), and in 13 patients with high PD-L1 expression (CPS≥50), mPFS reached 16.2 months (95% CI: 9.9-NE). Median overall survival was not reached. The findings suggested that iparomlimab and tuvonralimab plus chemotherapy was well-tolerated and demonstrated promising anti-tumor activity in the first-line treatment of recurrent/metastatic nasopharyngeal carcinoma.
Iparomlimab is a highly selective humanized monoclonal antibody targeting PD-1. Updated results from a pivotal single-arm, phase II clinical study (Abstract No. 3578), led by Professor Weijian Guo of Fudan University Shanghai Cancer CenterCancer Center and Professor Feng Bi of West China Hospital of Sichuan University, were presented at ASCO 2024. The updated results with 1-year follow-up after enrollment of last patient revealed that iparomlimab monotherapy showed promising efficacy in this patient population. Among those with solid tumors who had failed standard treatment, the ORR, assessed by the Independent Radiology Review Committee (IRRC), reached 50.0%, higher than the prespecified primary endpoint. The ORR was 57.9% in patients with colorectal cancer. At the time of data cutoff, the median duration of response (DOR), median progression-free survival (mPFS), and median overall survival had not yet been reached. These findings underscore the robust and durable efficacy of iparomlimab in patients with advanced dMMR/MSI-H solid tumors who have failed in standard treatment. Notably, prolonged treatment with iparomlimab remained safe and well-tolerated, without any new safety concerns emerging.
Graphical Abstract QLF31907: https://mma.prnewswire.com/media/2432113/Graphical_Abstract_QLF31907_FINAL_EN.pdf
QL1706 Graph: https://mma.prnewswire.com/media/2432114/QL1706_graph.pdf
SOURCE Qilu Pharmaceutical Co., Ltd
For more details,please visit the original website
The content of the article does not represent any opinions of Synapse and its affiliated companies. If there is any copyright infringement or error, please contact us, and we will deal with it within 24 hours.
Chat with Hiro
Hot reports
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.



