Is Tebentafusp approved by the FDA?
Tebentafusp received FDA approval on January 25, 2022. It is a prescription medication used to treat adults with uveal melanoma, a type of cancer in the eye that cannot be removed by surgery or has metastasized.
How Tebentafusp Works
Tebentafusp works by targeting a specific protein on the surface of cancer cells and activating the immune system to attack these cells. It is administered intravenously, typically once a week, with the initial doses requiring extended observation due to potential severe side effects.
Usage and Dosage
Tebentafusp is given as an intravenous solution, with the initial doses carefully monitored:
- Day 1: 20 mcg IV
- Day 8: 30 mcg IV
- Day 15: 68 mcg IV
- Thereafter: 68 mcg IV weekly
Patients must be confirmed as HLA-A*02:01 positive before starting treatment.
Side Effects
Common side effects of Tebentafusp include:
- Fever, chills
- Trouble breathing
- Confusion
- Severe vomiting or diarrhea
- Fast or irregular heartbeats
- Tiredness
- Nausea, stomach pain
- Rash, itching, dry skin
- Headache
- Swelling
- Abnormal liver function tests
Severe side effects such as cytokine release syndrome (CRS) may occur, characterized by fever, chills, trouble breathing, and severe gastrointestinal symptoms. Immediate medical attention is necessary if these symptoms develop.
Warnings and Precautions
Patients should inform their healthcare provider if they have a history of liver problems or are pregnant. Tebentafusp may harm an unborn baby, so effective contraception is recommended during treatment and for at least one week after the last dose. Breastfeeding is also not advised during this period.
Conclusion
Tebentafusp (Kimmtrak) is FDA approved for the treatment of uveal melanoma as of January 25, 2022. While it offers a new treatment option for this rare cancer, patients should be aware of the potential side effects and the necessity for regular monitoring during therapy.
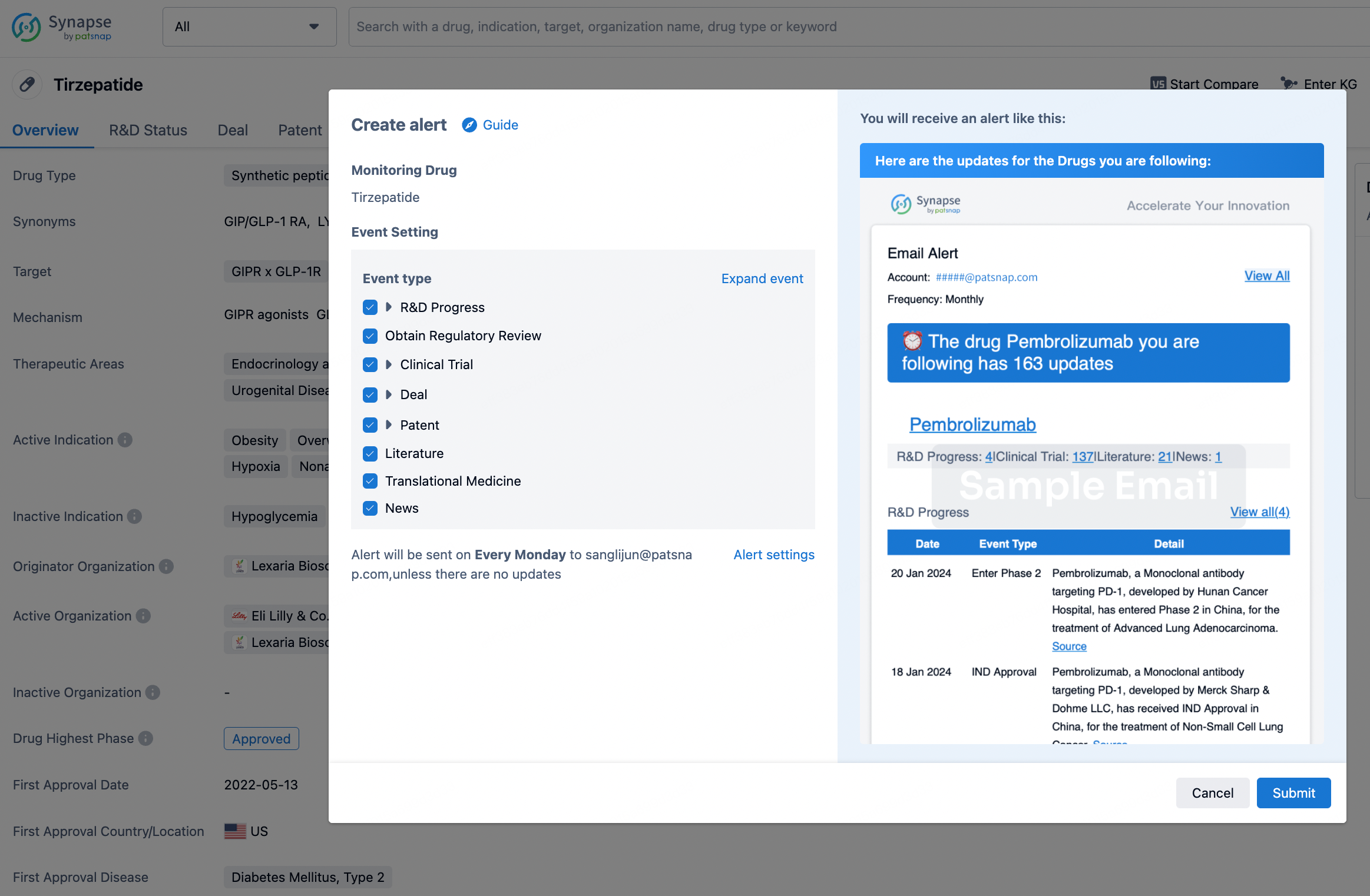
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!