Deep Scientific Insights on Cefazolin Sodium's R&D Progress, Mechanism of Action, and Drug Target
Cefazolin Sodium's R&D Progress
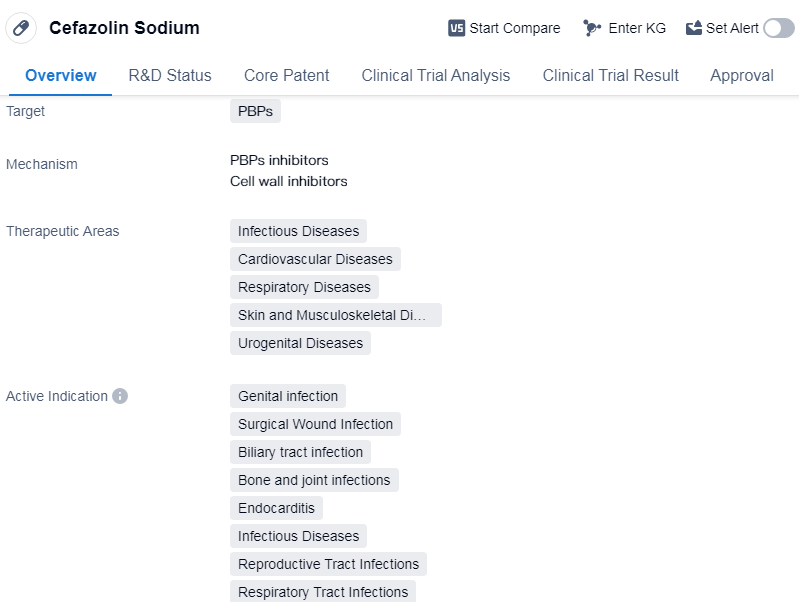
Cefazolin Sodium is a small molecule drug that belongs to the class of antibiotics known as cephalosporins. It primarily targets penicillin-binding proteins (PBPs) to exert its therapeutic effects. The drug has been approved for use in various diseases, including infectious diseases, cardiovascular diseases, respiratory diseases, skin and musculoskeletal diseases, and urogenital diseases.
In terms of active indications, Cefazolin Sodium is indicated for the treatment of genital infections, surgical wound infections, biliary tract infections, bone and joint infections, endocarditis, reproductive tract infections, respiratory tract infections, sepsis, skin and skin structure infections, urinary tract infections, and bacterial infections.
The drug was originally developed by GSK Plc, a renowned pharmaceutical company. It received first approval in the United States in October 1973, making it a well-established and widely used medication. Cefazolin Sodium has also obtained approvals in other countries, indicating its global recognition and acceptance.
With its broad spectrum of activity against various bacteria, Cefazolin Sodium has proven to be effective in treating a wide range of infections. Its approval in multiple therapeutic areas highlights its versatility and potential to address different medical conditions. The drug's mechanism of action, targeting PBPs, allows it to inhibit bacterial cell wall synthesis, leading to the disruption of bacterial growth and ultimately their elimination.
As a small molecule drug, Cefazolin Sodium offers advantages such as ease of formulation, administration, and manufacturing. These characteristics contribute to its widespread availability and accessibility in the market. The drug's long history of use since its first approval demonstrates its safety and efficacy profile.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
Mechanism of Action for Cefazolin Sodium: PBPs Inhibitors and Cell Wall Inhibitors
From a biomedical perspective, PBPs inhibitors refer to a class of drugs that target PBPs. PBPs are enzymes found in the cell wall of bacteria and are responsible for the synthesis of peptidoglycan, a crucial component of the bacterial cell wall. By inhibiting PBPs, these drugs disrupt the formation of peptidoglycan, leading to weakened cell walls and ultimately bacterial cell death. This mechanism of action makes PBPs inhibitors effective against bacterial infections.
On the other hand, cell wall inhibitors are a broader category of drugs that target various components involved in the synthesis or integrity of the cell wall. In the context of antimicrobial therapy, cell wall inhibitors primarily refer to drugs that interfere with the formation or stability of bacterial cell walls. These drugs can have different mechanisms of action, such as inhibiting enzymes involved in cell wall synthesis or disrupting the integrity of the cell wall structure.
In summary, PBPs inhibitors specifically target PBPs to disrupt bacterial cell wall synthesis, while cell wall inhibitors encompass a wider range of drugs that interfere with the formation or stability of bacterial cell walls.
Drug Target R&D Trends for Cefazolin Sodium
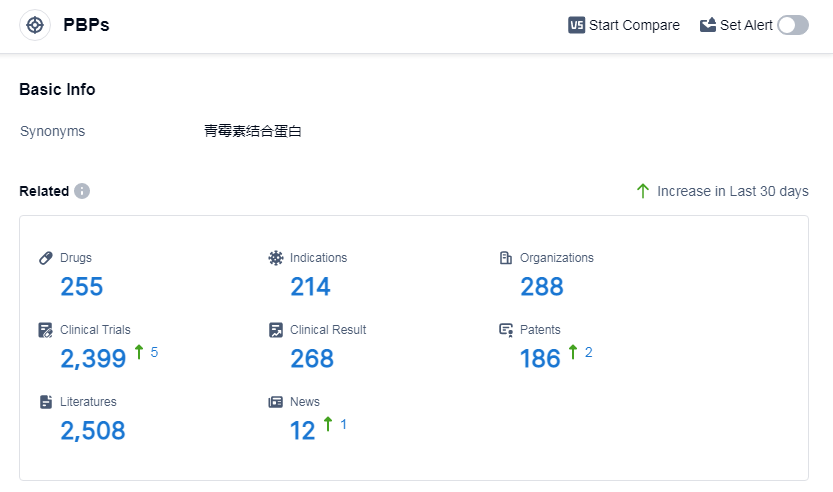
According to Patsnap Synapse, as of 14 Sep 2023, there are a total of 255 PBPs drugs worldwide, from 288 organizations, covering 214 indications, and conducting 2399 clinical trials.
The analysis of the target PBPs in the pharmaceutical industry reveals that Pfizer Inc., Meiji Holdings Co., Ltd., Takeda Pharmaceutical Co., Ltd., Shionogi & Co., Ltd., and Merck & Co., Inc. are the companies growing fastest under this target. Bacterial infections, infectious diseases, and urinary tract infections are the most common indications for drugs under the target PBPs. Small molecule drugs and synthetic peptides are the drug types progressing most rapidly. China, Japan, and the United States are the countries/locations developing fastest under the target PBPs. This analysis provides insights into the current competitive landscape and future development of target PBPs in the pharmaceutical industry.
👇Please click on the picture link below for free registration or log in directly if you have a freemium account, you can browse the latest research progress on drugs, indications, organizations, clinical trials, clinical results, and drug patents related to this target
Conclusion
In conclusion, Cefazolin Sodium is a well-established small molecule drug developed by GSK Plc. It has been approved for use in various therapeutic areas and is indicated for the treatment of numerous bacterial infections. With its broad spectrum of activity and long history of use, Cefazolin Sodium continues to be a valuable option in the pharmaceutical industry for combating infectious diseases and other related conditions.