Pentobarbital Sodium: Detailed Review of its Transformative R&D Success
Pentobarbital Sodium's R&D Progress
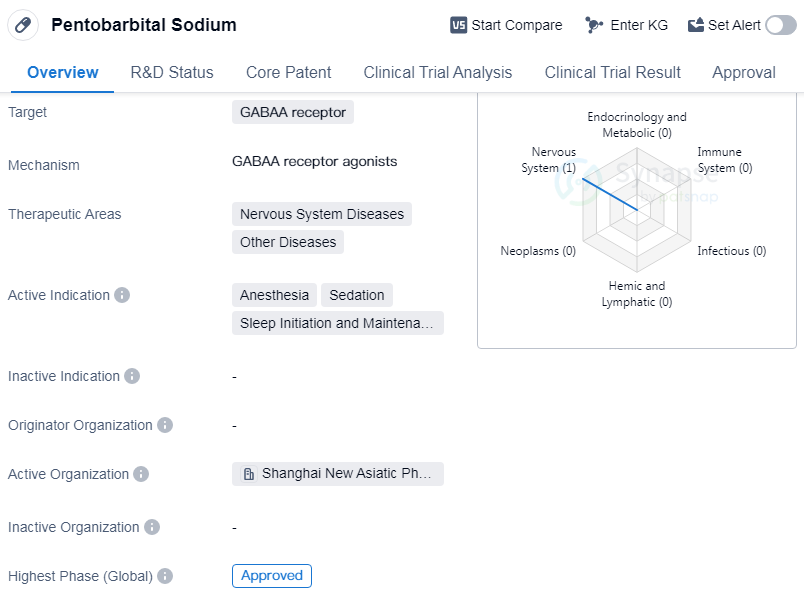
Pentobarbital Sodium is a small molecule drug that acts on the GABAA receptor. It is primarily used in the treatment of nervous system diseases and other diseases. The drug has been approved for use in anesthesia, sedation, and sleep initiation and maintenance disorders.
Pentobarbital Sodium has a long history in the pharmaceutical industry, with its first global approval dating back to 1930. It is considered a well-established drug in the field of biomedicine. The drug's mechanism of action involves targeting the GABAA receptor, which is a neurotransmitter receptor involved in the regulation of neuronal excitability.
In terms of therapeutic areas, Pentobarbital Sodium is primarily used in the treatment of nervous system diseases. This includes epilepsy, anxiety disorders, insomnia, etc. Additionally, it is also used in the treatment of other diseases, although specific details about these diseases are not provided.
The drug has achieved the highest phase of approval globally, indicating its widespread acceptance and use in medical practice. This suggests that Pentobarbital Sodium has undergone rigorous testing and evaluation to ensure its safety and efficacy.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
Mechanism of Action for Pentobarbital Sodium: GABAA receptor agonists
GABAA receptor agonists are a type of drugs that bind to and activate the GABAA receptors in the brain. GABAA receptors are a type of neurotransmitter receptor that respond to the neurotransmitter gamma-aminobutyric acid (GABA), which is the main inhibitory neurotransmitter in the central nervous system.
When GABAA receptor agonists bind to these receptors, they enhance the inhibitory effects of GABA, leading to an overall decrease in neuronal activity. This can result in sedative, anxiolytic (anti-anxiety), anticonvulsant, and muscle relaxant effects.
These drugs are commonly used in the treatment of various conditions such as anxiety disorders, insomnia, epilepsy, and muscle spasms. By enhancing the inhibitory actions of GABA, GABAA receptor agonists help to reduce excessive neuronal activity and promote relaxation and calmness.
It is important to note that GABAA receptor agonists can have sedative effects and may cause drowsiness, impaired coordination, and cognitive impairment. They should be used with caution and under medical supervision to minimize potential side effects and ensure appropriate dosing.
Drug Target R&D Trends for Pentobarbital Sodium
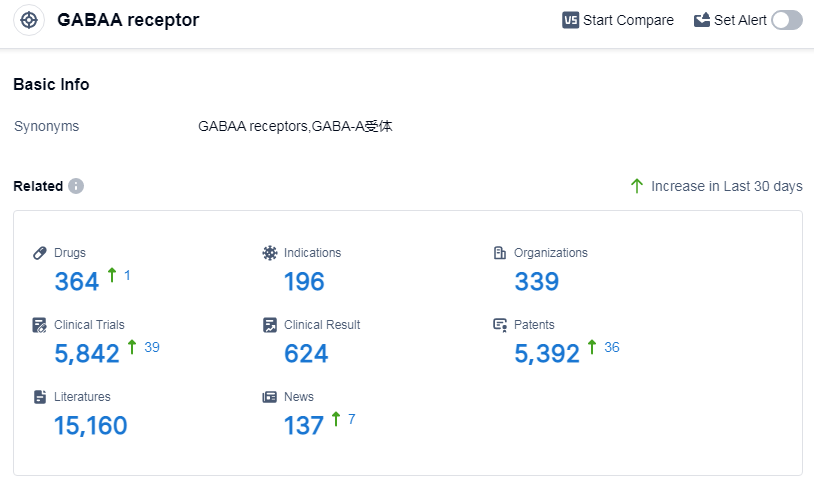
According to Patsnap Synapse, as of 14 Sep 2023, there are a total of 364 GABAA receptor drugs worldwide, from 339 organizations, covering 196 indications, and conducting 5842 clinical trials.
The analysis of the target GABAA receptor reveals a competitive landscape with multiple companies actively involved in research and development. Pfizer Inc., Sumitomo Chemical Co., Ltd., Teva Pharmaceutical Industries Ltd., and Jiangsu Nhwa Pharmaceutical Co., Ltd. are some of the companies with significant R&D progress. The drug's indications include sleep initiation and maintenance disorders, anesthesia, anxiety, epilepsy, seizures, etc. Small molecule drugs dominate the drug types progressing rapidly. Japan, China, and the United States are the leading countries/locations in the development of drugs under the target GABAA receptor. The future development of this target holds potential for advancements in the treatment of various indications and competition among pharmaceutical companies.
👇Please click on the picture link below for free registration or log in directly if you have a freemium account, you can browse the latest research progress on drugs, indications, organizations, clinical trials, clinical results, and drug patents related to this target
Conclusion
Overall, Pentobarbital Sodium is a small molecule drug that targets the GABAA receptor and is primarily used in the treatment of nervous system diseases. It has been approved for use in anesthesia, sedation, and sleep initiation and maintenance disorders. With a long history of use dating back to 1930, Pentobarbital Sodium is considered a well-established drug in the field of biomedicine. Its approval in global markets further highlights its significance and widespread use.