Request Demo
Pharmaceutical Insights
Navigate pharmaceutical trends with our insights on targets, institutional pipelines, clinical advances, and new drugs.

Recent blog posts
Drug Insights
6 min read
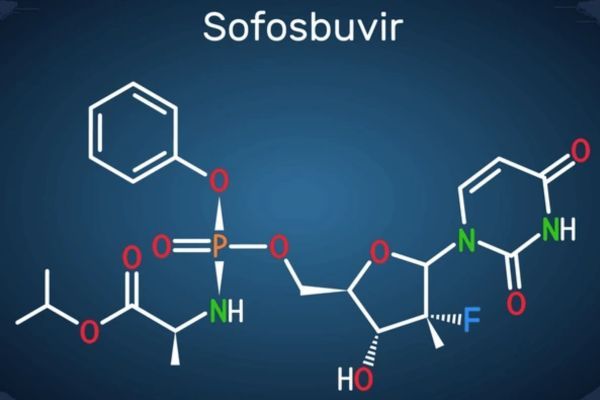
Market Analysis of Sofosbuvir (Sovaldi) in the USA: Patents, Exclusivity, and Entry Opportunities
28 August 2025
Comprehensive market analysis of Sofosbuvir (Sovaldi) in the USA, covering FDA approvals, patents, exclusivity, clinical insights, and strategic recommendations for market entry.
Drug Insights
6 min read
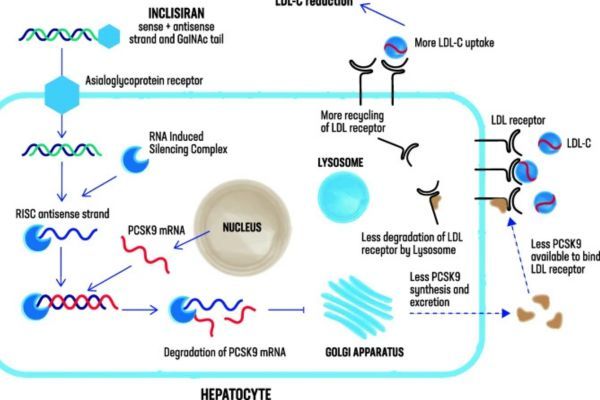
Inclisiran Market Analysis in the USA: Patents, Exclusivity, and Market Entry Outlook
28 August 2025
Comprehensive analysis of Inclisiran in the USA, covering FDA approvals, patents, clinical results, regulatory exclusivity, and strategic insights for innovators and potential generic challengers.
Drug Insights
6 min read
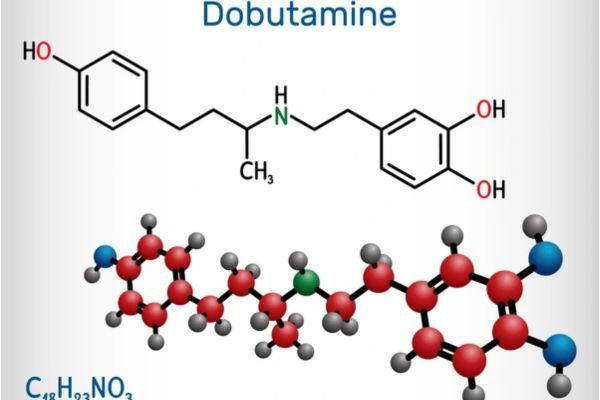
Dobutamine Hydrochloride Market Analysis in the USA: Generic Landscape, Patents, and Clinical Insights
27 August 2025
Comprehensive market analysis of Dobutamine Hydrochloride in the USA, including FDA approvals, patent barriers, clinical data, regulatory risks, and strategic recommendations for generic manufacturers.
Drug Insights
6 min read
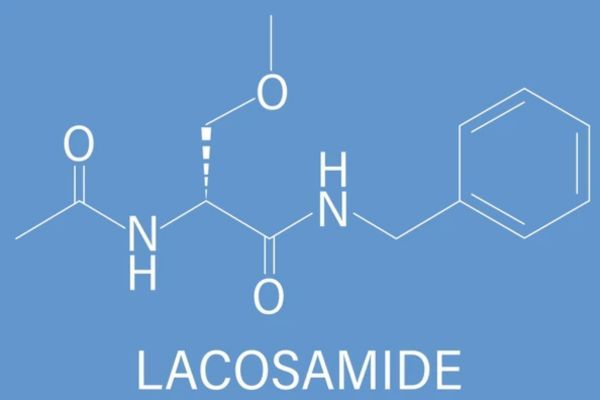
Lacosamide Market Analysis in the USA: Patent Landscape, Clinical Insights, and Strategic Entry
27 August 2025
Explore the US market analysis of Lacosamide, including FDA approvals, patent landscape, clinical results, regulatory risks, and strategic recommendations for generic and innovator companies.
Drug Insights
6 min read
Amisulpride Market Landscape: Patent Barriers, Clinical Insights, and Global Entry Strategies
26 August 2025
Discover the global market landscape of Amisulpride, including patent barriers, clinical insights, regulatory risks, and strategic opportunities in the US, China, and EU markets.
Drug Highlight
12 min read
June 2025 Drug Approvals: Breakthroughs in Gout, Cancer & RSV Prevention
18 July 2025
Explore the latest FDA & China-approved drugs in June 2025, including Genakumab (gout), Taletrectinib (NSCLC), and Clesrovimab (RSV). Key clinical benefits & market impact.
DrugDeal Decode
14 min read
May 2025 Biotech Boom: $12B+ in Deals Reshape Next-Gen Therapies from AI to RNA Editing
18 July 2025
Explore how CSPC ($5B deal), Sanofi, and Eli Lilly invested $12B+ in AI drug discovery, RNA editing & in vivo CAR-T in May 2025.
Drug Highlight
23 min read
May 2025 Drug Approval Wave: From KRAS-Targeted Combinations to Novel ADCs in NSCLC
4 July 2025
In May 2025, the global pharmaceutical landscape witnessed a wave of innovation.
Drug Highlight
20 min read
April 2025 Therapeutic Breakthroughs: From FcRn-Targeted mAbs to COL7 Gene Therapy in DEB
3 July 2025
In April 2025, a series of innovative drugs targeting various disease areas received regulatory approval, ranging from rare diseases.
Hot Spotlight
10 min read
The mRNA Therapeutics Revolution: Market Growth, Mechanism of Action, and Global Development Landscape
20 June 2025
Explore the booming mRNA therapeutics market, projected to exceed USD 100 billion by 2028. Learn how mRNA drugs work, their key advantages, and the global leaders driving innovation in vaccines and cancer treatment.
Hot Spotlight
8 min read
Nav1.8 Inhibitors: The Next-Generation Non-Opioid Analgesics Reshaping Pain Management
17 June 2025
Nav1.8 inhibitors represent a breakthrough in pain therapeutics, offering a non-addictive alternative to opioids.
Bio Sequence
8 min read
Chemical Modifications and Consequences: A Deep Dive into ASO Safety Profiles
17 June 2025
Explore the safety challenges of ASO drugs, and learn how chemical modifications and targeted delivery strategies can mitigate risks while preserving therapeutic efficacy in precision medicine.