Is Lonafarnib approved by the FDA?
Yes, Lonafarnib (Zokinvy) is an FDA-approved medication for treating Hutchinson-Gilford Progeria Syndrome and certain processing-deficient Progeroid Laminopathies. Approved on November 20, 2020, it offers a vital treatment option for managing these rare genetic conditions.
What is Lonafarnib?
Lonafarnib, marketed under the brand name Zokinvy, is an oral medication used to treat specific rare genetic conditions that cause premature and rapid aging. It is indicated for use in adults and children at least 12 months old with a body surface area of at least 0.39 square meters.
How Lonafarnib Works
Lonafarnib works by inhibiting a specific enzyme involved in the abnormal aging process seen in HGPS and certain Progeroid Laminopathies. By blocking this enzyme, Lonafarnib helps to reduce the harmful effects associated with these genetic conditions.
Indications
- Hutchinson-Gilford Progeria Syndrome (HGPS): Lonafarnib is used to lower the risk of death in patients with HGPS.
- Processing-Deficient Progeroid Laminopathies: It is also used to treat certain types of Progeroid Laminopathies that are processing-deficient.
Lonafarnib is not indicated for use in patients with non-HGPS Progeroid Syndromes or processing-proficient Progeroid Laminopathies.
Administration and Dosage
- Dosage Form: Oral capsules (50 mg; 75 mg)
- Administration: Typically taken with morning and evening meals. It should be swallowed with a full glass of water. If unable to swallow the capsule whole, the contents can be mixed with Ora Blend SF, Ora-Plus, orange juice, or applesauce and consumed immediately.
Potential Side Effects
Common Side Effects:
- Electrolyte imbalances (low levels of potassium, sodium, or calcium)
- Infection
- Increased blood pressure
- Stomach pain, nausea, vomiting, diarrhea
- Decreased appetite, weight loss
- Headache
- Fatigue
- Muscle or joint pain
- Cold symptoms (stuffy nose, sneezing, sore throat, cough)
- Abnormal lab tests
Serious Side Effects:
- New or worsening vision problems
- Severe ongoing nausea, vomiting, diarrhea
- Loss of appetite causing weight loss
- High blood pressure
- Kidney problems
- High potassium level
- Low potassium level
- Low sodium level
- Low calcium level
- Signs of infection
Patients should seek emergency medical help if they experience signs of an allergic reaction such as hives, difficulty breathing, or swelling of the face, lips, tongue, or throat.
Warnings and Precautions
- Drug Interactions: Many drugs can interact with Lonafarnib, and some should not be used together. Inform your doctor about all your current medications.
- Pregnancy and Fertility: Lonafarnib may harm an unborn baby and affect fertility. Effective birth control should be used to prevent pregnancy.
- Breastfeeding: It may not be safe to breastfeed while using this medication. Consult your doctor about any risks.
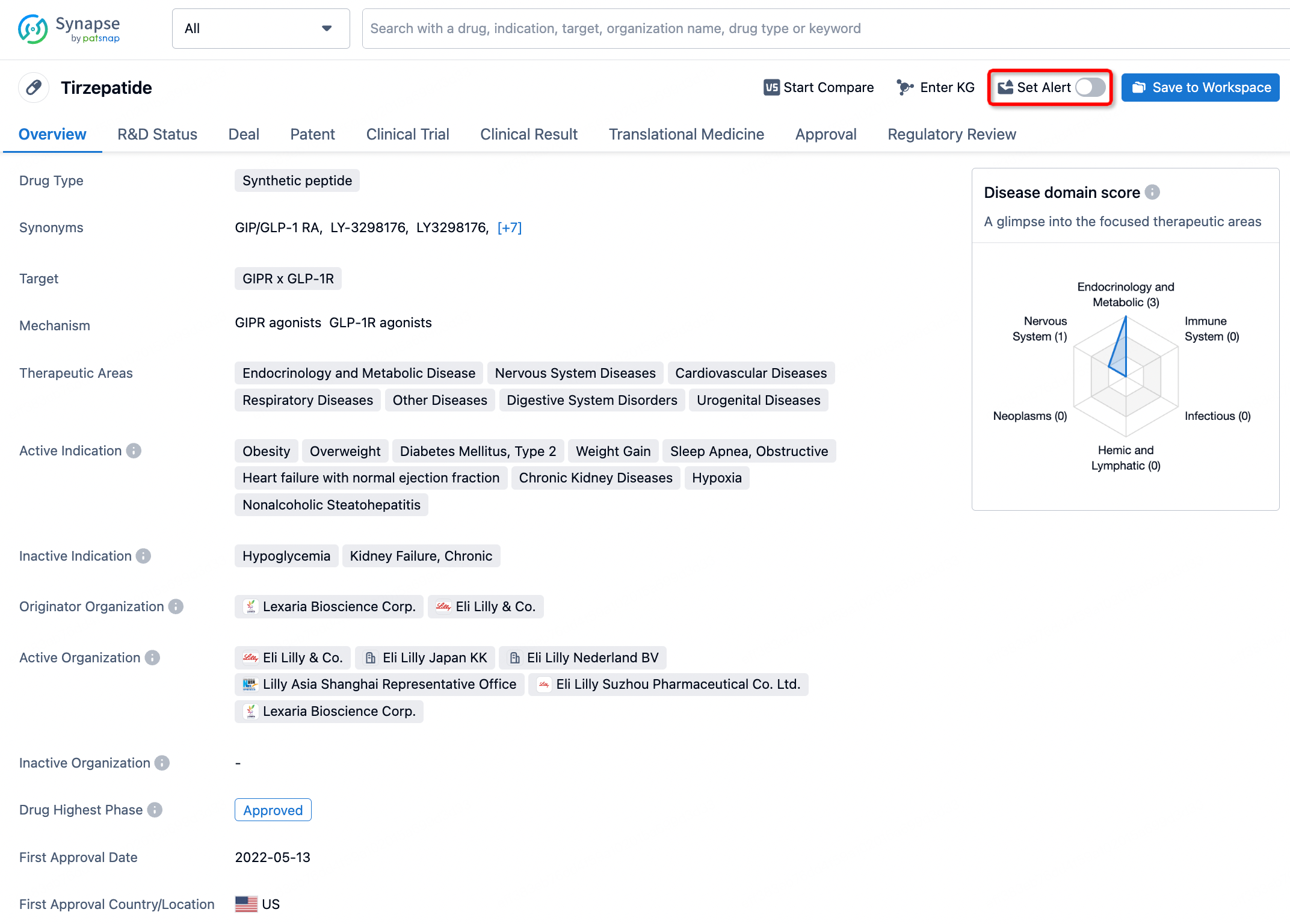
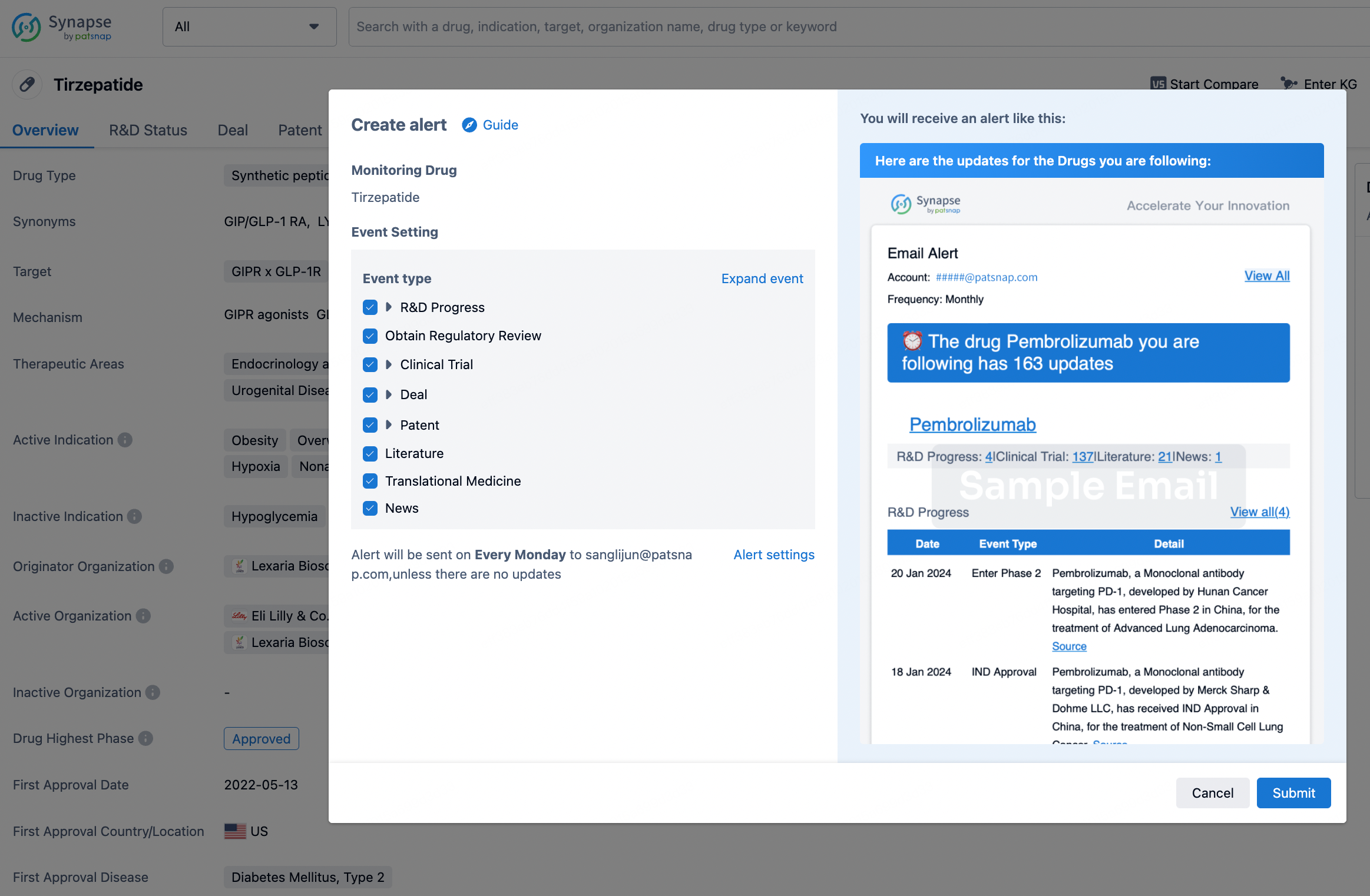
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!